Signalment:
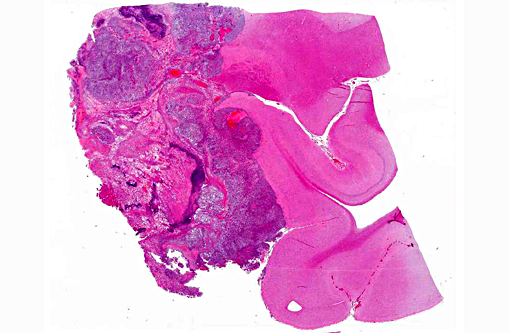
Gross Description:
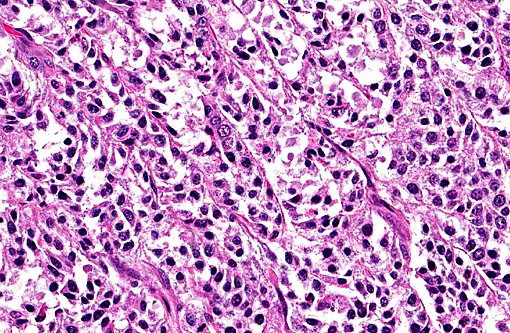
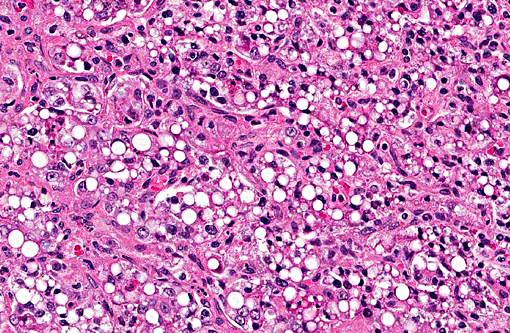
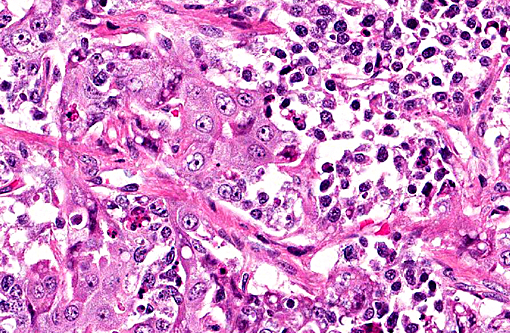
Histopathologic Description:
The primary cytoplasm. Occasionally hepatoid type cells have apical brush borders. The nuclei are large, vesicular, irregularly round, central or peripherally located with finely stippled chromatin and one or two nucleoli. The second population of cells is smaller (round and epithelial type) and they are scattered throughout the neoplasm. These cells have scant eosinophilic cytoplasm, indistinct cell borders and round nuclei with clumped chromatin. Mitotic figures range from 3-5 per 400X field in some areas. There is marked anisocytosis and anisokaryosis, and individual cell necrosis. There are extensive areas of necrosis characterized by cellular and eosinophilic debris, mild hemorrhage, degenerate neutrophils, plasma cells and MOTT cells interspersed between neoplastic cells. Within the adjacent gray and white matter, there is multifocal moderate gliosis with occasional glial nodules, satellitosis and scattered neuronal necrosis.
Morphologic Diagnosis:
Lab Results:
Condition:
Contributor Comment:
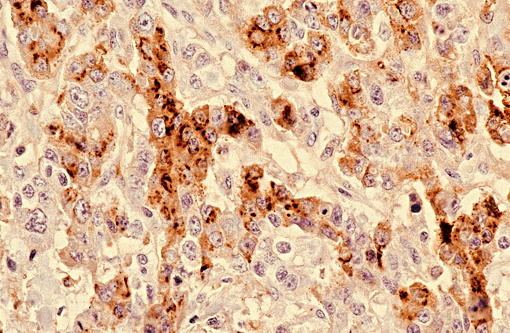
Immunohistochemically, these tumors express alphafetoprotein, vimentin and keratin. Alphafetoprotein is a positive marker for germ cell tumors in humans and dogs, and is produced by yolk sac tumors, enteric elements of teratomas and some embryonal carcinomas.(6) The diagnosis of the suprasellar germ cell tumor is based on three criteria: 1) midline suprasellar location, 2) presence within the tumor of several distinct cell types (histomorphology), and 3) positive staining for alphafetoprotein, VM and CK. Within the sellar region, the WHO classification of tumors from the nervous system of domestic animals has four tumors that include pituitary adenoma, pituitary carcinoma, craniopharyngioma and suprasellar germ cell tumor.(4)
The neoplasm was considered as a suprasellar germ cell tumor as a primary differential based on the pleomorphism of the neoplastic cells with different patterns and gross location. However, alphafetoprotein, which is an important marker for this type of tumor, was unavailable in the laboratory. In addition, this tumor was highly cellular and invasive with extensive areas of necrosis and frequent mitotic figures. Anaplastic meningioma was also considered as differential diagnosis since the microscopic location is associated with the meninges, the tumor has features of malignancy and the neoplastic cells are CK and VM positive. However, meningiomas are usually present in middle aged to older dogs. Furthermore, anaplastic astrocytoma and craniopharyngioma should also been considered since they share several histomorphological features with suprasellar germ cell tumor. Craniopharyngioma has areas resembling ameloblastoma and usually the mitotic rate is low.
JPC Diagnosis:
Conference Comment:
The differential diagnosis list for neoplasms in the suprasellar region were discussed including craniopharyngioma. This tumor arises from remnants of Rathkes pouch.
Craniopharyngiomas are composed of polygonal to columnar cells arranged in solidly cellular areas but may also be seen arranged in cysts or tubules. These tumors generally have areas of squamous differentiation and ciliated cells lining cystic spaces, neither of which were present in the tumor in this case. They can have multifocal areas of necrosis, and cholesterol crystals may be seen. Pituitary adenoma and pituitary carcinoma were also discussed. Pituitary adenomas are composed of polygonal to spindle shaped cells which can be arranged in solidly cellular areas as well as a sinusoidal pattern, and they lack the characteristic of multiple cell types as seen in the tumor in this case. The nuclei in pituitary adenomas are vesiculate, similar to the tumor in this case, but the cells have a moderate to abundant amount of granular cytoplasm. Pituitary carcinomas are similar to adenomas but are more invasive with greater cellular atypia and higher mitotic rate.(4)
Grossly, suprasellar germ cell tumors are grey-white in color, located on midline and usually obscure the pituitary and compress overlying neuroparenchyma. Pituitary tumors are white to brown in color and can be quite large, also compressing the adjacent neuroparenchyma.(4) Craniopharyngiomas are also large tumors that grow along the ventral brain, but may extend dorsally into the neuroparenchyma.(5) Each of these is a reasonable gross differential diagnosis for a mass located in the most ventral region of the brain, on midline and caudal to the optic chiasm.
References:
1. Barnhart KF, Wojcieszyn J, Storts RW. Immunohistochemical staining patterns of canine meningiomas and correlation with published immunophenotypes. Vet Pathol. 2002; 39:311-321.
2. Ferreira AJA, Peleteiro MC, Carvalho T, Correia JMJ, Shulman FY and Summers BA. Mixed germ cell tumor of the spinal cord in a young dog. J Small Anim Pract. 2003; 44(2):81-84.
3. Hoei-Hansen CE, Sehested A, Juhler M, Y-FC Lau, Skakkebaek NE, Laursen H and Rajpert-De Meyts E. New evidence for the origin of intracranial germ cell tumors from primordial germ cells: expression of pluripotency and cell differentiation markers. J. Pathology. 2006; 209 (1):25-33.
4. Koestner A, Bilzer T, Fatzer R, Schulman FY, Summers BA, Van Winkle TJ. Histological Classification of Tumors of the Nervous System of Domestic Animals. 2nd series. Vol. V. Washington DC: Armed Forces Institute of Pathology; 1999:30-36.
5. Koestner A, Higgins RJ. Tumors of the nervous system. In: Meuten DJ, ed. Tumors Cytoplasm of the epithelial-like cells strongly positive for alpha fetoprotein. (anti-AFP, 200X) in Domestic Animals. 4th ed. Ames, IA: Blackwell Publishing Professional; 2002:623-626,728.
6. Patterson-Kane JC, Shulman FY, Santiago N, McKinney L and Davis CJ. Mixed germ cell tumor in the eye of a dog. Vet Pathol. 2001; 38(6):712-714.
7. Summers BA, Cummings and de Lahunta A. Tumors of the central nervous system. In: Summers BA, Cummings and de Lahunta A, eds. Veterinary Neuropathology. St. Louis, MO: Mosby; 1995:384-
385.
8. ACVP meeting, neuropathology mystery slides, case # 5. Presented by Dr. Amanda Fales-Williams (Iowa State University).