Signalment:
Gross Description:
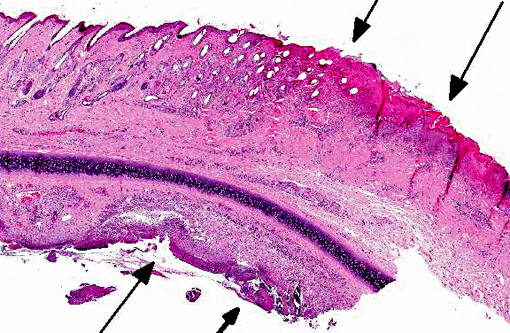
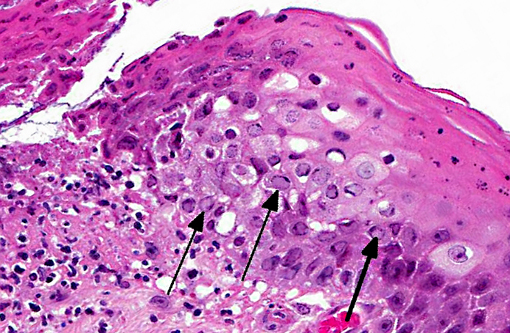
Histopathologic Description:
Morphologic Diagnosis:
1. Severe acute ulcerative conjunctivitis with intralesional intranuclear inclusion bodies consistent with feline herpesvirus infection.
2. Subacute ulcerative folliculitis with intralesional mites suggestive of Notoedres cati.
Condition:
Contributor Comment:
Most cats are infected as kittens, developing conjunctivitis, corneal ulceration or rhinitis of varying severity depending on viral strain and host susceptibility. The virus replicates in epithelial cells at temperatures below 37o Celsius and so its preferential proliferation sites are peripheral locations such as ocular and nasal mucosa. The virus damages tissue by direct cytolysis during replication and secondarily by immune-mediated inflammation. Cytolysis results in corneal ulceration, conjunctivitis with ulceration and rhinitis with erosions. Immunemediated injury is less common and results in stromal keratitis. In primary infections the most common presenting lesion is conjunctivitis.(2,7)
Up to 80% of infected cats go on to develop virus latency, typically in the trigeminal nerve, from which latency associated transcripts of FHV-1 have been demonstrated. More recently, evidence of persistent low-grade viral infection has been demonstrated in the cornea.(2,6,7) In approximately 50% of carriers, reactivation of infection can occur either spontaneously or during periods of stress or corticosteroid administration. Classically, this is associated with centrifugal spread of the virus from the trigeminal nerve along the sensory nerves to the original epithelial site of invasion. Upon reactivation, viral shedding recommences and this can be with or without recrudescence of clinical signs. With reactivation, the most common presenting clinical sign is conjunctivitis.(2)
Notoedres cati (feline scabies) is a rare, highly contagious ectoparasite of cats causing alopecia with adherent crusts around the head and ears, particularly the ear pinna. Histologically the parasite triggers superficial dermatitis with eosinophil-rich perivascular inflammation.(3)
JPC Diagnosis:
1. Eyelid: Blepharoconjunctivitis, ulcerative, focally extensive, severe with intranuclear viral inclusions.
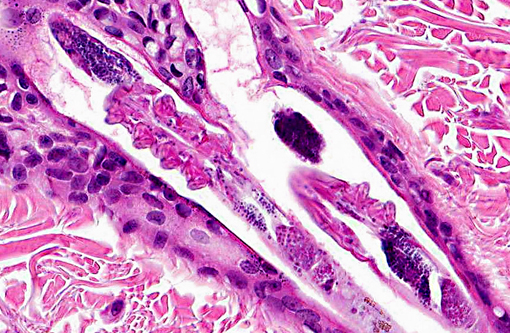
2. Haired skin, eyelid follicles and sebaceous glands: Adult arthropod parasites, etiology consistent with Demodex cati.
Conference Comment:
Dr. Chris Gardiner, JPC consultant in veterinary parasitology, believes these arthropods are more consistent with Demodex rather than Notoedres cati and provided by following justification: -�-�The mites in this specimen are elongate and found within the hair follicles; whereas, Notoedres cati mites are dorsoventrally flattened, similar to other members of the Family Sarcoptidae, and are typically observed in the stratum corneum.(1,4) Additionally, Demodex spp. are the only parasitic mites in which all four pairs of appendages originate together. Notoedres mites, on the other hand, have two pairs of two appendages that originate together separated by a space from another two pairs of two appendages that originate together. (C.H. Gardiner, personal communication).
References:
2. Gaskell R, Dawson S, Ramford A, Thiry E. Feline herpesvirus. Vet Res. 2007;38:337-354.
3. Ginn PE, Mansell JEKL, Rakich PM. Skin and appendages. In: Maxie MG, ed. Jubb, Kennedy and Palmers Pathology of Domestic Animals. 5th ed. vol. 1. Philadelphia, PA: Elsevier Saunders; 2007:721-722.
4. Hargis Am, Ginn PE. The integument. In: Zachary JF, McGavin MD, eds. Pathologic Basis of Veterinary Disease. St. Louis MO: Elsevier Mosby; 2012:1043.
5. Lopez A. Respiratory system, mediastinum, and pleurae. In: Zachary JF, McGavin MD, eds. Pathologic Basis of Veterinary Disease. St. Louis MO: Elsevier Mosby; 2012:473.
6. Maggs DJ. Update on pathogenesis, diagnosis and treatment of feline herpesvirus type 1. Clin Tech Small Anim Pract. 2005;20:94-101.
7. Wilcock BP. Eye and ear. In: Maxie MG, ed. Jubb, Kennedy and Palmers Pathology of Domestic Animals. 5th ed. vol. 1. Philadelphia, PA: Elsevier Saunders; 2007:490-491.