CASE 1: 20-003333 (4155466-00)
Signalment:
8 month old, male-intact, Huacaya, Vicugna pacos, alpaca
History:
The cria presented for a simple, closed, spiral fracture of the distal left humerus which was repaired with surgical intervention. Following surgery, the cria was severely hypoglycemic and seizing. Despite intensive therapy, the cria died a few days later.
Gross Pathology:
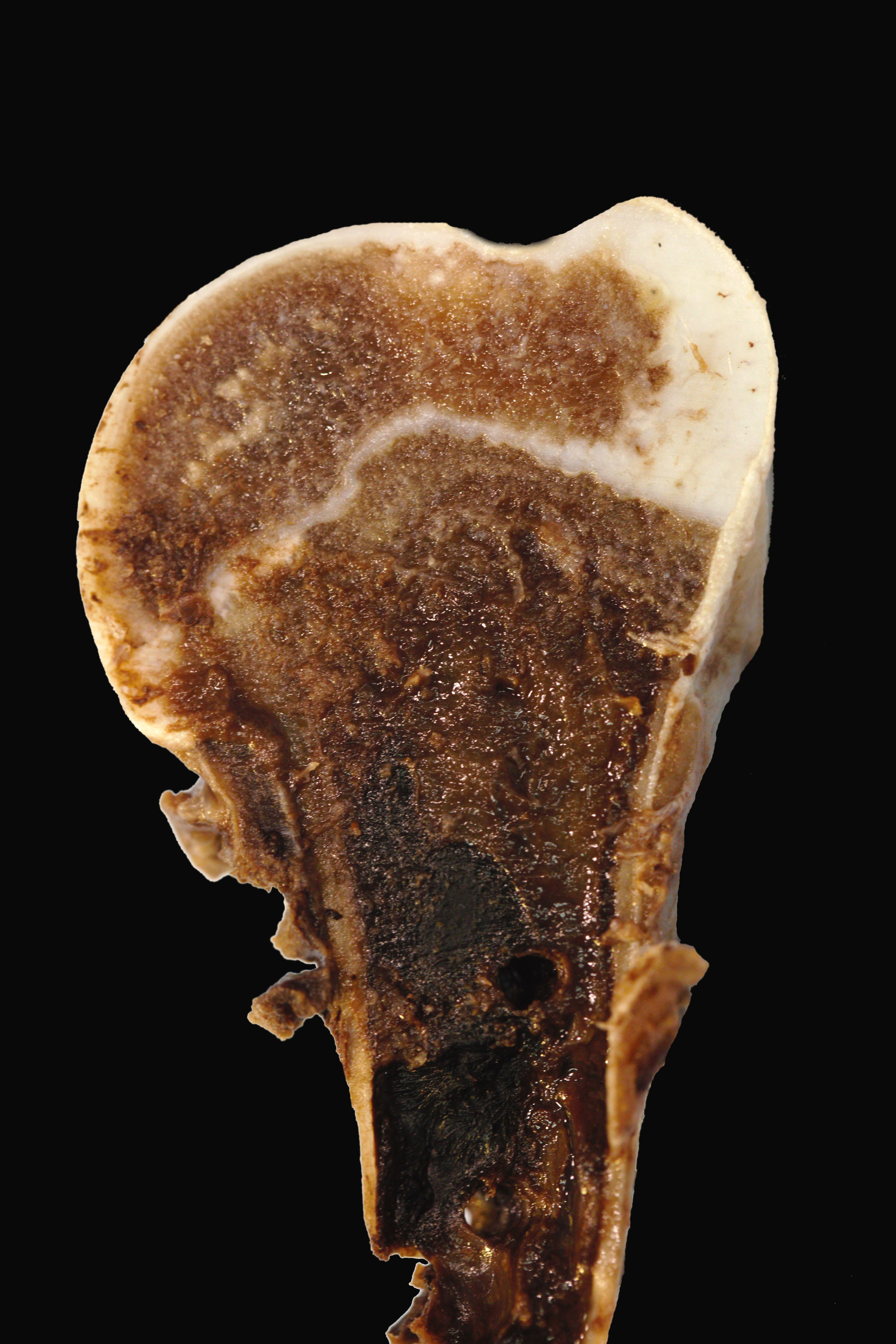
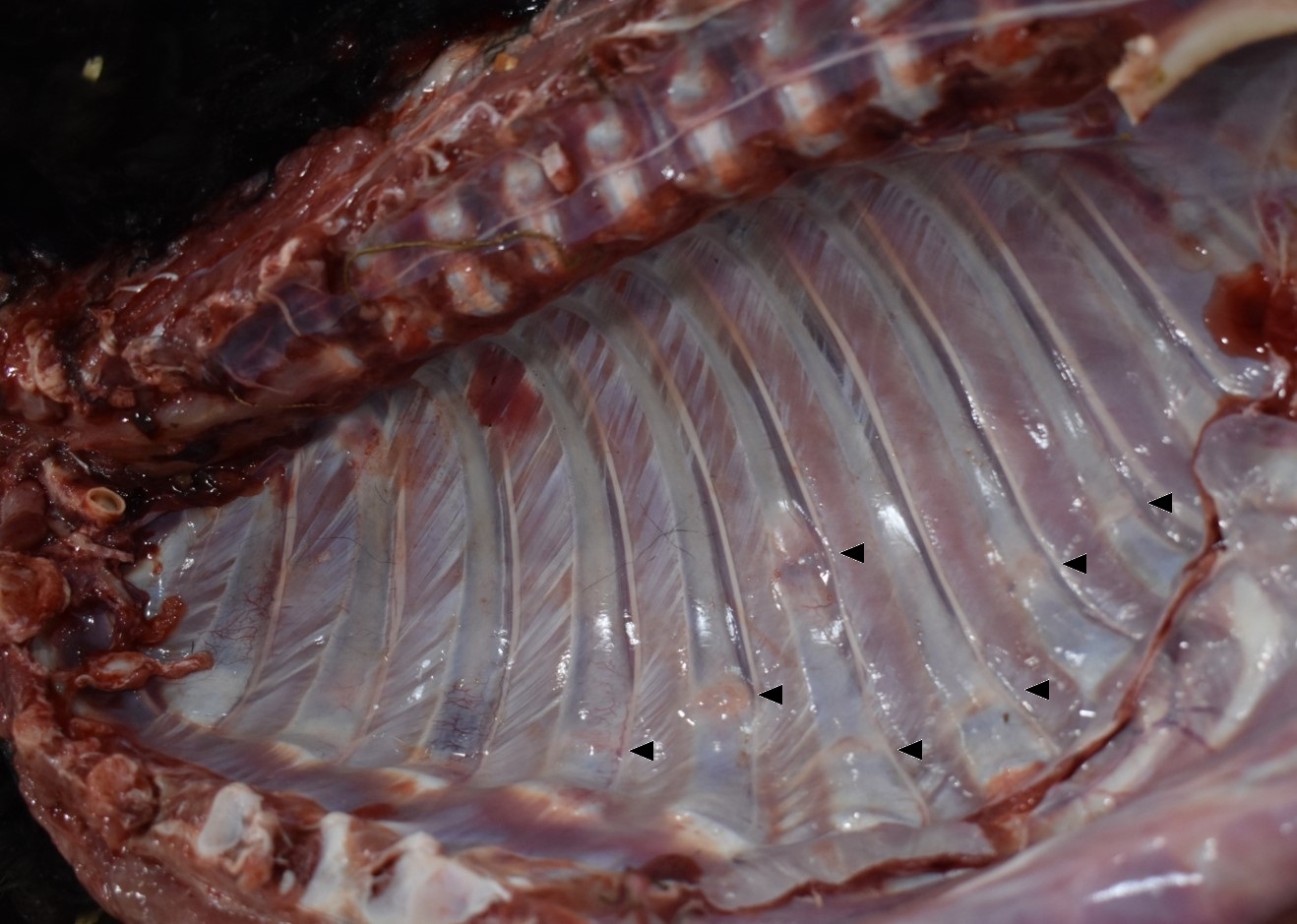
The alpaca presented to necropsy with a body condition score of 1/5. The fiber of the cria was black. The diaphysis of the left humerus was fractured without obvious formation of a fibrous callus and held in place with a metal plate. The cortical bone of the left humerus thin to segmentally non-discernable. The humeral fascia and skeletal muscles, extending to the proximal humerus to just distal the cubital joint, were edematous and hemorrhagic. Many transverse fractures with hard, bony fracture calluses were through the mid bodies of the right 5th ? 10th ribs. Subjectively, the ribs were easily broken. On longitudinal section of the left femur, the proximal physis was irregularly thickened. The epicardial fat was clear and gelatinous (serous atrophy).
Laboratory results:
A serum biochemistry was performed the day of death. Reference intervals are based on values published by UC Davis Veterinary Medical Teaching Hospital.8
SDH: 52.1 U/L (reference interval [RI]: 2 ? 5)
AST: 3249 U/L (RI: 107 - 199)
CK2: 13405 U/L (RI: 37 ? 108)
Total bilirubin: 0.36 mg/dL (RI: 0.0 ? 0.1)
BUN: 39.9 mg/dL (RI: 7 ? 22)
Calcium: 8.4 mg/dL (RI: 9.0 ? 9.9)
Phosphorous: 5.9 mg/dL (RI: 4.3 ? 8.3)
Microscopic description:
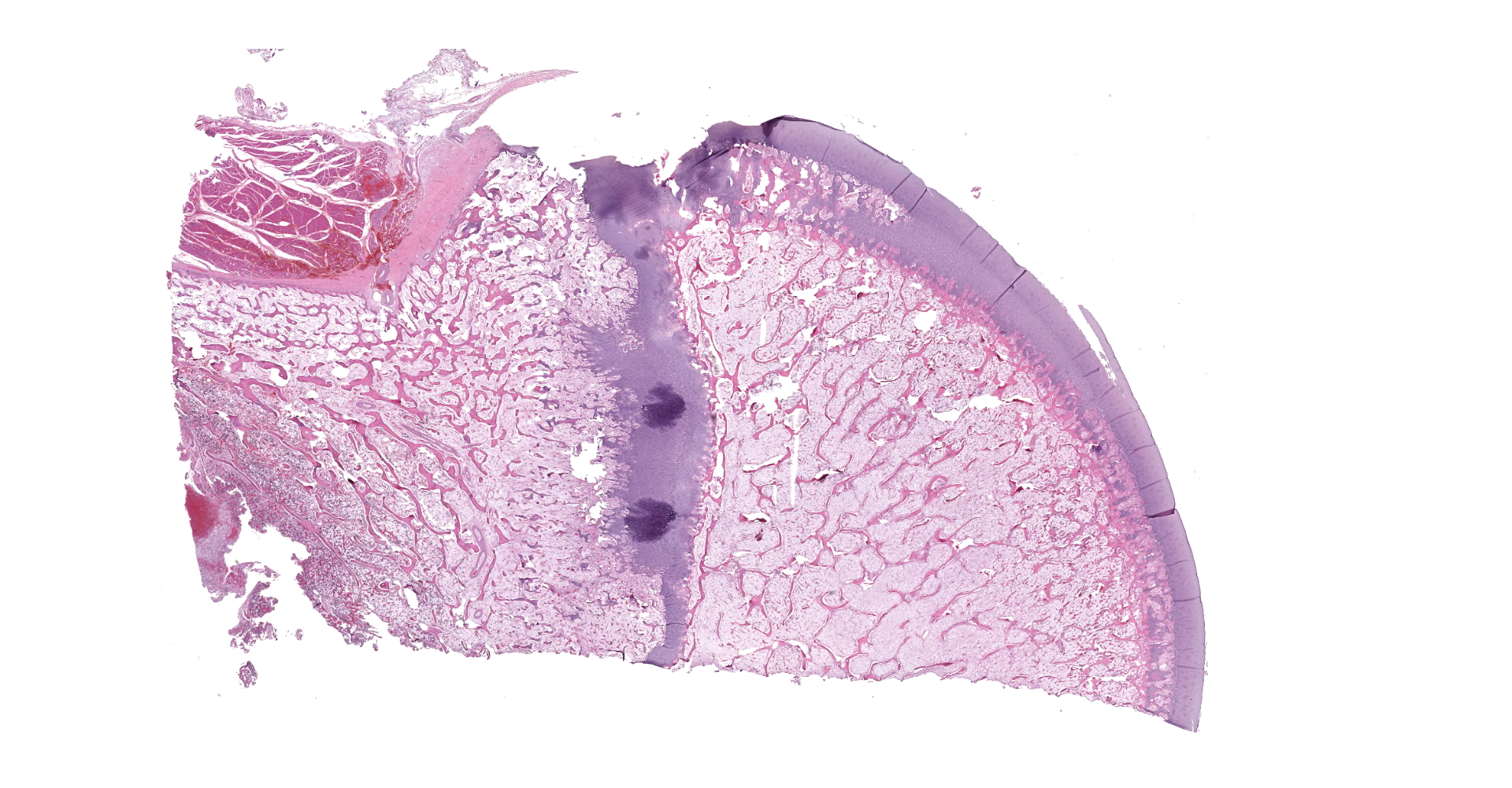
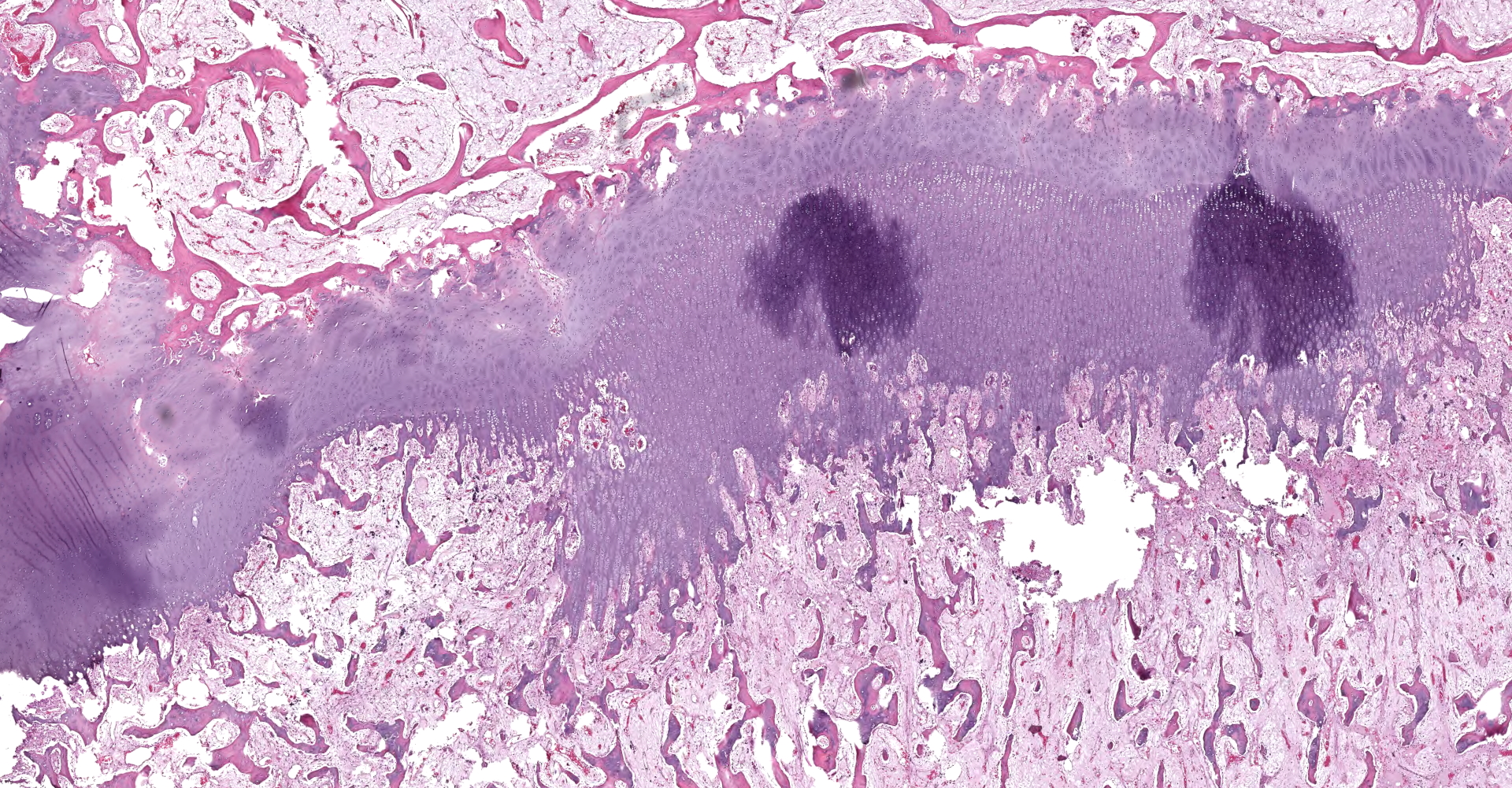
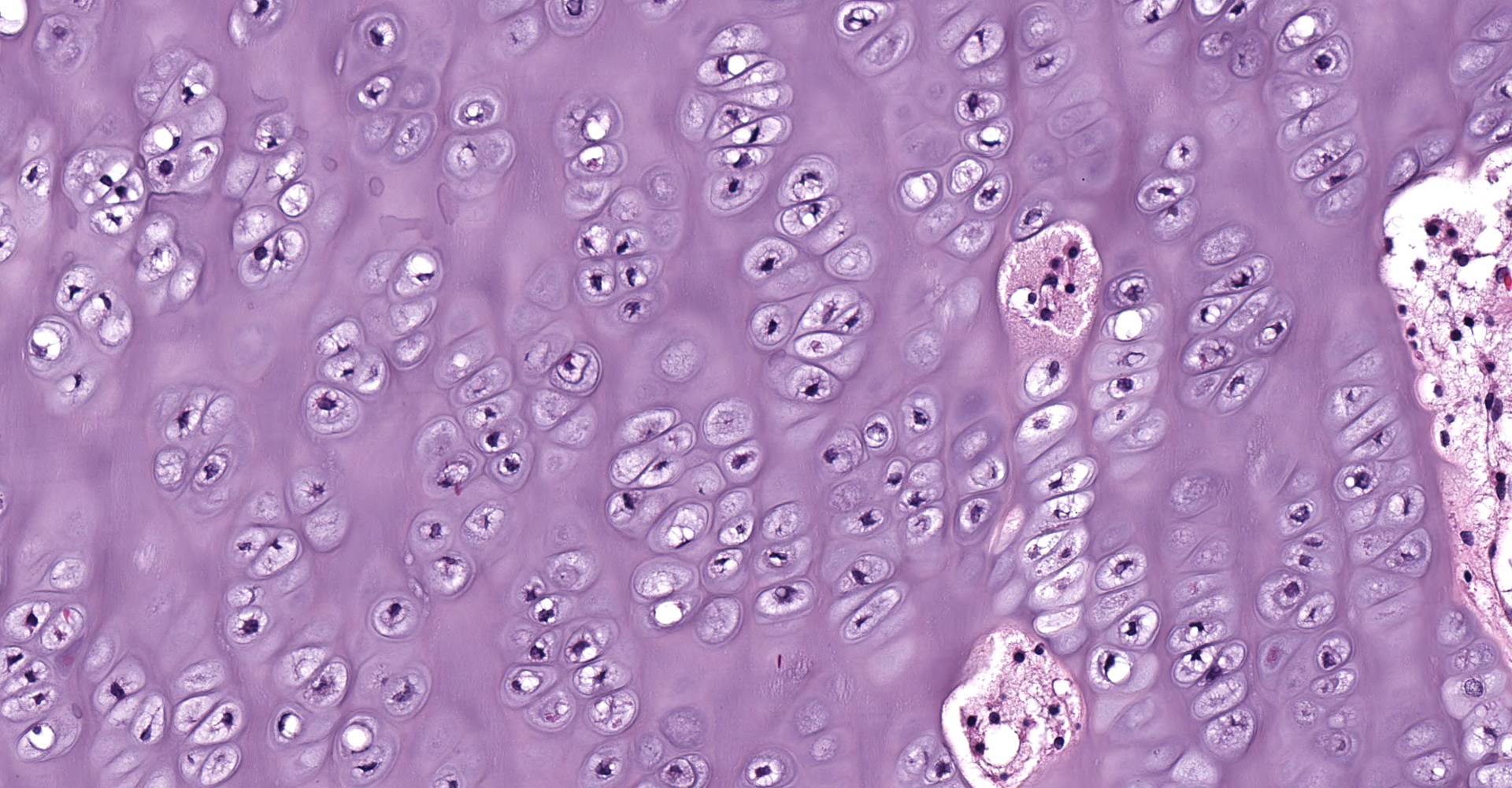
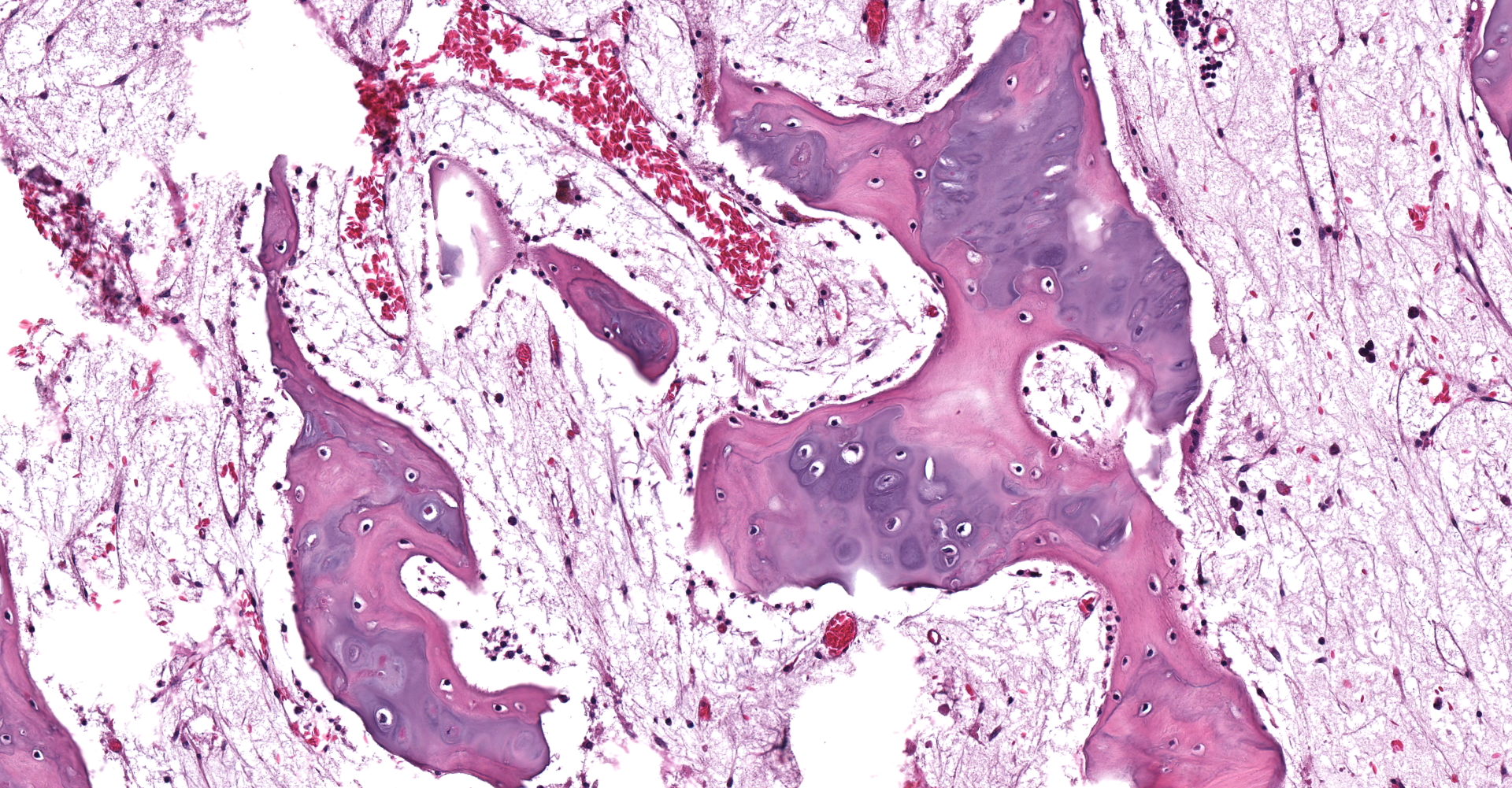
Left proximal humerus: The segmentally and irregularly thickened physeal cartilage has persistent tongues and disorganized clusters of hypertrophic, degenerate and necrotic chondrocytes that extend into the ill-defined primary and secondary spongiosa. The slender and frequently fragmented trabecular bony spicules have central cartilage, surrounded by woven and lamellar bone. The intertrabecular space is composed of a loose fibrovascular meshwork with intermixed rare, mature adipocytes and predominately later-stage hematopoietic elements. The metaphyseal cortex is flared, and the markedly attenuated to absent cortical bone is covered by an approximately 150 µm thick layer of fibrous periosteum. A focus of moderate hemorrhage is within the periosteum and the adjacent endomysium and perimysium. A cleft in the proximal diaphysis (previous location of screw) is surrounded by hemorrhage and displaces the bony spicules to the periphery.
Contributor's morphologic diagnosis:
Left proximal humerus: Physeal chondrodystrophy and delayed endochondral ossification (rickets).
Left proximal humerus: Fibrous osteodystrophy with osteopenia.
Contributor's comment:
Randomly distributed, moderate, neutrophilic acute hepatitis and minimal, acute, neutrophilic, interstitial pneumonia were diagnosed with histological examination of the standard tissues. These findings, along with the history of profound hypoglycemia, pointing to likely peracute septicemia as the cause of death. The emaciation (supported by a poor body condition and serous atrophy of epicardial fat) is considered to be a contributing cause of death for this case leading to an inability to mount a sufficient immune response.
Ultraviolet B dermal radiation converts 7-dehydrocholesterol (7-DHC) to pre-vitamin D3 in keratinocytes, which then undergoes thermal isomerization to vitamin D3. Vitamin D3, as well as dietary vitamin D2, are bound to vitamin D-binding protein and transported to the liver. In the liver, hepatic cytochrome 450 enzymes (such as CYP27A1, CYP3A4, CYP2R1, and CYP2J3) carry out 25-hydroxylation of vitamin D, converting it to 25-hydroxyvitamin D. Under the tight regulation of parathyroid hormone and plasma phosphorous, the kidney produces either the most active form of vitamin D, 1,25-dihydroxyvitamin D, or the inert metabolites, 24,25-dihydroxyvitamin D or 1,24,25-trihydroxyvitamin D.1,2
Melanin in the skin competes with 7-DHC for ultraviolet photons, so animals with pigmented skin require more UVB exposure for previtamin D3 formation. Animals at higher altitudes are exposed to higher levels of UVB and, therefore, may not need prolonged exposure to the sun for sufficient previtamin D3.2
Vitamin D targets, indirectly or directly, the kidneys, parathyroid glands, intestine, and bone. In the intestine, vitamin D promotes the uptake of calcium and increase active phosphate absorption. Transient receptor potential vallinoid 6 (TRPV6) transports calcium into the intestinal enterocyte to bind to calbindin D. Calbindin D transports calcium to PMCA1b and NCX1 to excrete calcium into the bloodstream. Both TRPV6 and calbindin D are upregulated by vitamin D.2
Vitamin D maintains the ionized blood calcium concentration by mobilizing calcium from bone stores. Vitamin D likely stimulates RANKL, thereby increasing binding to RANK on osteoclasts and stimulates the maturation of osteoclast progenitor cells to osteoclasts (osteoclastogenesis). The decoy receptor osteoprotegerin (OPG) is downregulated by vitamin D. Binding of RANLK to OPG inhibits osteoclastogenesis.
Vitamin D is partially balanced by parathyroid hormone. Vitamin D deficiencies result in hypocalcemia, stimulating the parathyroid gland to secrete parathyroid hormone. Excess vitamin D can suppress the parathyroid gland by decreasing growth factors, inhibit cell growth, and increasing ionized calcium sensitivity of the parathyroid gland.2
Black-coated alpacas are predisposed to developing vitamin D deficiency. Previous research has demonstrated that black-coated alpacas have lower serum vitamin D3 all year, with vitamin D3 levels that do not fluctuate with yearly solar radiation levels, unlike lighter-coated alpacas.6 Bone density in light-coated alpacas, along with serum vitamin D3 and phosphorous concentrations, does fluctuate with, but lags behind, seasonal levels of solar radiation. Due to this lag, the nadir of bone density and vitamin D3 in the northern hemisphere is from January to May despite solar irradiation being lowest in December.5
Rickets is a disease in immature and growing animals, such as in this case of an 8-month-old animal, caused by a deficiency in vitamin D and/or phosphorous. Affected animals with rickets will fail to mineralize their bone and have subsequent deformities, pathological fractures, and bone pain. In rickets, this failure of mineralization and endochondral ossification causes the growth plates to be irregularly thickened. This thickening of the growth plates is especially prominent in the costochondral junctions, forming a row of nodular thickenings ? classically termed 'rachitic rosary' due to the string of beads-like appearance.4
Vitamin D deficiency histologically differs from phosphorous deficiency rickets in bone. Both forms will develop the characteristic physeal failure of endochondral ossification. However, lack of vitamin D impairs the absorption of calcium from the intestine and the reabsorption of calcium from bone, leading to hypocalcemia and secondary hyperparathyroidism. Because of the hypocalcemia, younger animals with vitamin D deficiency have lesions that overlap with rickets and fibrous osteodystrophy.1 This is most notable in the cortical and trabecular bone. With phosphorous deficiency, the trabecular and cortical bone contains excess unmineralized osteoid leading to failed osteoclast resorption and impaired bone modeling/remodeling. With vitamin D deficiency, there is extensive resorption and replacement of the trabecular and cortical bone with fibrous connective tissue (features of fibrous osteodystrophy).4
Contributing Institution:
Auburn University - Department of Pathobiology
https://www.vetmed.auburn.edu/academic-departments/dept-of-pathobiology/
JPC diagnosis:
- Long bone: Physeal and epiphyseal osteodysplasia, diffuse, with regional delayed endochondral ossification (rickets).
- Long bone: Osteopenia, trabecular and cortical, diffuse.
- Long bone: Serous atrophy of fat, diffuse, marked.
JPC comment:
The moderator emphasized that these lesions may be due, in part, to rickets. Other portions of the bone, such as the fewer and thinner bony trabeculae, are features of osteopenia. The clinical history and gross findings may also support malnourishment in this animal, contributing to the lesions observed in this animal. The moderator described three features likely occurring concurrently in this animal: fibrous osteodystrophy, delayed endochondral ossification, and chondrodystrophy. Contributing factors may have been protein and caloric restriction, hypocalcemia, and vitamin D deficiency.
Vitamin D balance is crucial for calcium and phosphorus homeostasis, and its deficiencies are well illustrated by this case. Different species depend on vitamin D for this balance to different degrees, and it has been hypothesized that these differences can be attributed to evolutionary adaptation to different environments. The movement of animal life from aquatic environments, which is relatively higher in calcium and lower in phosphorus than land environments, necessitated new mechanisms to absorb calcium and regulate the balance with phosphorus needs. Successful adaptations included the increased absorption of calcium from the gastrointestinal system, more flexible bone remodeling which acts as a calcium reservoir, and the maturation of three intertwined hormones that regulate the calcium and phosphorus balance. Parathyroid hormone, vitamin D, and fibroblast growth factor 23 (FGF23) all act together to provide feedback to the absorption, reservoir, and elimination mechanisms within the body to provide an optimum balance for life.7
While the effects of PTH and vitamin D are regularly described and detailed, FGF23 is more recently characterized. FGF23 is secreted by osteoblasts and osteocytes when stimulated by 1,25(OH)2D3, PTH, or chronically elevated levels of serum calcium and phosphorus. The elaboration of FGF23 provides negative feedback to PTH secretion, inhibits renal absorption of phosphorus, and reduces the capacity of the kidney to activate vitamin D.7
The moderator emphasized that FGF23 sits at an interesting intersection of rickets and fibrous osteodystrophy/osteopenia, providing critical feedback to the systems regulating these processes. In this case, it is not immediately clear whether FGF23 is implicated in the development of disease, or might be a useful target for therapy.
Defects in renal 1-?-hydroxylase results in vitamin D-dependent rickets, Type 1, where 25(OH)D3 cannot be transformed to 1,25(OH)2D3. This key enzyme is encoded by the gene CYP27B1 (type 1a), or CYP27R1 (type 1b), and mutations have been documented as autosomal recessive disease in humans, Hannover pigs, a few cats, and one Saint Bernard dog.1 This disease manifestation in cats is due to mutated CYP27B1 genes, but there have been different mutation locations and types, indicating non-specific initiating events. Additional research may focus on interactions in non-coding regions supporting these exons.3
References:
1. Craig LE, Dittmer KE, Thompson KG. Bones and Joints. In: Vol. 1, Jubb, Kennedy & Palmer's Pathology of Domestic Animals: Volume 1. Elsevier; 2016:16-163.e1.
2. Dittmer KE, Thompson KG. Vitamin D Metabolism and Rickets in Domestic Animals. Vet Pathol. 2011 Mar 15;48:389?407.
3. Grahn RA, Ellis MR, Grahn JC, Lyons LA. A novel CYP27B1 mutation causes a feline vitamin D-dependent rickets type Ia. J Feline Med Surg. 2012;14(8):587-590.
4. Olson EJ, Carlson CS. Bones, Joints, Tendons, and Ligaments. In: Zachary JF, ed. Pathologic Basis of Veterinary Disease. St. Louis: Elsevier; 2017:954-1008.e2.
5. Parker JE, Timm KI, Smith BB, et al. Seasonal interaction of serum vitamin D concentration and bone density in alpacas. Am J Vet Res. 2002 Jul;63:948?953.
6. Smith BB, Van Saun RJ. Seasonal changes in serum calcium, phosphorus, and vitamin D concentrations in llamas and alpacas. Am J Vet Res. 2001 Aug;62:1187?1193.
7. Uhl EW. The pathology of vitamin D deficiency in domesticated animals: An evolutionary and comparative overview. International Journal of Paleopathology. 2018; 23:100-109.
8. Veterinary Medical Teaching Hospital - University of California Davis. Clinical Chemistry Reference Intervals. 2010.