CASE 2: S 818/19 FB (4152315-00)
Signalment:
One-day-old, male piglet (Sus scrofa domesticus)
History:
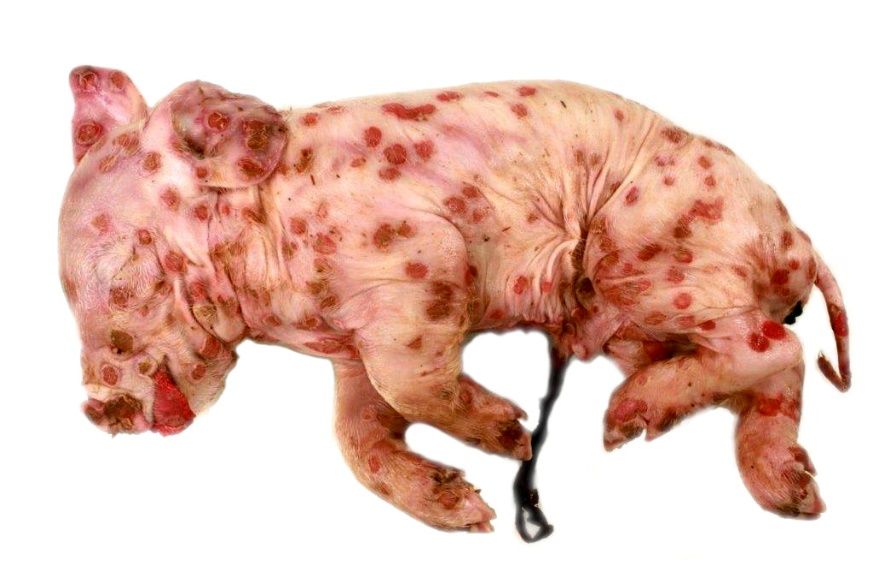
The piglet from a clinically healthy sow was born with multifocal severe skin lesions, affecting the whole body and died spontaneously one day post natum.
Gross Pathology:
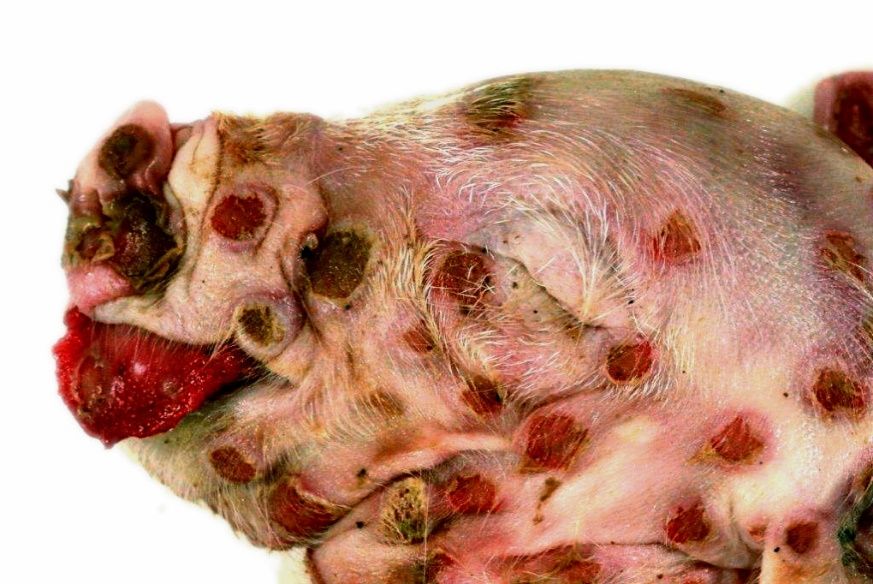
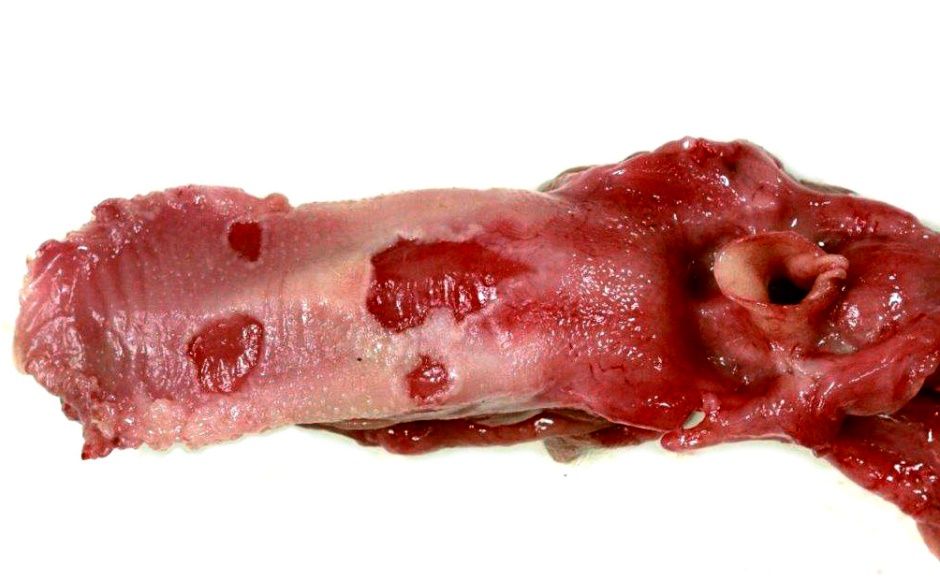
Macroscopically, the animal showed multifocal severe, erythematous maculae covering the whole body as well as the tongue and the cranial part of the esophagus. Lesions appeared as cutaneous and mucosal erythematous, round to coalescing papules with a depressed center and a raised, wall-like border. Partially, encrusted exudate was present on the surface of the alterations.
Laboratory results:
Using PCR, swinepox virus was detected in samples from representative lesions. Microbiologically, a moderate to high amount of Escherichia coli, Proteus sp., as well as coliform germs were isolated from lung, small intestine, liver and skin.
Microscopic description:
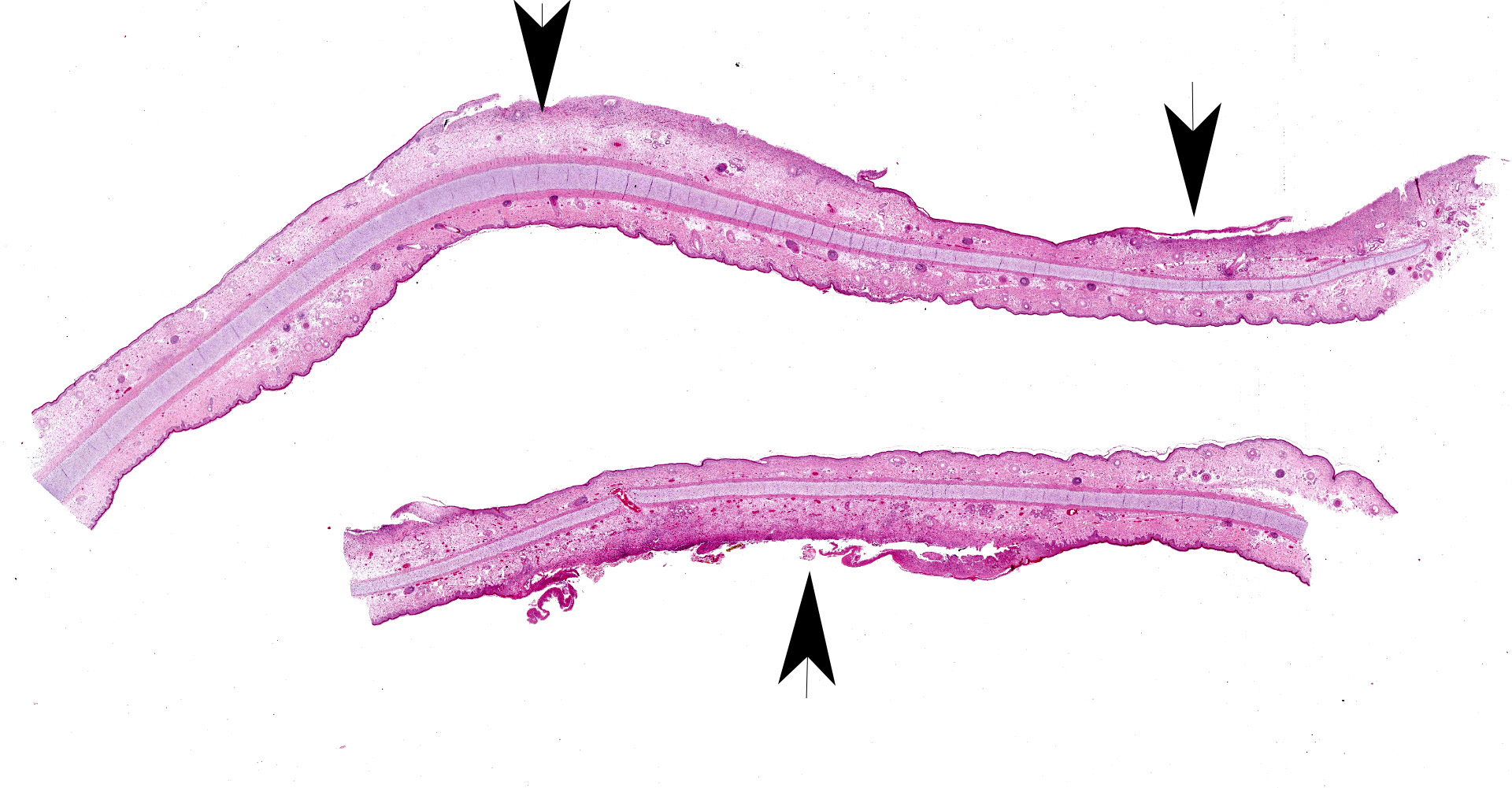
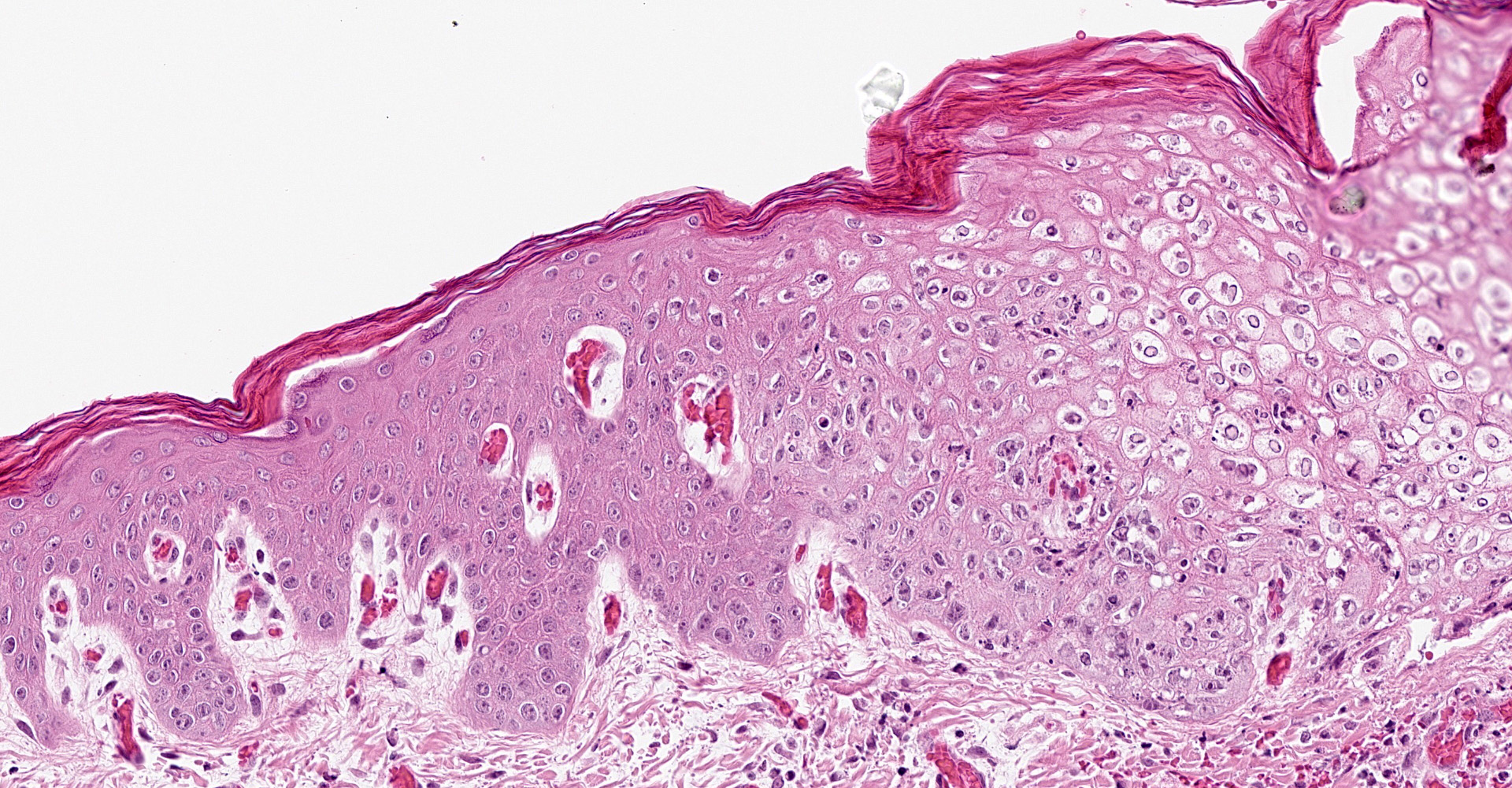
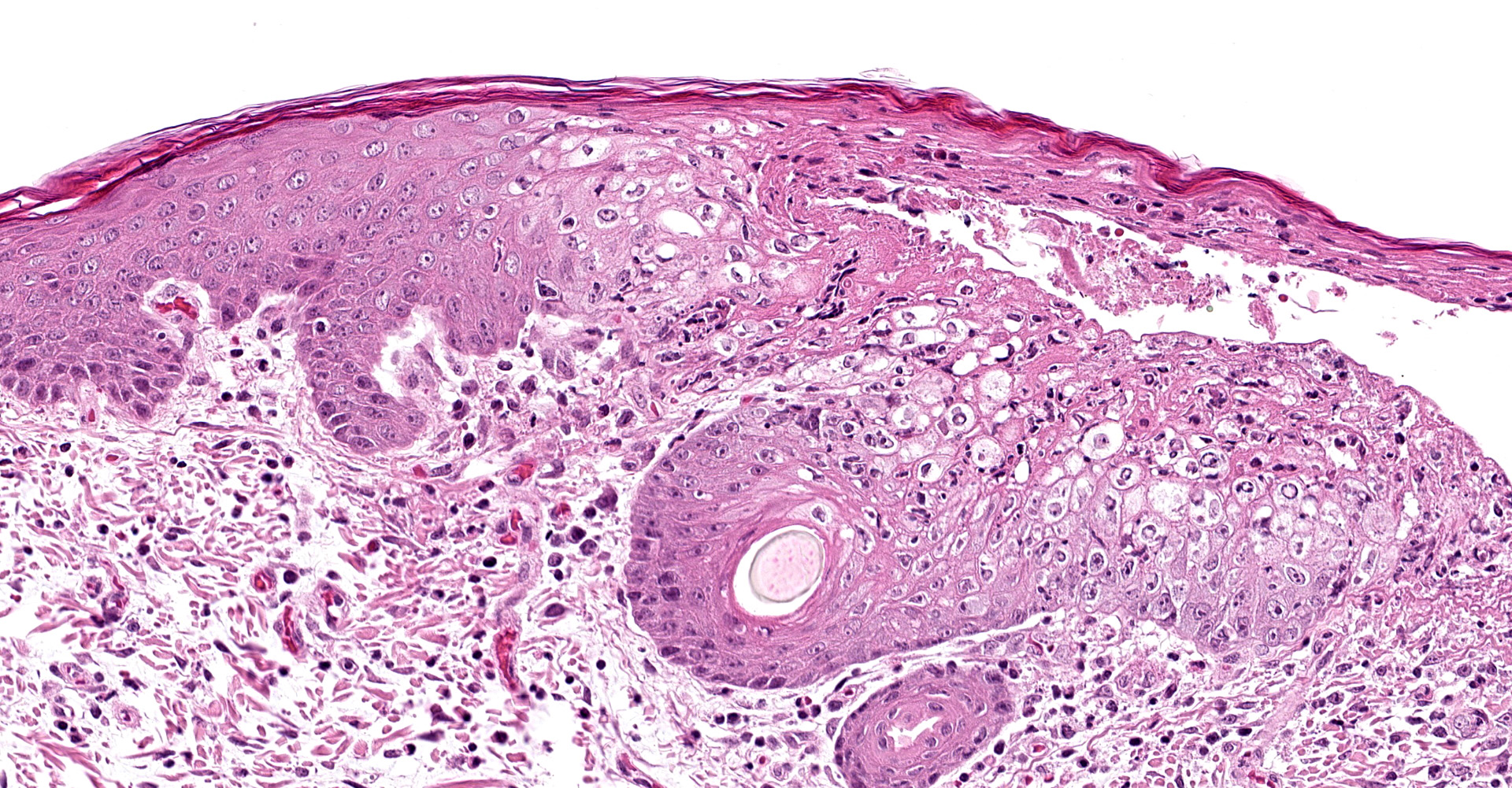
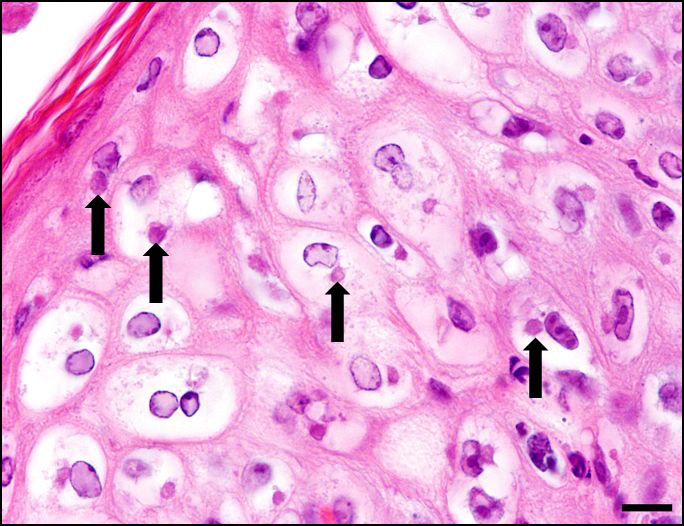
Skin, pinna: there are multifocal complete losses of the epidermis including the basal membrane (ulceration) with adjacent parts of the epidermis detached from the dermis, including areas of keratinocytes that show multifocally cellular shrinkage with nuclei undergoing karyopyknosis, karyorrhexis and karyolysis (necrosis). Multiple areas of the remaining epidermis show a moderate increase of cellular layers (hyperplasia) and cellular swelling with ground glass appearance of the cytoplasm and nuclear enlargement (ballooning degeneration). Within these keratinocytes often eosinophilic, cytoplasmic inclusion bodies are present. In several locations, the underlying dermis is infiltrated by a moderate amount of macrophages, lymphocytes and neutrophils, focally accompanied by large-scale necrosis. In some locations, the inflammatory process involves adnexal structures, and occasionally, some of the hair follicles show similar alterations as the epidermis.
Contributor's morphologic diagnosis:
Dermatitis, multifocal, severe, subacute, proliferative and ulcerative with ballooning degeneration of keratinocytes and cytoplasmic inclusion bodies; adnexal structures: folliculitis, multifocal, moderate, lympho-histiocytic and necrotizing with ballooning degeneration of keratinocytes and cytoplasmic inclusion bodies.
Contributor's comment:
The swinepox virus is the only member of the genus Suipoxvirus in the family Poxviridae and the subfamily Chordopoxvirinae.5,7 It occurs worldwide, is endemic in regions with intensive swine production, and is particularly associated with poor sanitary conditions.7,8,9 Pigs are the only natural hosts, though earlier studies have shown that experimental infection in rabbits is possible.3 The resistant virus persists in dried crusts from infected animals for up to a year.5,7,12 Virus transition occurs either by direct contact or by mechanical vectors, such as the blood sucking swine louse (Haematopinus suis),5,7 mosquitoes or biting flies,12 assisting infection by causing skin trauma.5,7
In affected herds, the morbidity may reach high levels.9 Predominantly, young growing piglets are affected but some authors also described lesions in neonates, which raises the theory of an intrauterine infection.5,7,10 A viremic stage of the disease has not yet been reported, although transplacental infection suggests its occurrence.12 The presented case of a one-day-old piglet supports an intrauterine infection because the histological investigation revealed reactive inflammatory changes indicative for a lesion duration of more than one day.
Usually, the course of disease is mild with low mortality, so the virus has caught rather little attention in the past.5,7 The severity is often age-related, as some authors report more severe alterations in younger piglets.9,10 After an incubation period of about seven days,13 the macroscopic changes typically appear on the ventral and lateral abdomen, lateral thorax, as well as the medial forelegs, and the thighs. They show the typical pattern of pox infections, beginning as erythematous papules that transform into umbilicated pustules.5,7
In contrast to the pathogenesis of poxvirus infections in other species, a vesicular stage is insignificant, though some authors reported the occurrence of vesicles in individual animals that have experimentally been infected.1,3,5,7 After healing, a white macule may remain.5,7 Sometimes the alterations are more prominent on the back.5,7 A periodic sequence of lesion development may result in multiple stages of pox lesions, that can be found simultaneously.13
Secondary dermatitis may accompany the virus-induced alterations.8,9 In cases of severe infections, the lesions may turn out to be generalized and affect alongside the integument the mucosa of the gastrointestinal tract from oral cavity to the stomach as well as the trachea and bronchi.5,7,10 Cytoplasmic inclusion bodies may be found frequently in keratinocytes of the stratum spinosum,1,3,9 however, even intranuclear inclusion have been reported sporadically.1 As etiological differential diagnosis vaccinia virus infection has to be considered as the morphological changes are quite similar to swinepox induced changes.3,7 Infection of pigs with the swinepox virus does not prevent infection with vaccinia virus.13
Contributing Institution:
Department of Pathology
University of Veterinary Medicine Hannover
Buenteweg 17
D-30559 Hannover
Germany
http://www.tiho-hannover.de/kliniken-institute/institute/institut-fuer-pathologie/
JPC diagnosis:
Ear, pinna, haired skin, epidermis and follicular epithelium: Dermatitis, proliferative and necrotizing, multifocal, moderate, with ballooning degeneration and intracytoplasmic viral inclusions.
JPC comment:
The contributor provides a good summary of swinepox. While the primary transmission routes are direct contact or through natural vectors, there are potential methods of transmission that are not often considered. When large ships travel oceans, in order to provide stability during the voyage, and during loading and unloading operations, they have ballast water. Vast quantities of ballast water are taken into tanks while the ship is in port, which is then pumped between different tanks for balance, and often discharged in a destination port. This transported water reservoir is the potential medium of delivering invasive species, as well as infectious organisms. A characterization of ballast water that had arrived in Busan, South Korea, a variety of viruses were found, including swinepox virus in ballast water from a port in Panama. There were a variety of other concerning viruses as well, including suid herpesvirus 1, Raccoonpox virus, a variety of bacteriophages, and a host of viruses that typically infect marine invertebrates, amoebas, and algae.6
Pox viruses have host specificity, with little spillover from the host specific viruses such as swinepox virus, sheeppox virus, myxoma virus, and yaba monkey tumor virus. Protein kinase R (PKR) is a normal and critical part of the host immune response and limits viral replication in an infected cell by phosphorylating eukaryotic initiation factor 2a (eIF2a). Pox viruses produce proteins E3 and K3 that antagonize PKR and allow viral activity within cells. E3 is a dsDNA binding protein that inhibits PKR activation, but K3 acts as an eIF2a analog substrate for PKR, limiting activation of eIF2a. Using vaccinia virus, E3 and K3 genes were deleted, which eliminated the virus's ability to replicate. Subsequently, E3 and K3 orthologs from host specific pox viruses were expressed in the modified vaccinia virus, and it was found that the expressed K3 orthologs restored the ability of the virus to replicate in host specific cells. This suggests that the K3 family proteins confer host specificity to these viruses.2
Because the swinepox virus genome is so large (146kb), is host specific, and usually causes a self-limiting form of the disease, it has become an ideal candidate for vaccine development. Introducing additional DNA (transgenes) into the genome is easily performed, and even multiple transgenes can be accommodated for multivalent vaccine development.4 A variety of recombinant vaccines have been investigated, using swinepox as the delivery mechanism, such as Streptococcus suis, porcine circovirus, swine influenza virus, swine infectious gastritis virus, and a number of other pathogens.11 This virus is likely to remain a useful tool for vaccine development.
References:
- Blakemore F, Abdussalam M. Morphology of the elementary bodies and cell inclusions in swine pox. J Comp Pathol. 1956;66:373?377.
- Cao J, Varga J, Deschambault Y. Poxvirus encoded eIF2a homolog, K3 family proteins, is a key determinant of poxvirus host species specificity. Virology. 2020;541:101-112.
- Datt NS. Comparative studies of pig pox and vaccinia viruses. I. Host range pathogenicity. J Comp Pathol. 1964;74:62?68.
- Fan HJ, Lin HX. Recombinant swinepox virus for veterinary vaccine development. In: Brun A, ed. Vaccine Technologies for Veterinary Viral Diseases: Methods and Protocols, Methods in Molecular Biology. New York, NY: Springer. 2016:163-175.
- Hargis AM, Ginn P. Viral infections. In: McGavin MD, Zachary JF. Pathologic Basis of Veterinary Disease. 4th ed. St. Louis: Mosby Elsevier, 2006:1175?1176.
- Hwang J, Park SY, Lee S, Lee TK. High diversity and potential translocation of DNA viruses in ballast water. Marine Pollution Bulletin. 2018;137:449-455.
- Mauldin EA, Peters-Kennedy J. Viral diseases of skin. In: Maxie G, ed. Jubb, Kennedy and Palmer`s Pathology of Domestic Animals. 6th ed. Vol. 1. St. Louis: Elsevier, 2016:616 ?625.
- Medaglia ML, Pereira A, Freitas TR, Damaso CR. Swinepox virus outbreak, Brazil. Emerg Infec Dis. 2011;17:1976?978.
- Olinda RG, Maia LA, Cargnelutti JF, et al. Swinepox dermatitis in backyard pigs in Northeastern Brazil. Pesq Vet Bras. 2016;36(6):468?472.
- Thibault S, Drolet R, Alain R, et al. Congenital swine pox: A sporadic skin disorder in nursing piglets. In: Swine Health Prod. 1998;6(6):276?278.
- Yuan X, Lin H, Li B, He K, Fan H. Swinepox virus vector-based vaccines: attenuation and biosafety assessments following subcutaneous prick inoculation. Vet Res. 2018;49:14.
- https://vetmed.iastate.edu/vdpam/FSVD/swine/index-diseases/pox
- https://www.msdvetmanual.com/integumentary-system/pox-diseases/swinepox