Wednesday Slide Conference 2022-2023
Conference 15
Case III:
Signalment:
13-month-old, female-intact, Sprague-Dawley rat, Rattus norvegicus
History:
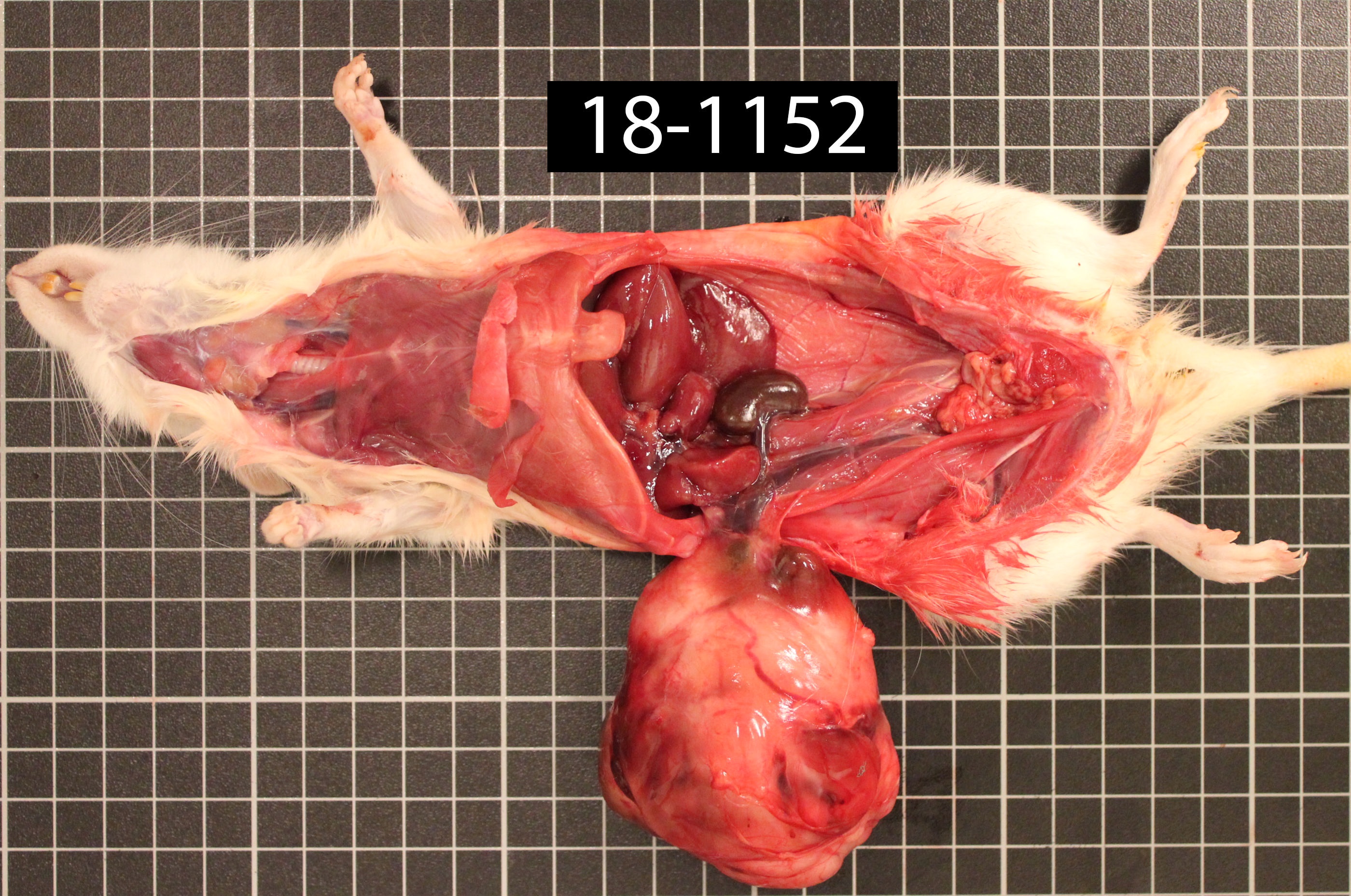
Sentinel rat, presented with abdominal distention. On physical examination, the clinician palpated a freely moveable, large, firm mass within the peritoneal cavity.
Gross Pathology:
The animal is underconditioned (2/5 score). The right kidney is 2.834 g, the left kidney is 122.0 g. The mass is surrounded but not adhered to omentum. Approximately 90% of the right kidney is replaced by a 7 x 5.5 x 5 cm, discrete, encapsulated, semi-firm, pale-tan to brown to red, multilobular mass. On cut section, the mass is composed of smooth, homogeneous, pale-tan regions surrounding a friable, dark-red to brown core.
Laboratory Results:
Immunohistochemistry and special stains:
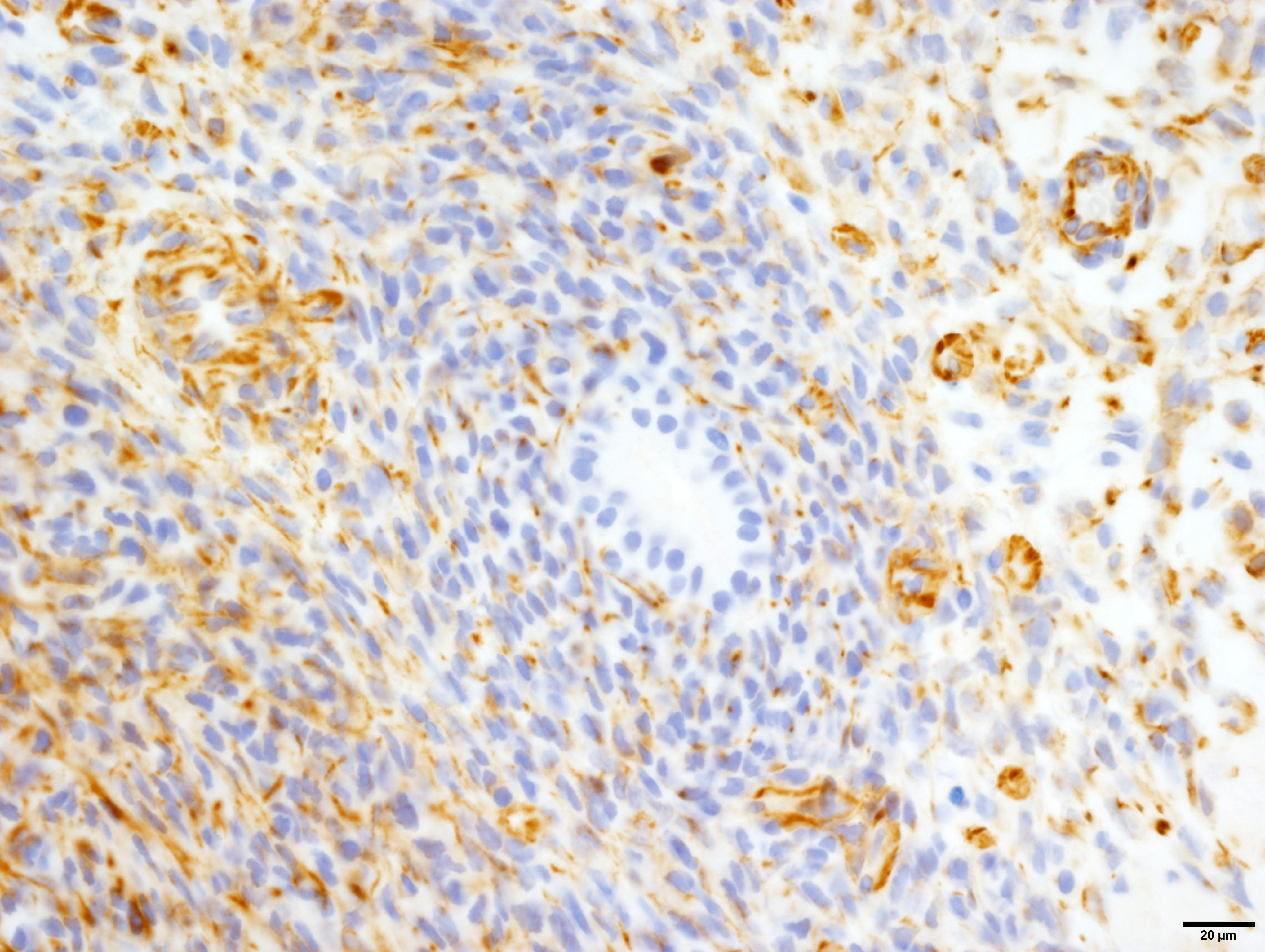
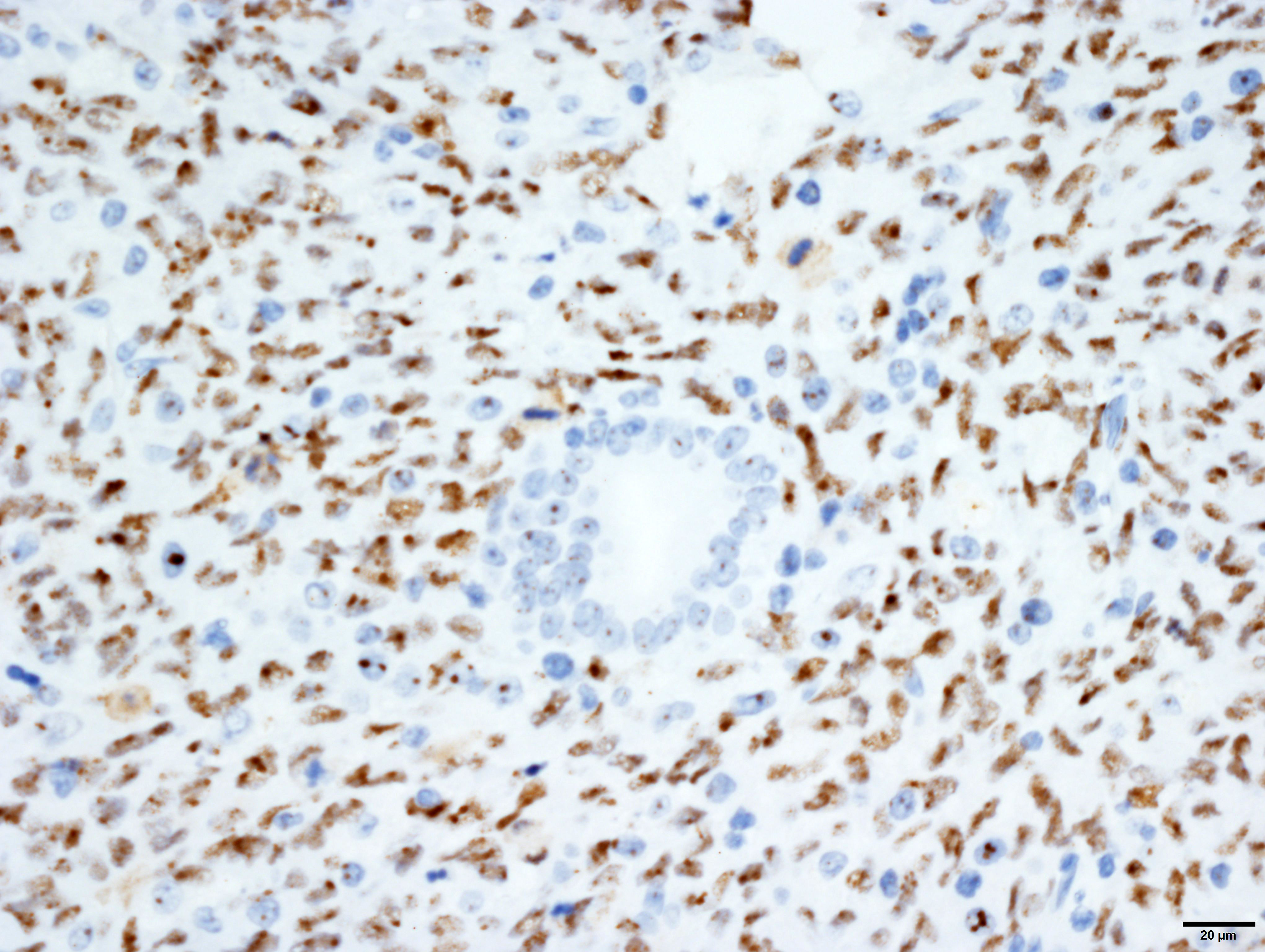
Vimentin: Strong, diffuse, cytoplasmic immunoreactivity of blastemal and stromal neoplastic populations. The primitive tubular epithelial structures are negative.
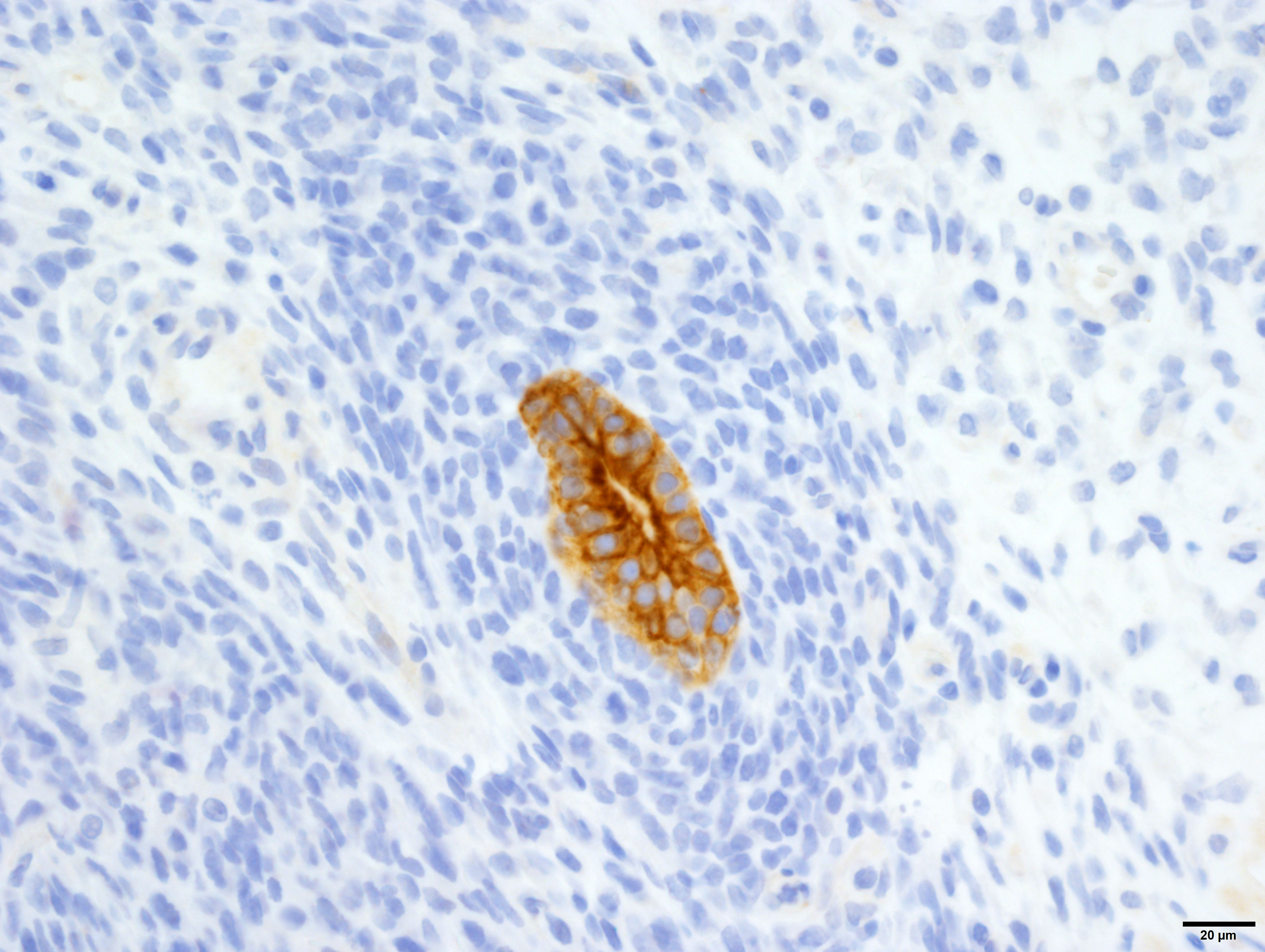
Cytokeratin wide-spectrum screening (WSS): Strong, diffuse, cytoplasmic immunoreactivity of tubular epithelial neoplastic cell populations. The blastemal and stromal components negative.
Wilms tumor protein (WT1): Moderate, diffuse, nuclear immunoreactivity of the blastemal with variable immunoreactivity of the stromal neoplastic population and rare, equivocal nuclear immunoreactivity of a few tubular structures.
N.b. Normal renal tubular and glomerular epithelium also has moderate, variable nuclear immunoreactivity.
Masson’s Trichrome: Masson’s trichrome confirms the presence of collagen within the tumor population (blue staining), and suggests differentiation into muscle (red staining).
Microscopic Description:
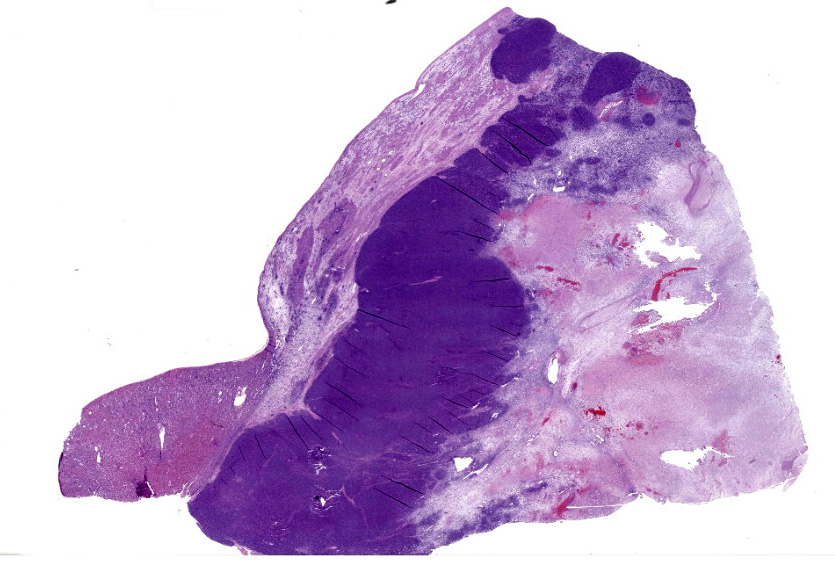
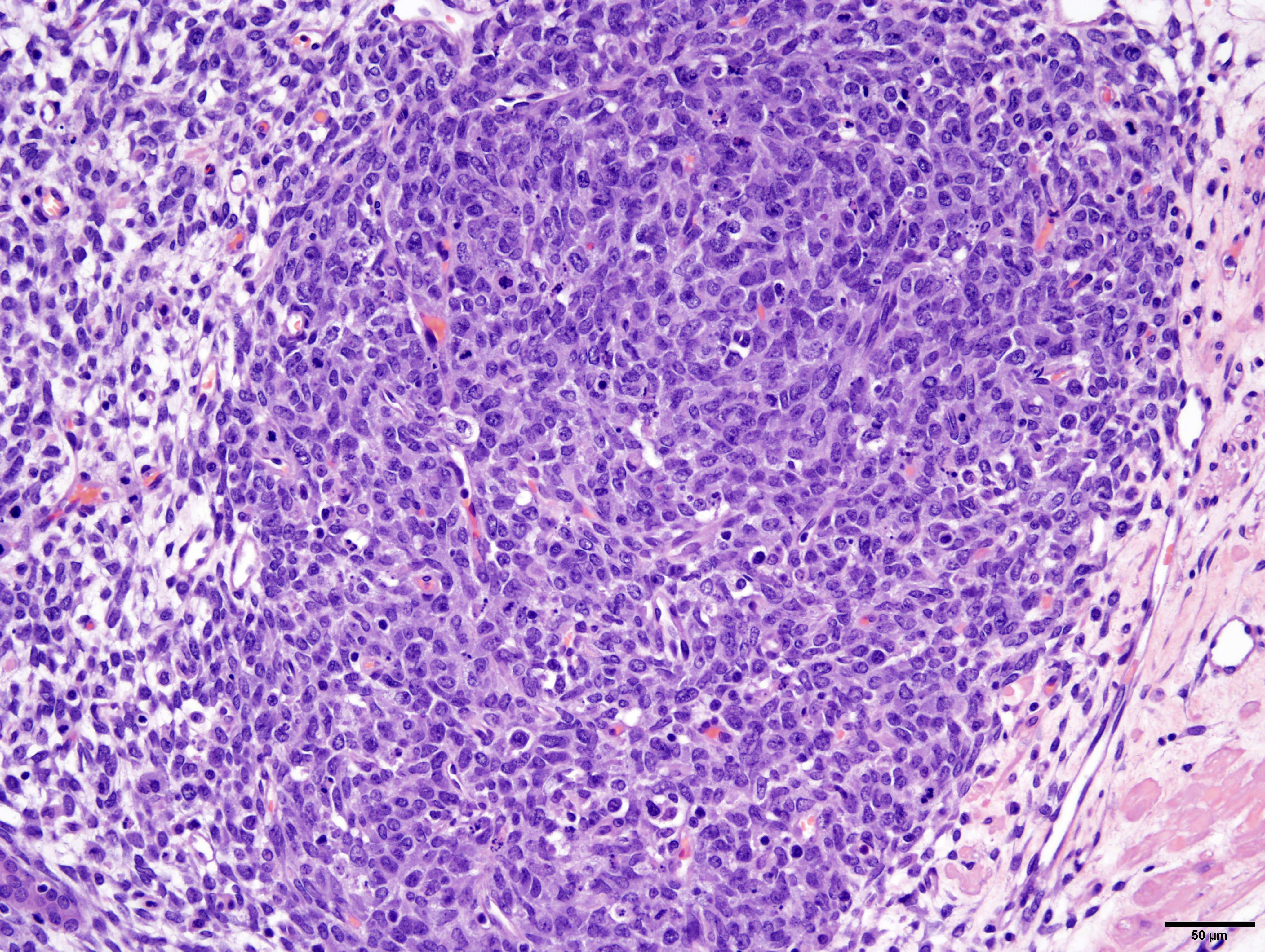
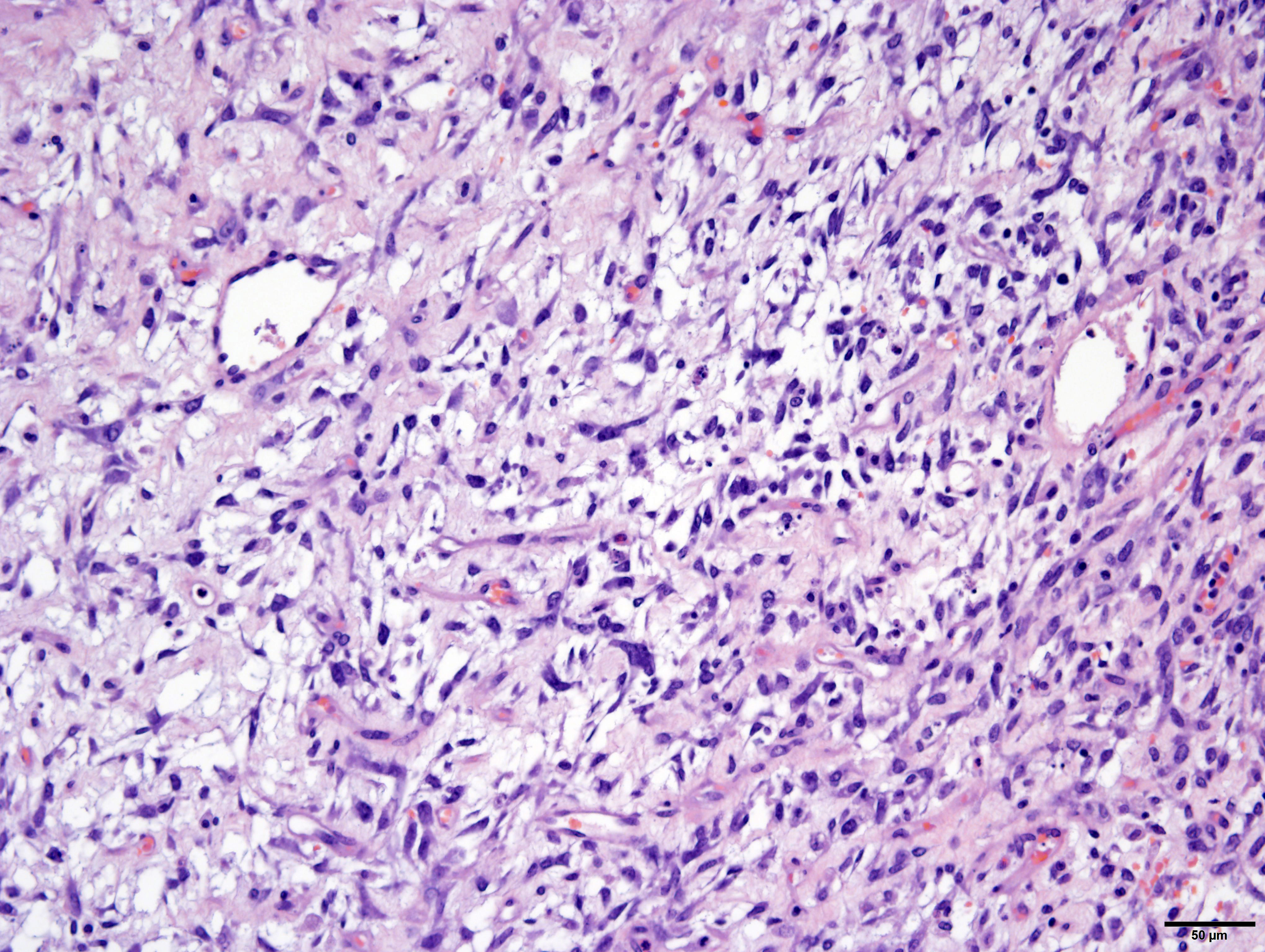
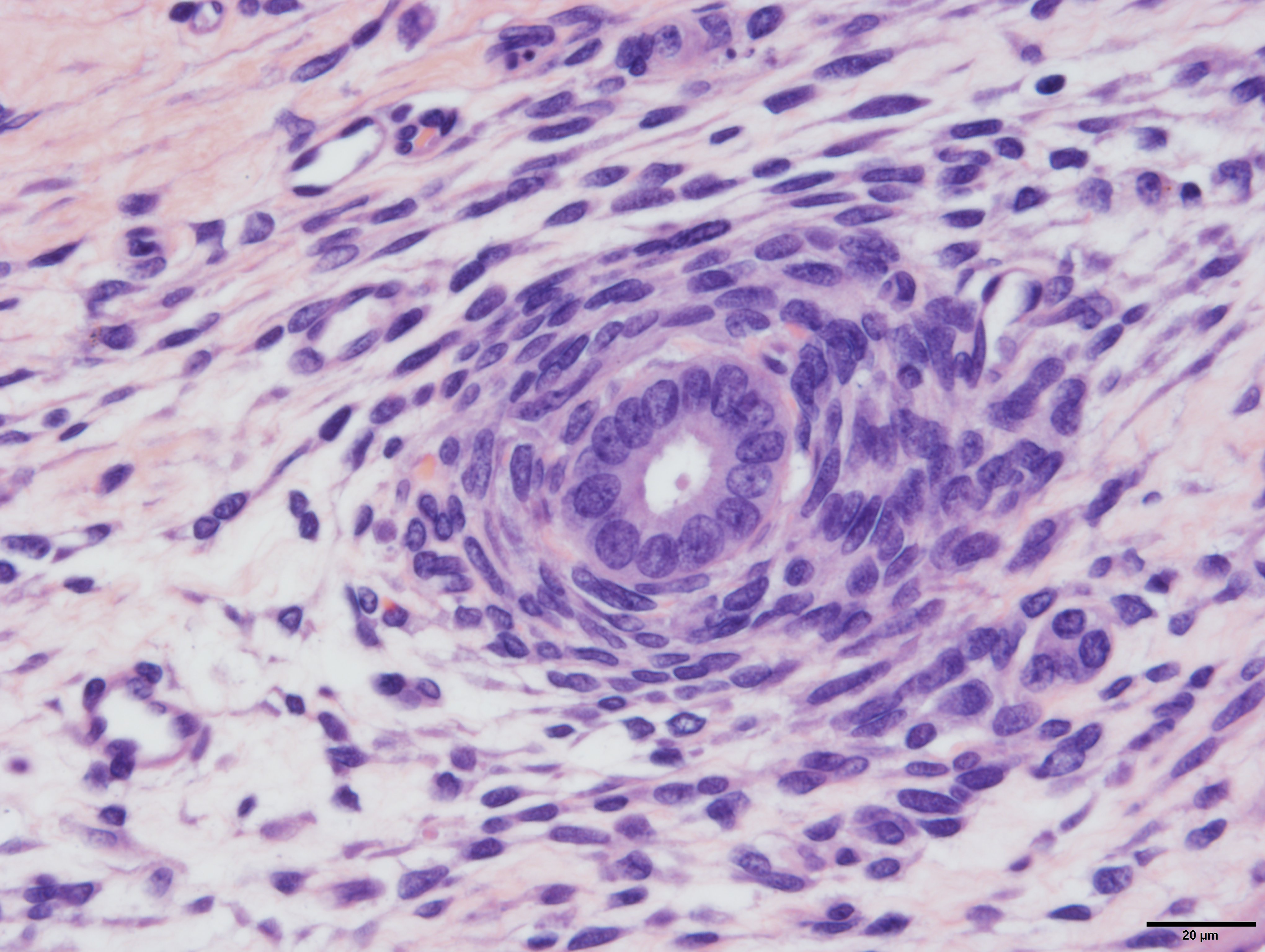
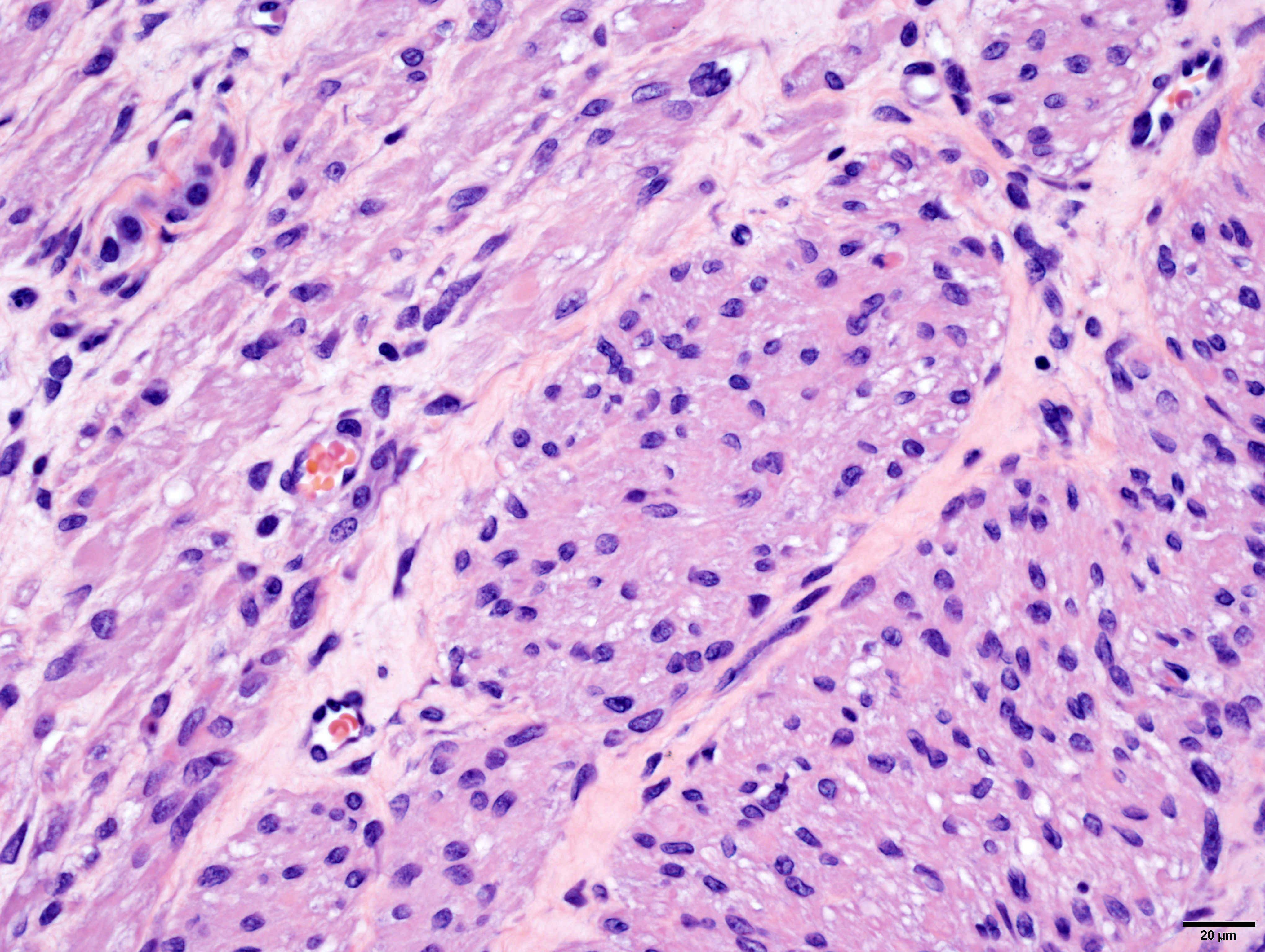
Arising from, compressing, and replacing approximately 50% of the renal parenchyma is a partially encapsulated, multilobular, ill-demarcated, expansile and densely cellular neoplasm that extends to cut borders. The neoplasm is composed of three haphazardly organized cell populations: epithelial, stromal and blastemal. The epithelial component is characterized by cuboidal to columnar cells which form tubules rimmed by a few layers of blastemal cells. The stromal population is composed of fusiform cells arranged in bundles and are supported by a loose fibrovascular stroma. Multifocally, there is differentiation of the mesenchymal cells into collagen, striated and smooth muscle. The blastemal population is composed of dense streams of basophilic polygonal cells with scant cytoplasm, and indistinct cell borders. Anisocytosis and anisokaryosis are mild. The mitotic count in the blastemal population is high, but low in the other populations. The mass has large lakes of necrosis, hemorrhage, edema, fibrin and rare mineralization, admixed with large numbers of degenerate and non-degenerate neutrophils, macrophages, and lesser numbers of lymphocytes and plasma cells. Multifocally, vessels are partially occluded by fibrin thrombi. At the margins of the mass, scattered within the stromal population, are small numbers of misshapen, partially developed and variably sclerotic glomeruli, occasionally surrounded by an ectatic Bowman’s space. Adjacent to these, there are small numbers of variably sized tubules, which are not surrounded by a rim of blastemal cells.
Contributor’s Morphologic Diagnoses:
Kidney: Triphasic nephroblastoma with striated and smooth muscle differentiation (syn: Wilms Tumor)
Contributor’s Comment:
A nephroblastoma, also known as a Wilms tumor (WT), is an undifferentiated, embryonal, mesodermal tumor with multipotent differentiation capabilities, that is thought to arise from the primitive renal stem cell.14 It is the most common renal tumor in childhood.9 In human pediatric patients, it is associated with germline and/or somatic mutations of the WT1 gene, but other genes are thought to be involved as well3. Precursor lesions of WT are so called nephrogenic rests and are defined as “a focus of abnormally persistent nephrogenic cells, meaning cells that can be induced to form a Wilms’ tumor”.3
Histologically, WT present with three distinct cell types: epithelial, stromal and blastemal. All three elements are not required for diagnosis, but when all are present, the term “triphasic” is used. Heterologous components, including collagen, smooth muscle, skeletal muscle, cartilage, and bone may also be present. Zhuang et al. showed that the blastemal, epithelial, stromal and other heterologous components of WT all have identical genetic changes, suggesting all features of this tumor are neoplastic.21
In laboratory animals, nephroblastomas have been frequently reported in rats, but are infrequent in mice and non-human primates.4,6 In rats, nephroblastomas occur both experimentally and spontaneously. Experimentally, they can be induced by chemical administration of N-ethyl-N-nitrosurea or N-methyl-N-nitrosourea, both alkylating agents.20 Spontaneously, they have been reported in Sprague-Dawley and F344 rats, with only rare reports of metastases to the lymph nodes and lungs.5,18 No metastases were identified in our case.
The diagnosis of triphasic WT was based on the presence of blastemal, epithelial and stromal components. Primitive tubular structures, surrounded by a rim of blastemal cells were identified, however, there was no evidence of primitive glomeruloid structures. In addition, there was widespread differentiation of the stromal components into striated and smooth muscle, and collagen. The diagnosis of WT was aided by immunoreactivity of both blastemal and stromal populations with WT1, vimentin and immuno-negativity for cytokeratin WSS. The epithelial component was positive for cytokeratin, equivocally immunoreactive for WT1 and negative for vimentin. However, from our search, the veterinary literature is inconclusive regarding immunohistochemical profiles of these tumors, especially regarding the epithelial component. This may in part be due to the challenge in differentiating the epithelial population from the blastemal population, the choice of antibody, or the up/downregulation of certain proteins at different cellular developmental stages. In our case, the identification of epithelial components was simplified by the presence of discrete tubular structures, which were immunoreactive with cytokeratin WSS.
The main differential diagnosis for a WT is a renal mesenchymal tumor (RMT). RMTs arise from multipotential spindle-shaped mesenchymal cells, and, like WTs, can also give rise to heterologous components, such as smooth or skeletal muscle, fibrous tissue, cartilage and/or bone.17 Further misleading pathologists, profiles of hyperplastic tubules are frequently found in RMTs, but these are considered preexisting entrapped renal tubules, rather than newly formed. The pathognomonic feature of WTs is the population of blastemal cells, which are immediately apparent in our case.
Finally, the significance of multiple, individual, partially developed, and variably sclerotic glomeruli, as well as variably sized tubules within the stromal population at the margins of the tumor is not determined. We suspect these may represent normal renal structures that were entrapped and separated from the renal parenchyma at the early onset of neoplasia. However, we cannot exclude that they may have arisen de novo.
Contributing Institution:
Laboratory of Comparative Pathology; Hospital for Special Surgery, Memorial Sloan Kettering Cancer Center, The Rockefeller University, Weill Cornell Medicine.
https://www.mskcc.org/research-areas/programs-centers/comparative-medicine-pathology
JPC Diagnosis:
Kidney: Nephroblastoma.
JPC Comment:
Nephroblastomas are the most common renal neoplasms in pigs, chickens, and fish.13 In pigs, they generally exhibit a benign behavior.13 Nephroblastomas also occur in a variety of other veterinary species, including dogs, cats, bovine fetuses, Japanese eels, guanacos, cottontail rabbits, and budgerigars.1,8,10,16 In recent literature, a primary nephroblastoma was reported in the nasopharynx of a 3-month-old Boer goat, and unilateral stromal-type nephroblastomas were reported in the kidneys of two hedgehogs.2,19
In dogs, nephroblastomas are the third most common renal neoplasm, accounting for approximately 5% of renal neoplasia.13 Up to 50% of canine nephroblastomas metastasize, and potential sites include the contralateral kidney, lung, liver, mesenteric lymph nodes, and spinal cord.7 A recent report also described the first documented case of gingival metastasis in an 8 year old miniature Pinscher.7 Primary spinal nephroblastomas also occur in young dogs, arising from nephrogenic rests between the dura and spinal cord, typically in the thoracolumbar region.12 A recent report in a 1-year-old male American pitbull terrier documented multifocal spinal nephroblastomas arising in the thoracolumbar region, cervical intumescence, sacral segment, and cauda equina.11 The authors believed that neoplastic seeding of the CNS resulted in the multifocal distribution, because vascular invasion was not observed but neoplastic cells expanded Virchow-Robbins spaces (which are continuous with the subarachnoid space).11
Differential diagnoses discussed by the contributor included renal cell carcinoma, renal mesenchymal tumor, amphophilic vacuolar tumor, renal sarcoma, and liposarcoma; diagnostic criteria for the tumors can be reviewed on the Global Open Registry Nomenclature Information System at goRENI.org.
References:
- Agnew D. Camelidae. In: Terio KA, McAloose D, St. Leger J, eds. Pathology of Wildlife and Zoo Animals. San Diego, CA: Elsevier. 2018; 193.
- Athey JM, Rice LE, Harvey AB, Washburn KE, Rodrigues-Hoffman A. Nasopharyngeal nephroblastoma in a 3-month-old Boer goat. J Vet Diagn Invest. 2021; 33(1):108-111.
- Beckwith J, Bruce NB, Bonadio K. Nephrogenic rests, nephroblastomatosis, and the pathogenesis of Wilms' tumor. Pediatr Pathol. 1990; 10:1-36.
- Castiglioni V, De Maglie M, Queliti R. Immunohistochemical characterization of a renal nephroblastoma in a trp53-mutant and prolyl isomerase 1-deficient mouse. J Toxicol Pathol. 2013; 26:423-427.
- Chandra M, Carlton WW. Incidence, histopathologic and electron microscopic features of spontaneous nephroblastomas in rats. Toxicol lett. 1992; 62: 179-190.
- Chandra M, Riley MGI, Johnson DE. Spontaneous renal neoplasms in rats. J Appl Toxicol. 1993; 13:109-116.
- Chen B, Li W, Wang F. A blastema-predominant canine renal nephroblastoma with gingival metastasis: case report and literature review. J Vet Diagn Invest. 2018; 30(3): 430-437.
- Delaney MA, Treuting PM, Rothenburger JL. Lagamorpha. In: Terio KA, McAloose D, St. Leger J, eds. Pathology of Wildlife and Zoo Animals. San Diego, CA: Elsevier. 2018; 486.
- Dumba M, Jawad N, McHugh K. Neuroblastoma and nephroblastoma: an overview and comparison. Cancer Imaging. 2014; 14:O15.
- Frasca Jr S, Wolf JC, Kinsel MJ, Camus AC, Lombardini ED. Osteichthyes. In: Terio KA, McAloose D, St. Leger J, eds. Pathology of Wildlife and Zoo Animals. San Diego, CA: Elsevier. 2018; 961.
- Henker LC, Bianchi RM, Vargas TP, de Olveira EC, Driemer D, Pavarini SP. Multifocal Spinal Cord Nephroblastoma in a Dog. J Comp Path. 2018; 158: 12-16.
- Higgins RJ, Bollen AW, Dickinson PJ, Siso-Llonch S. Tumors of the Nervous System. In: Meuten DJ, ed. Tumors in Domestic Animals. 5th ed. Ames, Iowa: Iowa State Press; 2017
- Meuten DJ, Meuten TLK. Tumors of the urinary system. In: Meuten DJ, ed. Tumors in Domestic Animals. 5th ed. Ames, Iowa: Iowa State Press; 2017:646-649.
- Pritchard-Jones K. Malignant origin of the stromal component of Wilms’ tumor. J Natl Cancer Inst. 1997; 89:1089–1091.
- Pritchard?Jones K, Vujanic G. Multiple pathways to Wilms tumor: how much is genetic?. Pediatr Blood Cancer.2006; 47:232-234.
- Reaville DR, Dorrenstein G. Psittacines, Coliiformes, Musophagiformes, Cuculiformes. In: Terio KA, McAloose D, St. Leger J, eds. Pathology of Wildlife and Zoo Animals. San Diego, CA: Elsevier. 2018; 784.
- Seely JC. Renal mesenchymal tumor vs nephroblastoma: revisited. J Toxicol Pathol. 2004; 17: 131-136.
- Tanaka, N, Takeshi I, Jyoji Y. Spontaneous nephroblastoma with striated muscle differentiation in an F344 rat. J Toxicol Pathol. 2017; 30:231-234.
- Ueda K, Imada T, Ueda A, Imada M, Ozaki K. Stromal-type Nephroblastoma with or without Anaplasia in Two Hedgehogs. J Comp Path. 2019; 172: 48-52.
- Yoshizawa K, Kinoshita Y, Emoto Y. N-Methyl-N-nitrosourea-induced Renal Tumors in Rats: Immunohistochemical Comparison to Human Wilms Tumors. J Toxicol Pathol. 2014; 26:141-148.
- Zhuang Z, Merino MJ, Vortmeyer AO. Identical genetic changes in different histologic components of Wilms’ tumors. J Natl Cancer Inst. 1997; 89:1148-52.