Joint Pathology Center
Veterinary Pathology Services
Wednesday Slide Conference
2018-2019
Conference 6
October 3, 2018
CASE II: A16-38521 (JPC 4102645-00).
Signalment: Seven-year-old neutered male mixed breed dog, Canis familiaris
History:The dog presented to a veterinary college neurology service one month prior to necropsy following a 3-week history of circling toward the left and altered mentation. On neurologic examination, the dog had inappropriate, dull mentation and a left head turn. He was ambulatory with no weakness or ataxia but circled to the left with right-sided postural reaction deficits in the thoracic and pelvic limbs. There was an inconsistent menace response on the right side. His neuroanatomic localization was to the left prosencephalon. Magnetic resonance imaging (MRI) revealed a T2-weighted homogenously hyperintense, non-contrast enhancing mass lesion within the left thalamus and midbrain with secondary obstructive hydrocephalus, characterized by distension of the lateral and third ventricles. The dog was started on corticosteroid therapy, but gradually declined until the owners elected to euthanize.
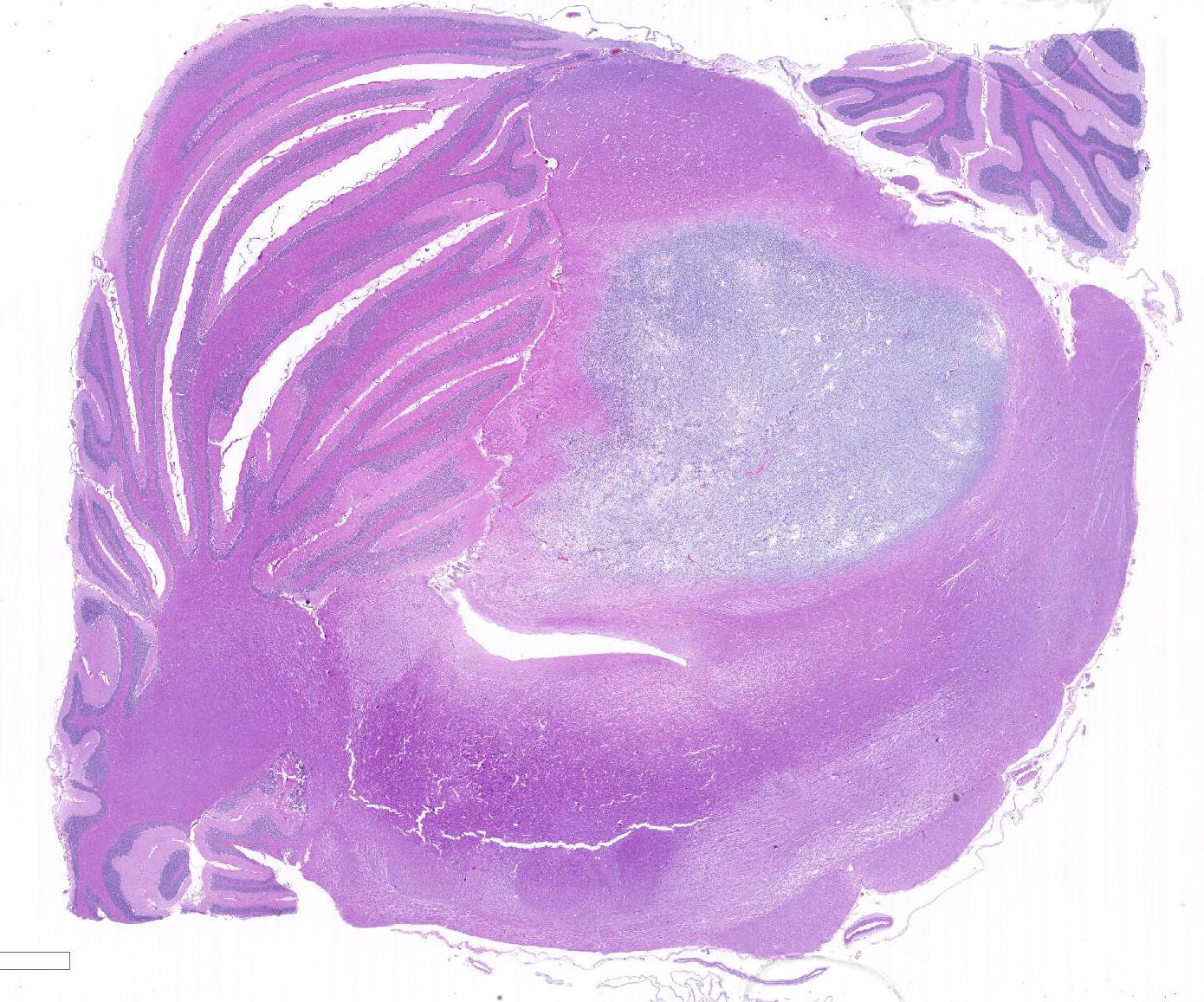
Gross Pathology: The brain was mildly edematous and there was thinning of the cerebral cortex bilaterally with distension of the lateral ventricles. Serial sectioning revealed a well-circumscribed, well demarcated, pale, gelatinous mass on the left side effacing the mesencephalon with compression of the third ventricle and mesencephalic aqueduct.
Laboratory results: None given.
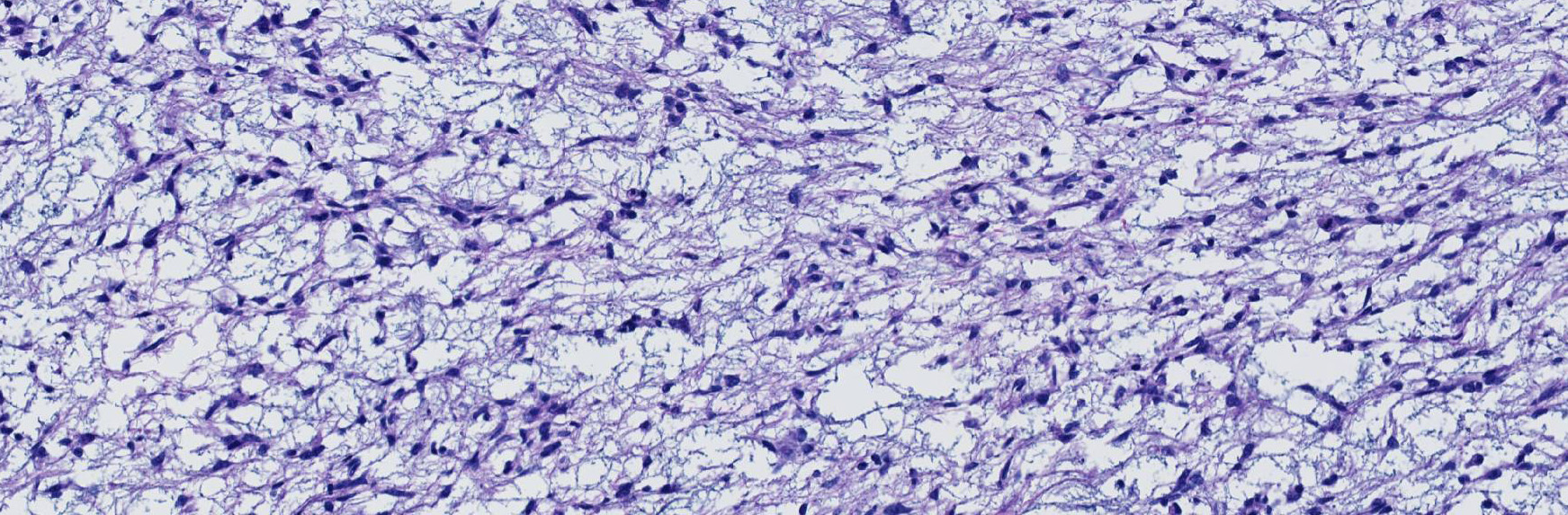
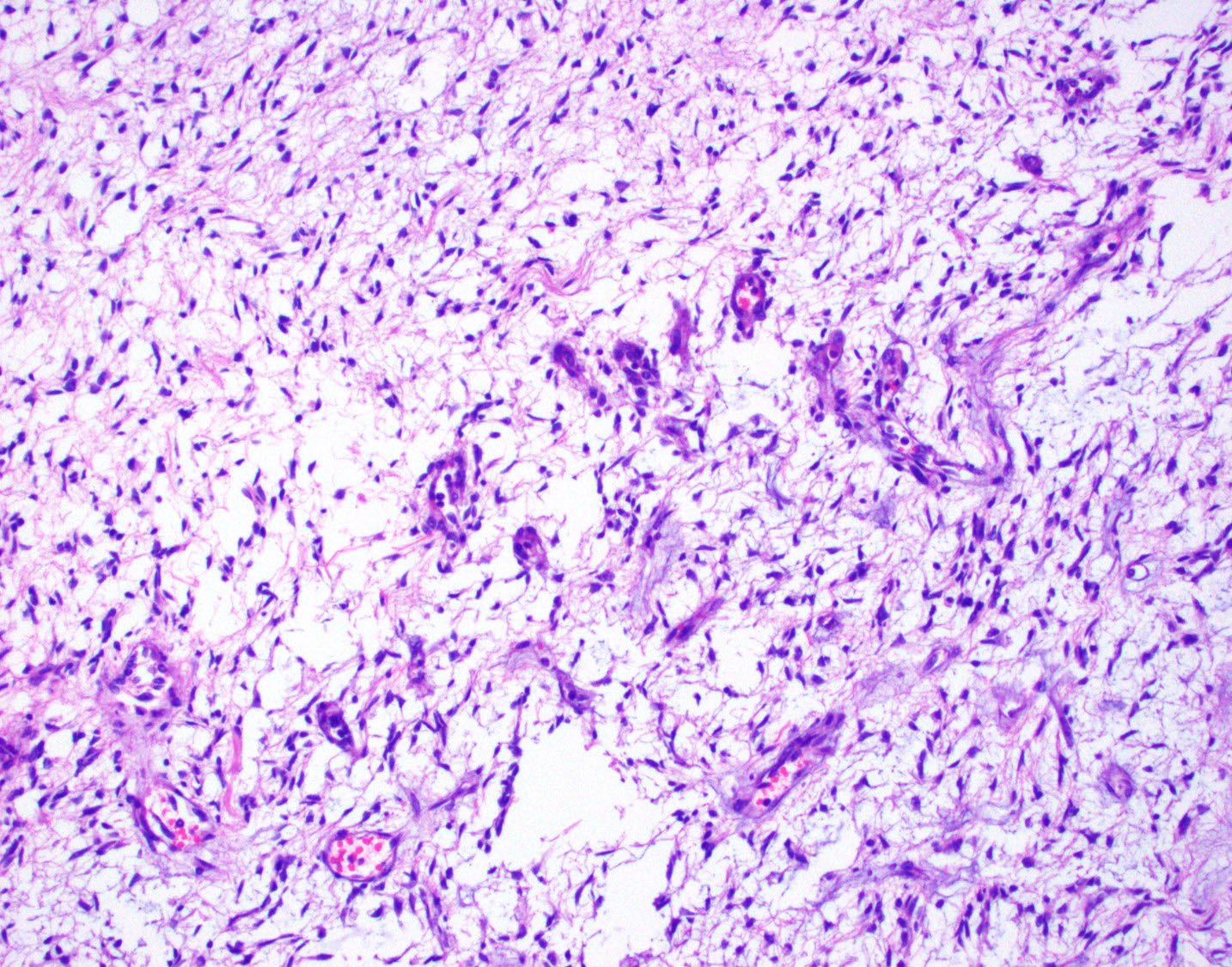
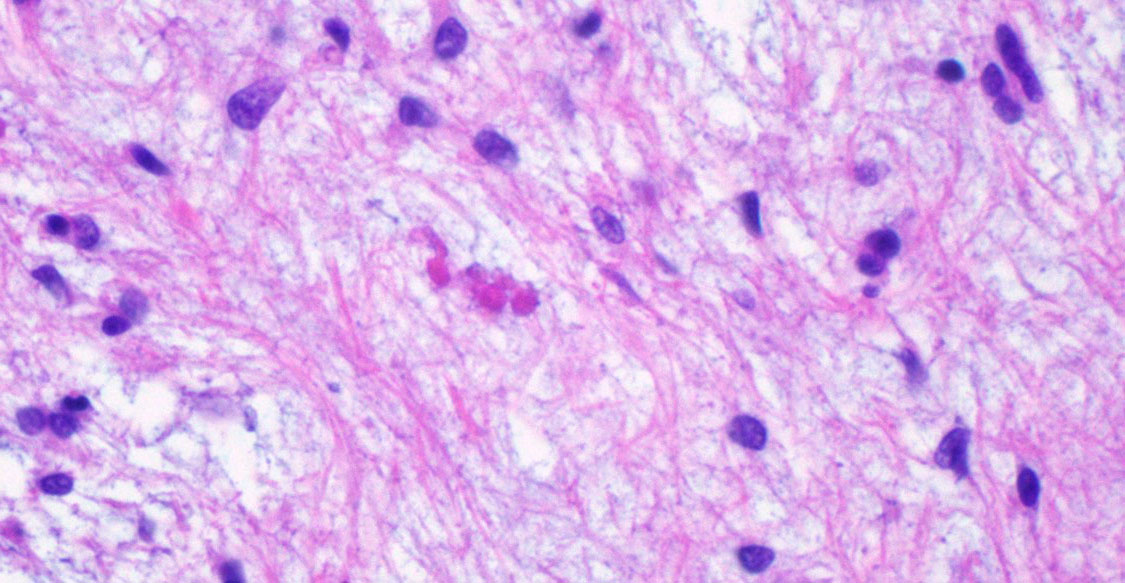
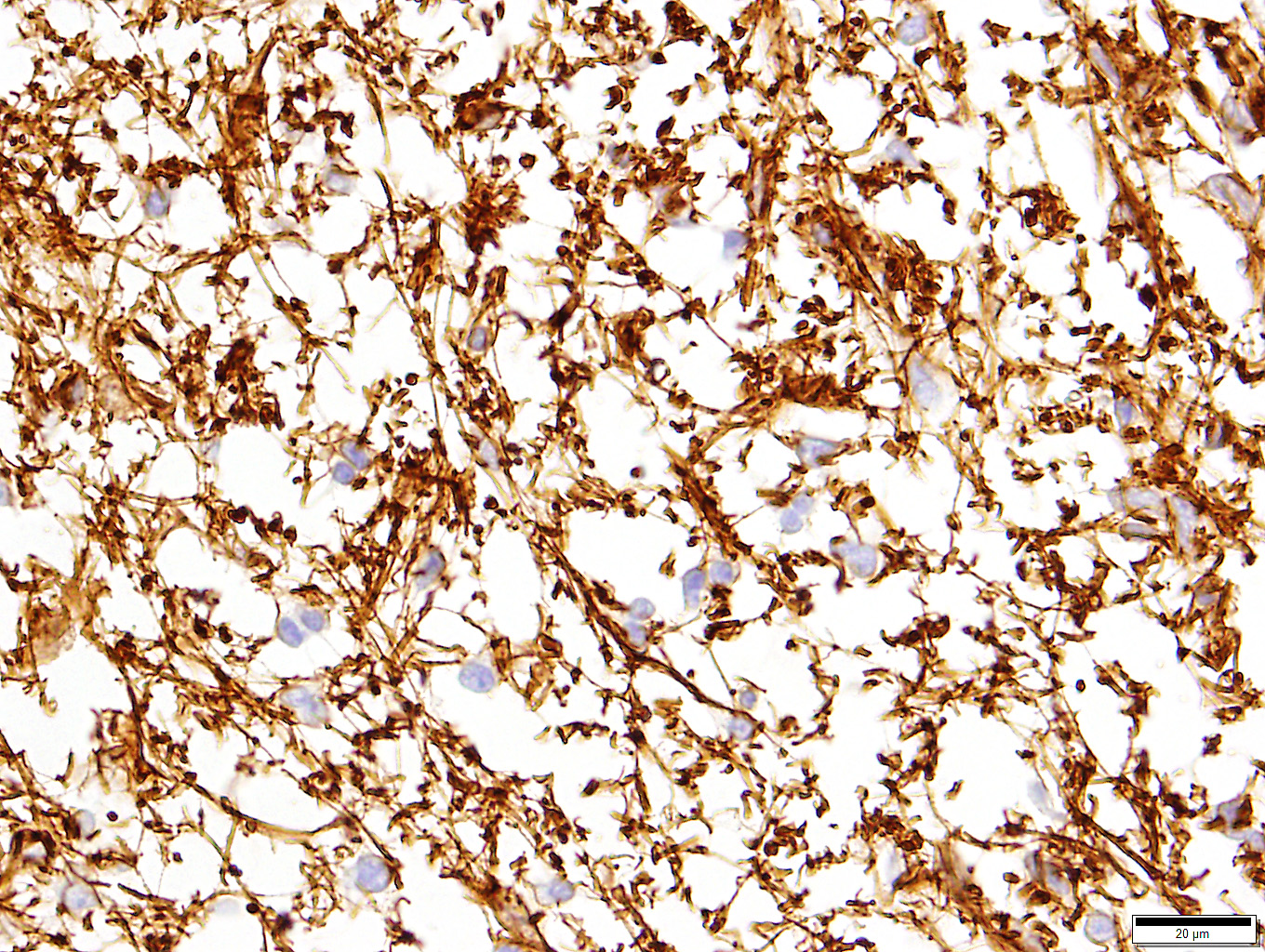
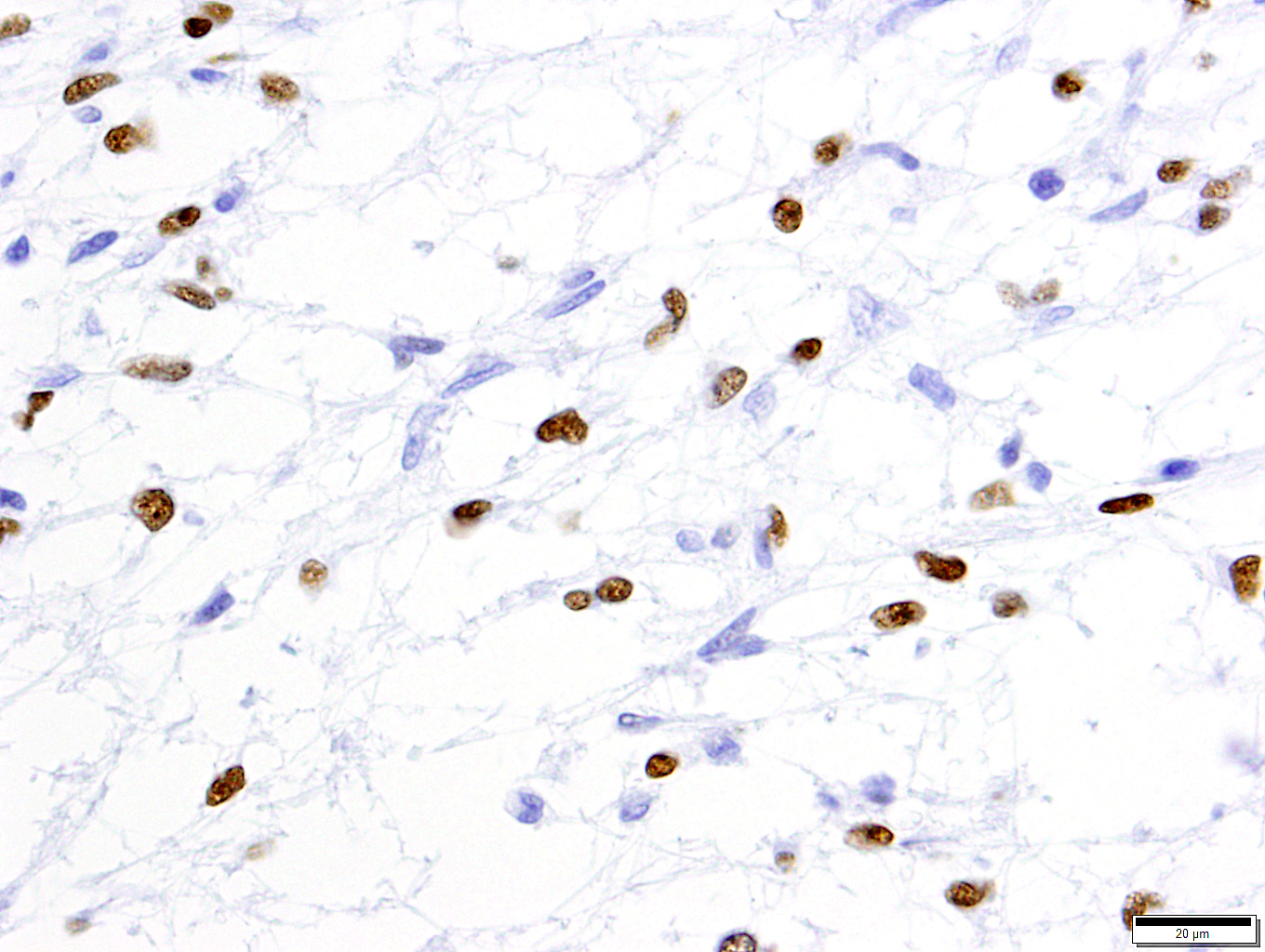
Microscopic Description: Extending from the thalamus caudally to the cerebellar peduncles caudally and expanding the mesencephalon with compression of the lateral ventricles and mesencephalic aqueduct was a well demarcated, unencapsulated, expansile, moderately cellular mass. There were two morphologically distinct cellular populations within the mass: bipolar, spindloid cells with small ovoid nuclei and long thin wispy cytoplasmic processes, and proliferative, small, round, individualized cells with round to indented, condensed nuclei and scant, granular cytoplasm. Cells were embedded within an abundant loose myxoid matrix that was positive on Alcian blue pH 2.5. Throughout the mass, there were scattered proliferations of hypertrophied endothelial cells, frequently forming glomeruloid capillary loops. There was pallor and mild vacuolation of the surrounding neuroparenchyma. Most of the large, bipolar cells and their associated spindloid processes were strongly positive for GFAP on immunohistochemistry. The nuclei of the smaller round cells stained positively for Olig2, but did not stain with GFAP.
Contributor’s Morphologic Diagnoses: Neoplasia, oligoastrocytoma, thalamus to brain stem, left side with bilateral hydrocephalus
Contributor’s Comment: There is a diffuse mixture of GFAP-positive and Olig2-positive cells within the mass. The Olig2-positive cells, interpreted as oligodendrocytes, are too proliferative to be considered reactive cells and therefore are likely neoplastic. There is also proliferation of GFAP-positive astrocytic cells, but it is less clear whether these are reactive or neoplastic. Astrocytes will commonly proliferate into and around oligodendrogliomas in response to the neuroparenchymal destruction5. This reactive response should comprise no more than 30% of the mass, which is far less than the proportion of astrocytic, GFAP-positive cells observed in this mass, which led us to the diagnosis of oligoastrocytoma. Oligoastrocytomas, or mixed gliomas, have been described previously in dogs, where both glial lineages have neoplastic features. These two populations are often separated and distinguishable within the mass, but more heterogeneous and mixed masses are reported.2,5,6It has also been proposed gliomas originating from more primitive cell lines may be dually positive for astrocytic and oligodendroglial markers.5 In this case, however, there appear to be two distinct populations.
Contributing Institution:
University of Georgia, College of Veterinary Medicine, Department of Pathology, (www.vet.uga.edu/VPP)
JPC Diagnosis: Brainstem, adjacent to 4th ventricle: Astrocytoma, noninfiltrative, low grade.
JPC Comment: A 2018 publication by the Comparative Brain Tumor Consortium4 has laid the groundwork for a sweeping reclassification of canine glial tumors. Currently the World Health Organization (WHO) Tumor Fascicle Histological Classification of Tumors of the Nervous System of Domestic Animals, published in 1999, still serves as the gold standard for diagnosis of canine glial tumors; however, its diagnostic content is based largely on morphology alone (with a small amount of immunohistochemical data– then in its infancy – on some of the more common neoplasms). The human side of the WHO has published not one, but two editions in their series on Tumors of the CNS, grouping these neoplasms into expected outcomes based on morphologic features, but also incorporating molecular data as part of the diagnostic and prognostic algorithms. This new publication on canine tumors lays the important groundwork for similar advances when they become more widely available.
The new publication also has an immediate impact on the current methods of diagnosis of canine glial tumors, simplifying the classification of these tumors into three groups: astrocytoma, oligodendroglioma, and “undefined glioma”. A takeaway message that will immediately simplify classification is the recommendation that any tumor in which greater than 80% of the neoplastic cells exhibit morphology and immunoreactivity of either astrocytic or oligodendroglial differentiation, that the tumor be classified into one of those two groups. “Undefined” gliomas are characterized as glial neoplasms which do not have predominant features of either an oligodendroglioma or an astrocytoma; the geographic distribution of the neoplastic cells in these tumors might be either segregated or intermingled.4
A number of excellent tips regarding the interpretation of immunohistochemistry on canine gliomas is also presented in this article. The authors recommend the following panel in the diagnosis of canine gliomas: GFAP, Olig2, CNPase (a component of myelin expressed exclusively by oligodendroglia in the CNS), and Ki67. It should be noted that despite its name, Olig2 is not specific for oligodendroglia and may be expressed by neoplastic astrocytes as well. A lack of Olig2 staining is of diagnostic importance as well as a result of its specificity for oligodendro- and astroglia, and a lack of staining should lead the pathologist to strongly consider non-glial tumors at that point in the diagnostic process. The article also comments on the interpretation of immunohistochemical results, with decreased staining of GFAP, CNPase and Ki67 all being negatively impacted by time to fixation (which may be important in the processing of necropsy samples) as well as time in fixation, and fixation buffering. 4
Finally, the paper suggests morphologic criteria (albeit subjective) that may be useful in classifying gliomas into low- or high-grade, to include necrosis, pseudo-palisading of neoplastic cells along areas of necrosis, mitotic activity, microvascular proliferation, and cellular features of malignancy (atypia, anisocytosis, anisokaryosis, and nuclear pleomorphism). Interestingly, in this study, oligodendrogliomas had an overall increased rate of high-grade tumors than astrocytomas, and in keeping with anecdotal observations by many veterinary neuropathologists, oligodendroglial tumors in general far outnumbered astrocytic tumors.4
A number of features in this particular neoplasm are key to its classification as an astrocytoma (notwithstanding the diffuse immunoreactivity for GFAP, as this information was not provided to attendees.). The minimal amount of cytoplasm of the neoplastic cells, elongate nuclei, bipolar cell morphology, lack of necrosis, and lack of any apparent nuclear rowing (aka “interfascicular queuing”) were considered more appropriate for the diagnosis of astrocytoma in this case. It is important to note that in pilocytic astrocytomas in humans (which this neoplasm strongly resembles in appearance and location), smaller Olig2 cells can be interspersed with the bipolar astrocyte population and do not confer a diagnosis of oligoastrocytoma. The presence of microcysts and formation of Rosenthal fibers by some of the neoplastic cells was also considered of diagnostic importance (a feature that has been well established in human astrocytomas)
During the discussion of this case, the moderator acknowledged the difficulty of identifying true edema in neurologic sections and the lack of well-defined critieria for it, but believes that the area of rarefaction surrounding the neoplasm and extending into the adjacent parenchyma in this case is a good example of spongiosis.
References:
- Ide T, Uchida K, Kikuta, F, Suzuki K, Nakayama H. Immunohistochemical characterization of canine neuroepithelial tumors. Vet Pathol 2010; 47:741-50.
- LeCouteur RA, Withrow SJ. Tumors of the nervous system. In: Withrow SJ, Vail DM, eds. Small Animal Clinical Oncology. St Louis, MO: Saunders Elsevier; 2007:659-670.
- Higgins RJ, Bollen AW, Dickinson PJ, Siso-Lonch S. Tumors of the nervous system. In: Meuten DJ, ed, Tumors of Domestic Animals, 5th ed. Ames IA, Wiley-Blackwell, 2017: 848-849.
- Koehler JW, Miller AD, Miller CR, Porter BR, Aldape K, Beck J, Brat D, Cornax I, Corps K, Frank C, Giannini, Horbinski C, Huse JT, O’Sullivan MG, Rissi DR, Simpson RM, Woolard K, Shih JH, Mazcko C, Gilbert MR, LeBlanc AK. A revised diagnostic classification of canine glioma: towards validation of the canine glioma patient as a naturally occurring preclinical model for human glioma. J Neuropathol Exp Neurol 2018; doi 10.1093/jnen/nly085
- Summers BA, Cummings JF, deLaunta A. Veterinary Neuropathology. St. Louis, MO: Mosby, 1995:370-373.
- Vandvelde M, Higgins RJ, Oevermann O. Neoplasia. In Veterinary Neuropathology. West Susses, United Kingdom, Wiley-Blackwell; 2012: 139-140.