Signalment:
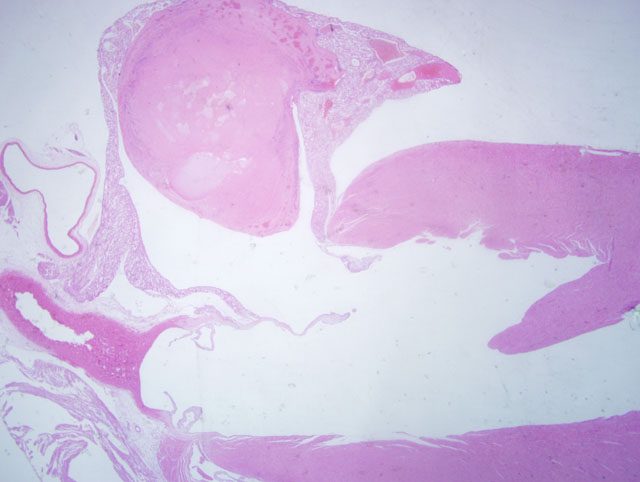
Gross Description:
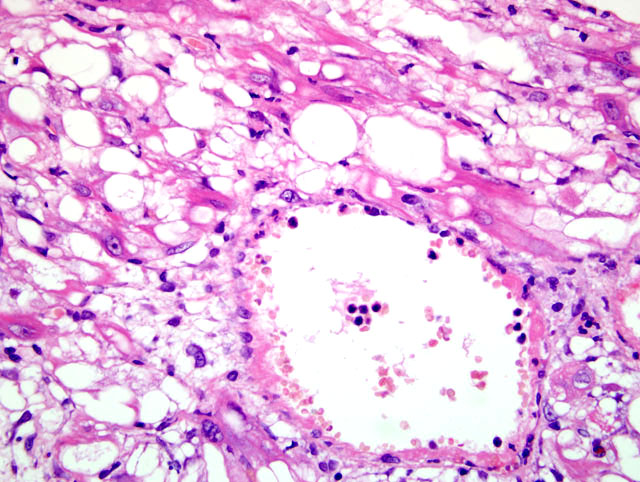
Histopathologic Description:
Additional histologic lesions in these rats referable to doxorubicin administration are present in the kidneys, testes, epididymides and lungs. Kidney lesions included tubular degeneration/ regeneration, glomerular atrophy and vacuolation and hyperplasia/ hypertrophy of Bowmans capsule. Testicular lesions consisted of germ cell loss, with only Sertoli cells remaining within seminiferous tubules in severe cases. A secondary oligospermia was present within the epididymides. Within the lung there was degeneration, with prominent vacuolation, of the muscular media in large arteries, sometimes accompanied by inflammation and fibrinoid necrosis.
Morphologic Diagnosis:
1. Heart: Multifocal cardiac myocyte degeneration with prominent vacuolation, and rare necrosis.
2. Heart, left atrium: Atrial thrombosis and neutrophilic endocarditis, with intralesional bacteria.
Condition:
Contributor Comment:
The doxorubicin-induced lesions in this study increased in incidence and/or severity with increasing duration of dosing. Both the cumulative effect and the morphologic characteristics of the lesions were consistent with findings reported in the literature. The myocardial degeneration induced in the ventricles and septum in this study in rats was representative of that reported in mice in the literature; however, atrial lesions were much more severe. A similar pattern was described previously in mice treated with doxorubicin,(3) although the bulk of the literature describing heart lesions caused by doxorubicin is based on examination of transverse tissue sections taken through the midventricular and septal areas, which do not include evaluation of the atria. In mice, vacuolation and degeneration of atrial myocytes are shown with electron microscopy to be dilation of the sarcoplasmic reticulum and increased numbers of normal and/or degenerate mitochondria.(3) Atrial interstitial inflammatory cell infiltrates within the myocardium and endocardium are also reported.(3) Additionally, mice treated with doxorubicin show an incidence of atrial thrombosis approaching 75%.(3) The proposed cause of the atrial thrombosis in the mice was endothelial inflammation accompanied by abnormal blood flow secondary to the myocardial damage.(3)
JPC Diagnosis:
Conference Comment:
Discussion then focused on the secondary effects of the histologic lesions in the atria. Residents concluded that the primary lesion is the myocardial change, which results in ineffective myocardial contraction, endothelial damage, release of prothrombotic substances and thrombosis. Three perturbations promoting thrombus formation, colloquially referred to as Virchows triad, are endothelial injury, altered normal blood flow (turbulence or stasis), and hypercoagulability.(4) Endothelial cell damage is not restricted to physical damage; any damage capable of disrupting the prothrombotic-antithrombotic balance favoring thrombosis is included in this category.(4)
Normal, non-activated endothelial cells have antithrombotic activity; activation or damage resulting from bacterial endotoxin, cytokines or changes in hemodynamic properties promote thrombus formation. The following chart summarizes the mechanisms for these actions:(4)
| Antithrombotic Properties of Endothelial Cells | Prothrombotic Properties of Endothelial Cells | ||
| Antiplatelet effects |
| Platelet effects | -�-� Endothelial injury exposes subendothelial ECM â platelet adherence via von Will |
| Anticoagulant Effects | -�-� Heparin-like molecules enhance thrombin inactivation via antithrombin III: 1. Thrombomodulin â binds thrombin â activates protein C â inactivates factors Va and VIIIa â inhibits clotting 2. Produces protein S and tissue factor pathway inhibitor â direct inhibition of factor VIIa (tissue factor) and factor Xa |
Procoagulant effects | -�-� Cytokine (TNF or IL-1) or endotoxin â endothelial production of tissue factor â activation of the extrinsic clotting cascade -�-� Augment the catalytic function of activated factor IXa and Xa |
| Fibrinolytic effects | -�-� Produce tissue plasminogen activator â cleaves plasminogen to plasmin â leaves fibrin â thrombus dissolution | Antifibrinolytic effects | -�-� Secrete inhibitors of plasminogen activator â reduces fibrinolysis |
Platelets play an equally important role in hemostasis. Once contact is made with extracellular matric (ECM) proteins, platelets have three functions:(4)
- Adhere to collagen with glycoprotein Ib via vWF; additional glycoprotein receptors bind other ECM components.
- Secretion of α- and dense-granule contents promoting thrombus formation. P-selectin-coated α-granules contain fibrinogen, fibronectin, platelet factor 4, PDGF, TGF-β, and factors V and VIII. Dense (δ) granules have ionized calcium, histamine, serotonin, epinephrine, ADP and ATP.
- Platelets aggregate and change shape, forming the primary hemostatic plug.
References:
2. Ayaz SA, Bhandari U, Pillai KK. Influence of DL α-lipoic acid and vitamin-E against doxorubicin-induced biochemical and histological vhanges in the cardiac tissue of rats. Indian J Pharmacol. 2005;37(5):294-299.
3. Fujihira S, Yamamoto T, Matsumoto M, et al. The high incidence of atrial thrombosis in mice given doxorubicin. Tox Pathol. 1993;21(4):362-368.
4. Mitchell RN. Hemodynamic disorders, thromboembolic disease and shock. In: Kumar V, Abbas AK, Fausto N, Aster JC eds. Robbins and Cotran Pathologic Basis of Disease. 8th ed. Philadelphia, PA: Elsevier Saunders; 2009:115-123.
5. Speth PAJ, van Hoesel Q, Haanen C. Clinical pharmacokinetics of doxorubicin. Clin Pharm. 1988;15:15-31.