CASE 4: C008 (4153941-00)
Signalment:
6-year-old, female, Rasa Aragonesa, Ovine (Ovis orientalis aries)
History:
The animal was part of an experimental study that aimed to characterize the evolution of arthritis in sheep naturally infected by small ruminant lentiviruses (SRLV). At clinical examination, the animal showed bilateral asymmetric carpal arthritis and mild diffuse increase of respiratory sounds.
Gross Pathology:
At necropsy, the sheep showed body score of 2/5. Carpal joints were swollen and showed marked synovitis and moderate cartilage erosion. Lungs showed rubbery consistence and were diffusely increased in size.
Laboratory results:
The animal was seropositive to small ruminant lentivirus (SRLV, Elitest MVV/CAEV, Hyphen Biomed). Carpal joint qPCR (EXOone Maedi Visna ? CAEV, Exopol) were performed on carpal swaps and the results were Ct: 35,07 and Ct: 32,47, respectively for each carpi (considered positive if Cq?38). qPCR against Mycoplasma agalactiae, Chlamydia abortus, Streptococcus dysgalactiae, Erysipelothrix rhusiopathiae were negative.
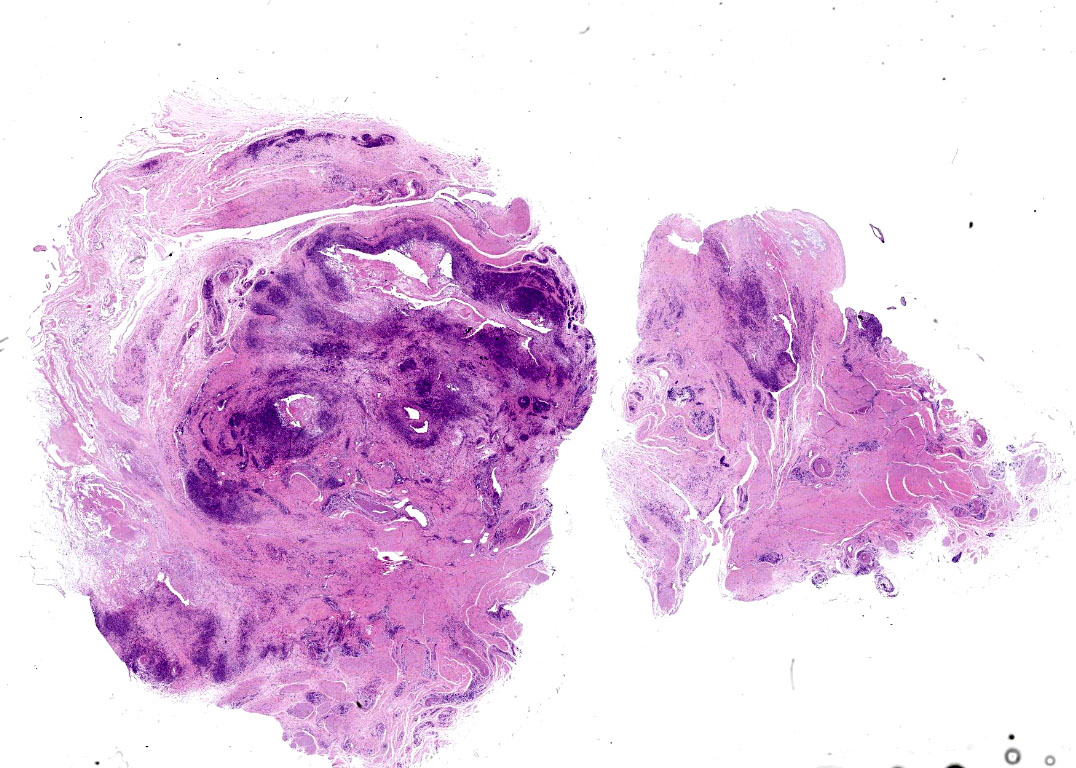
Microscopic description:
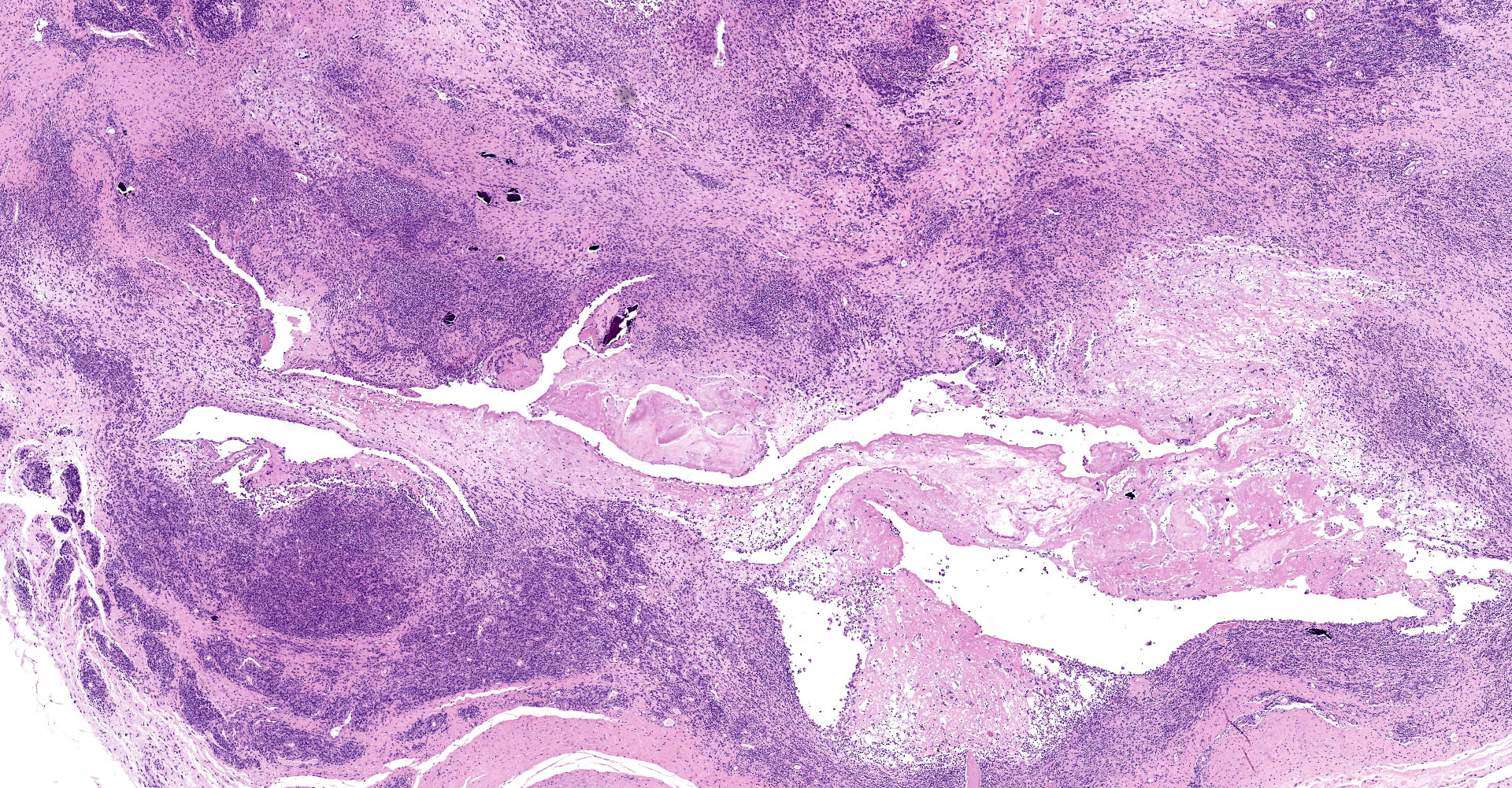
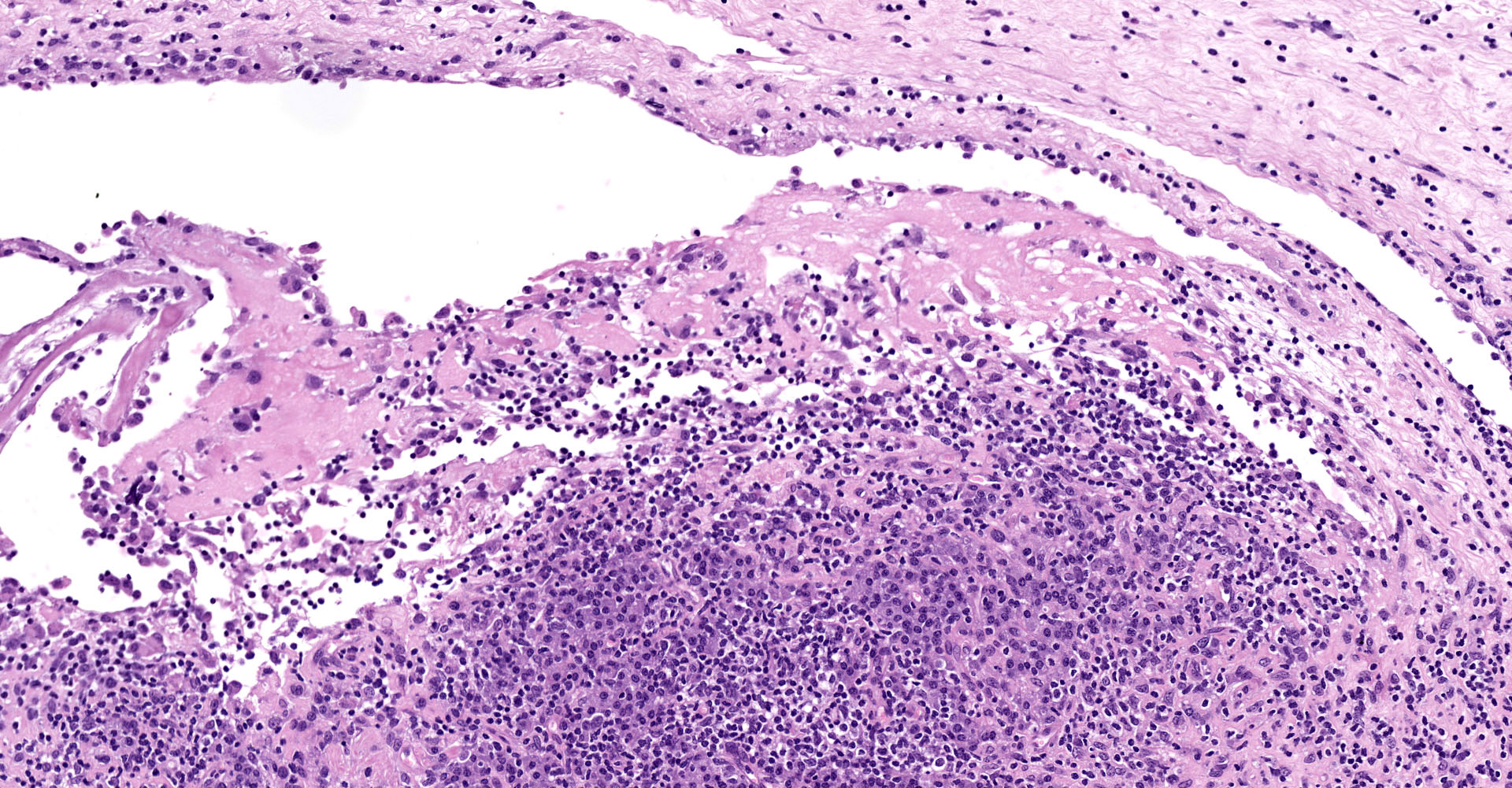
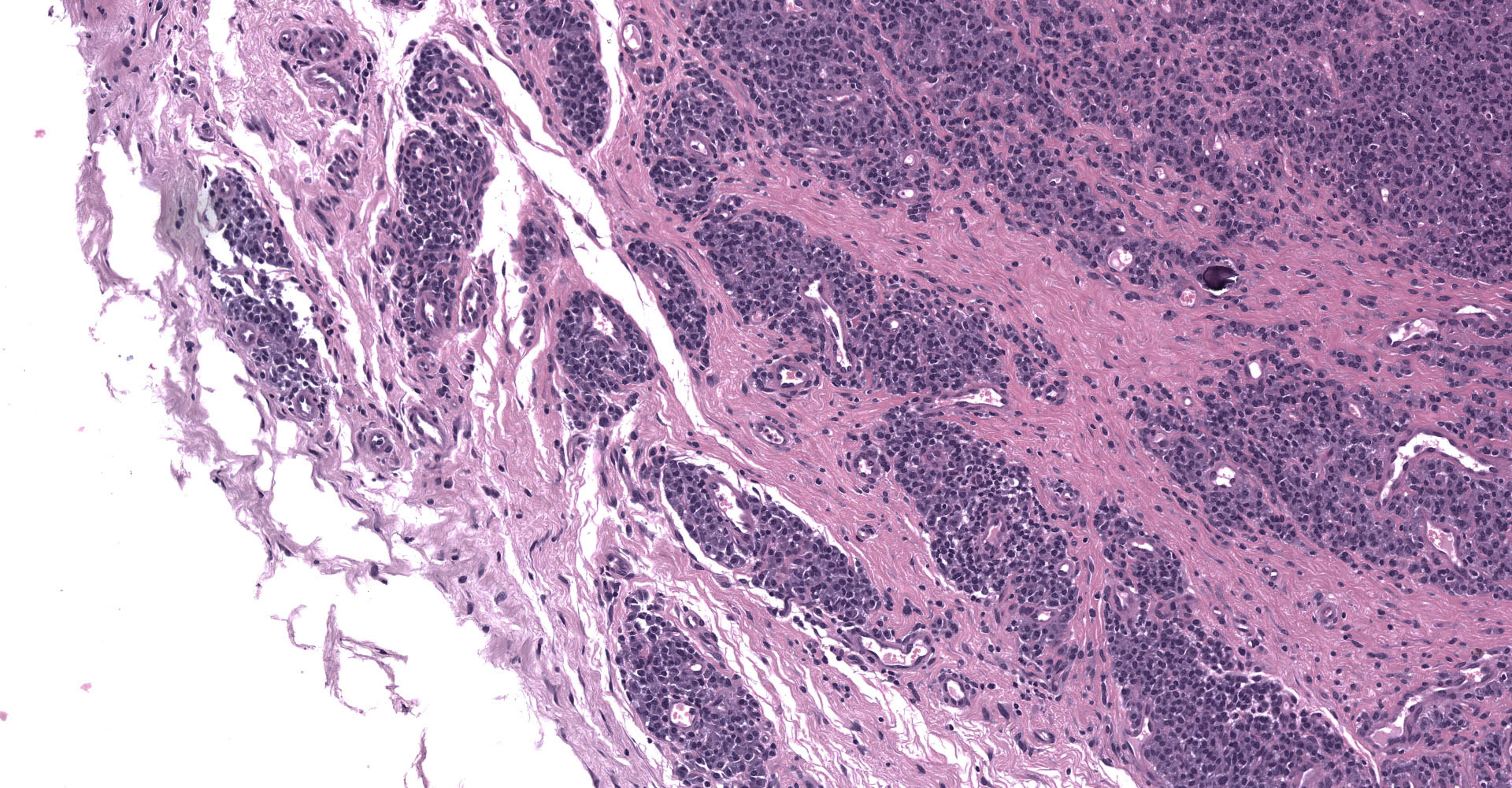
Joint capsule, synovia. Diffusely expanding the stroma there is an inflammatory process associated with moderate synovial membrane villar proliferation. Stroma in thickened (up to 1 cm) by abundant lymphocytes, plasma cells and macrophages occasionally arranged in lymphoid-like structures and rare neutrophils admixed with increased numbers of plump fibroblast and matures collagen fibers (fibrosis) and proliferation of neovessels. Additionally, some macrophages contain abundant brown-golden intracytoplasmic pigment (hemosiderin) and there are multifocal areas of basophilic mineral deposition (calcification). Stromal arteries show marked thickening of the tunica media with smooth muscle hypertrophy and reactive endothelium and lymphatics are dilated. Diffusely, synovial membrane is thickened (up to 3 times) and show fingerprint projections towards the lumen lined by swollen synoviocytes with finely vacuolated cytoplasm (hypertrophy) that occasionally piles up (hyperplasia). Additionally, there are multifocal areas of synoviocyte loss (erosions and ulcers) and replacement by moderate amounts of fibrillary eosinophilic polymerized material (fibrin), occasional cellular debris and rare neutrophils.
Contributor's morphologic diagnosis:
Joint capsule, synovia. Lymphoplasmacytic synovitis. Diffuse. Severe. Chronic. With synovial hypertrophy and hyperplasia and stromal fibrosis.
Contributor's comment:
This is the articular form of small ruminant lentiviruses (SRLV), a highly prevalent chronic infection of sheep and goats linked to important deleterious effects in animal production and animal trade restrictions.4
The SRLVs are a group of non-oncogenic exogenous retroviruses with marked tropism for the mononuclear-phagocyte system. SRLV are single stranded RNA viruses with high mutational potential.13 Indeed, four main genotypes (A, B, C, and E) and more than 35 subgroups with significant antigenic heterogeneity have been already characterized.8 These viruses induce chronic and persistent inflammation that usually remains subclinical. When it becomes clinical, the disease can express mainly in four different locations: lung, joints (mainly carpus), central nervous system, and mammary gland.9 The expression of each form as well as the severity of the lesion depends on the host immune response and the viral factors.5,11
The main routes of vertical and horizontal transmission are via colostrum and via inhalation of nasal secretions, respectively.9 The virus infects a variety of cell types (mammary epithelium, fibroblast, endothelial cells, monocytes, choroid plexus) but its replication mainly occurs in mature macrophages.13 Lentiviral infected macrophages produce cytokines that recruit and activate other leukocytes but also enhance lentiviral replication. Indeed, clinical signs and histopathological lesions are the result of the inflammatory process instead of direct viral damage to the organ. There is neither treatment nor commercial vaccines for the disease, what makes challenging the immunization of the ovine population against the virus.14
The articular form, characterized by arthritis, is mainly caused by genotype B2 strains.7,10 It mainly targets carpal joint although tarsal joints may also become affected. Typical gross findings include, serosanguinous joint effusion, thickened joint capsule and tendon sheaths, fibrin attached to synovium, and hygromas (flattened, cystic, subcutaneous distensions over the anterior carpus filled with serosanguinous fluid containing fibrinous or gelatinous masses). In most severe cases, fibrillation and erosion of cartilage can be present. Histologically, lentiviral arthritis is characterized by villous synovial hyperplasia, subsynovial lymphoplasmacytic inflammation with occasional lymphoid follicle formation and stromal fibrosis.
The respiratory form is the most common presentation of the disease in sheep and consists of progressive pneumonia with marked dyspnea and weight loss.2 Lungs are characterized by a rubbery firm texture and lack of collapse when the thorax is opened. Indeed, this animal presented with a diffuse interstitial pneumonia with BALT hyperplasia.
The neurologic form is characterized by demyelinating leukoencephalomyelitis that leads to ataxia and profound weight loss.12 This form progresses faster than the respiratory one and usually occurs in adult sheep and goat kids (2-4 months).
The mammary form is difficult to detect and mainly consists of mammary gland induration, progressive bilateral atrophy, and milk production loss that leads to deficient lamb growth.6,9 Both mammary glands are usually symmetrically affected. Microscopical lesions are characterized by abundant lymphocytes and plasma cells that infiltrate the interstitial and periductal connective tissue and even form lymphoid nodules. In advanced lesions degeneration and loss of acinar and ductal epithelium may happen as a consequence of inflammation rather than direct virus effect.
The main differential diagnosis for this kind of arthritis in the Mediterranean countries are Mycoplasma agalactiae, Chlamydia abortus, Streptococcus dysgalactiae, Erysipelothrix rhusiopathiae.
Contributing Institution:
Universidad de Zaragoza
Departamento de Patología Animal
https://patologiaanimal.unizar.es
JPC diagnosis:
Joint capsule: Synovitis, proliferative and lymphoplasmacytic, chronic, diffuse, marked, with fibrin, synovial ulceration, granulation tissue, and mineralization.
JPC comment:
Numerous countries have active eradication programs for small ruminant lentivirus (SRLV), including Belgium. Recent phylogenetic analysis of the gag-pol region of samples from sheep and goats of various provinces in Belgium showed that 13 of 14 sheep SRLV strains belonged to subtype A1, with 7 of 7 goat SRLV strains belonging to subtype B1. One sheep sample was found to be of the B1 subtype, illustrating the ability of these viruses to cross species and cause disease. Interestingly, analysis of the pol region of sheep all clustered in subtype A1, but three goats clustered in subtype B1, two clustered in subtype A1, and two samples clustered in a new separate cluster within genotype B. The new subtype has been designated subtype B5.8
A robust monitoring and eradication program was tested in Switzerland when a flock of seropositive goats was imported. ELISA and western blot results were consistent and indicated a high prevalence of infection in the new animals. Isolation of the virus was most successful from the spleen, confirmed B1 subtype, and PCR analysis of the gag-pol and env regions revealed a high degree of heterogeneity within viral sequences, indicating multiple infections within single animals, and within the flock. Excellent infrastructure and communication allowed this program to succeed, in large part.3
Numerous aspects of the host immune response have previously been investigated, including Toll-like receptors, antiviral proteins (APOBEC family, tetherin, TRI-M5alpha), and various cytokines, with recent research focused on miRNAs. A total of 212 miRNAs were identified, with 46 highly conserved across species. However, 12 novel miRNAs were identified in infected sheep. Differences were found in expression of these miRNAs between uninfected sheep versus seropositive sheep. However, no differences in miRNA expression were found between asymptomatic seropositive animals and those with clinical disease. While some differential expression of miRNAs has been found, additional research may be warranted.1
References:
1. Bilbao-Arribas M, Abendano N, Varela-Martinez E, Reina R, de Andres D, Jugo B. Expression analysis of lung miRNAs responding to ovine VM virus infection by RNA-seq. BMC Genomics. 2019;20:62.
2. Blacklaws BA. Small ruminant lentiviruses: Immunopathogenesis of visna-maedi and caprine arthritis and encephalitis virus. Comp Immunol Microbiol Infect Dis. 2012;35:259?269.
3. De Martin E, Golomingi A, Zahno ML, et al. Diagnostic response to a cross-border challenge for the Swiss caprine arthritis encephalitis virus eradication program. Schweiz Arch Tierheilkd. 2019;161(2):93-104.
4. Echeverría I, De Miguel R, De Pablo-Maiso L, et al. Multi-Platform Detection of Small Ruminant Lentivirus Antibodies and Provirus as Biomarkers of Production Losses. Front Vet Sci. 2020 Apr 30;7.
5. Gayo E, Polledo L, Balseiro A, et al. Inflammatory Lesion Patterns in Target Organs of Visna/Maedi in Sheep and their Significance in the Pathogenesis and Diagnosis of the Infection. J Comp Pathol. 2018;159:49?56.
6. Gayo E, Polledo L, Magalde A, et al. Characterization of minimal lesions related to the presence of visna/maedi virus in the mammary gland and milk of dairy sheep. BMC Vet Res. 2019 Apr 10;15:109.
7. Glaria I, Reina R, Crespo H, et al. Phylogenetic analysis of SRLV sequences from an arthritic sheep outbreak demonstrates the introduction of CAEV-like viruses among Spanish sheep. Vet Microbiol. 2009 Jul 2;138:156?162.
8. Michiels R, Adjadj NR, De Regge N. Phylogenetic analysis of belgian small ruminant lentiviruses supports cross species virus transmission and identifies new subtype B5 strains. Pathogens. 2020 Mar 1;9.
9. Minguijón E, Reina R, Pérez M, et al. Small ruminant lentivirus infections and diseases. Vet Microbiol. 2015;181:75?89.
10. Pérez M, Biescas E, Reina R, et al. Small Ruminant Lentivirus?Induced Arthritis. Vet Pathol. 2015;52:132?139.
11. Pinczowski P, Sanjosé L, Gimeno M, et al. Small Ruminant Lentiviruses in Sheep: Pathology and Tropism of 2 Strains Using the Bone Marrow Route. Vet Pathol. 2017 May 1;54:413?424.
12. Polledo L, González J, Benavides J, et al. Patterns of Lesion and Local Host Cellular Immune Response in Natural Cases of Ovine Maedi-Visna. J Comp Pathol. 2012;147:1?10.
13. Pritchard GC, McConnel I. Maedi-Visna. In: Aitken ID, ed. Diseases of sheep. Oxford (UK): Blackwell Publishing; 2007:217?223.
14. Reina R, de Andrés D, Amorena B. Immunization against small ruminant lentiviruses. Vol. 5, Viruses. 2013:1948?1963.