Signalment:
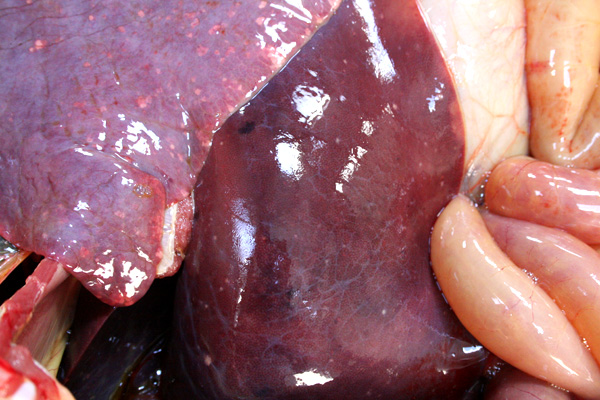
Gross Description:
Histopathologic Description:
Scattered randomly within the adrenal cortex are areas of congestion, hemorrhage and necrosis. Immediately adjacent to the cytoclastic debris are cells that contain eosinophilic nuclear inclusion bodies that peripheralize the chromatin.
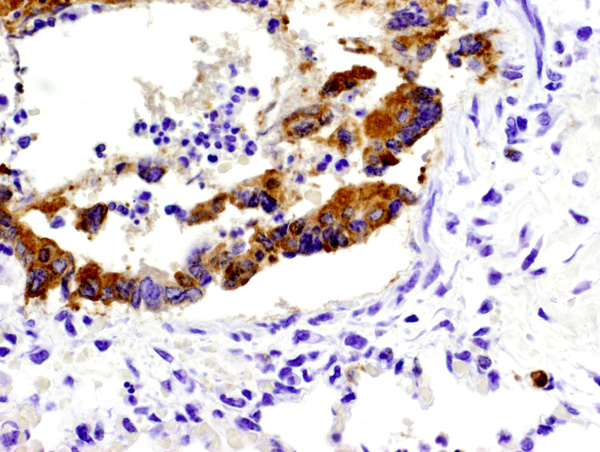
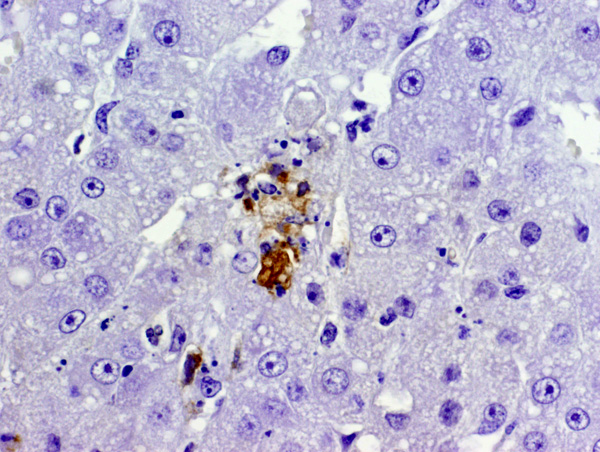
Equine herpesvirus 1 (EHV-1) antigen was detected by immunohistochemisty within the nucleus and cytoplasm of several epithelial cells and leukocytes in the lung (figure 2) and adrenal gland (figure 3). Appropriate positive and negative controls were used and examined and worked accordingly.
Morphologic Diagnosis:
Adrenal gland: adrenalitis, necrotizing, acute, multifocal, moderate with eosinophilic nuclear inclusion bodies.
Lab Results:
Condition:
Contributor Comment:
The pathogenesis of EHV-1 abortion is not fully elucidated11. Virus is translocated from the maternal circulation to the uterus and placenta. Uterine lesions consist of vasculitis in the small arterioles of the endometrium11. In some cases, abortion can occur without fetal lesions or virus spread to the fetus, presumably from widespread virus-related thrombosis and infarction leading to premature placental separation and expulsion of the fetus14. Placental lesions in these cases consist of chorionic necrosis and fibrinoid vascular necrosis of chorionic blood vessels with fibrin thrombi13,14. EHV-1 has been detected in endometrial and chorionic endothelial cells in experimental and spontaneous cases of abortion by ISH and immunohistochemistry12,13,15.
More commonly virus spreads to the fetus. In addition to placental endothelial cells, DNA ISH also has identified EHV-1 in necrotic debris associated within infarcted microcotyledons, debris within endometrial glands and also trophoblasts, suggesting trophoblast infection results from diffusion of virus from sites of endometrial infarction and also from emptying of debris from infected glands directly onto the surface of trophoblasts12.Â
EHV-1 infection of the fetus results in well described and documented lesions. Grossly, the aborted fetus is usually fresh with subcutaneous edema and petechiae of the mucous membranes. The lungs are edematous and the trachea may contain a fibrinous cast. The liver contains miliary white foci of necrosis. The spleen may contain prominent lymphoid follicles2,9. Histologic lesions consist of necrosis and eosinophilic intranuclear inclusion bodies in parenchymal organs, especially the liver and adrenal glands, with minimal inflammatory cell infiltrate, lymphocytolysis in the thymus and bronchointerstitial pneumonia2,6. Syncytia formation in EHV-1 infection, as seen in this case, is rarely described. Previous reports include syncytia in the lungs of aborted fetuses6 and in experimental neurologic disease3.Â
JPC Diagnosis:
1. Lung: Pneumonia, bronchointerstitial, necrotizing, acute, multifocal, moderate, with fibrin, edema, syncytia, and eosinophilic intranuclear inclusion bodies, Thoroughbred (Equus caballus).
2. Adrenal gland, cortex: Necrosis, multifocal, with rare eosinophilic intranuclear inclusion bodies.
Conference Comment:
There are three types of Equine Alphaherpes viruses:
- EHV-1: Equine viral abortion, myeloencephalopathy, respiratory disease
- EHV-3: Equine coital exanthema
- EHV-4: Rhinopneumonitis virus
EHV-1 and EHV-4 both can cause abortion, although it occurs more often with EHV-1. EHV-1 and EHV-4 both can cause respiratory disease, although it is more common with EHV-4.
Slide variation includes some slides with syncytia in the adrenal cortex.
References:
2. Foster RA: Female Reproductive System. In: Pathologic Basis of Veterinary Disease, ed. Mc Gavin MD, Zachary JF, 4th ed., pp1298-1299. Mosby Elsevier, St Louis, Missouri, 2007
3. Jackson TA, Osburn BI, Cordy DR, Kendrick JW: Equine herpesvirus 1 infection of horses: studies on the experimentally induced neurologic disease. Am J Vet Res 38: 709-719, 1977
4. LeBlanc, MM: Abortion. In: Equine Medicine and Surgery, Volume II, eds. Colahan PT, Mayhew IG, Merritt AM, Moore JN, 5th ed., pp1205-1206. Mosby St Louis, Missouri 1999
5. Maxie MG, Youssef S: Nervous system. In: Jubb, Kennedy, and Palmers Pathology of Domestic Animals, ed. Maxie MG, 4th ed., vol. 1, pp. 431-431. Elsevier Limited, St. Louis, MO, 2007
6. Mukaiya R, Kimura T, Ochiai K, Wada R, Umemura T: Demonstration of Equine Herpesvirus-1 gene expression in the placental trophoblasts of naturally aborted equine fetuses. J com Path 123:119-125, 2000
7. Murphy FA, Gibbs EPJ, Horzinek MC, Studdert MJ: Veterinary Virology, pp 301-303. Academic Press, San Diego, California, 1999
8. Paradis MR: Equine respiratory viruses. In: Large Animal Internal Medicine, ed. Smith BP, 2nd ed., pp. 587-588, Mosby, St. Louis, MO, 1996
9. Rooney JR, Robertson JL: Equine Pathology pp 246-248. Iowa State University Press, Ames, Iowa, 1996
10. Schlafer DH, Miller RB: Female genital system. In: Jubb, Kennedy, and Palmers Pathology of Domestic Animals, ed. Maxie MG, 4th ed., vol. 3, pp. 532-533, Elsevier Limited, St. Louis, MO, 2007
11. Slater J: Equine Herpesviruses. In: Equine Infectious Diseases, eds. Sellon DC, Long MT, 1st ed., pp 134-145. Saunders Elsevier, St Louis, Missouri, 2007
12. Smith KC, Borchers K: A study of the pathogenesis of Equid Herpesvirus-1 (EHV-1) abortion by DNA in-situ hybridization. J comp Path 125:304-310, 2001
13. Smith KC, Mumford, JA, Lakhani K: A comparison of Equine Herpesvirus-1 (EHV-1) vascular lesions in the early versus late pregnant equine uterus. J Comp Path 114:231- 247, 1996
14. Smith KC, Whitwell KE, Blunden AS, Bestbier ME, Scase TJ, Geraghty RJ, Nugnet J, Davis-Poynter NJ, Cardwell JM: Equine herpesvirus-1 abortion: atypical cases with lesions largely or wholly restricted to the placenta. Equine Vet J 36:79-82, 2004
15. Szeredi L, Aupperle H, Steiger K: Detection of Equine Herpesvirus-1 in the fetal membranes of aborted equine fetuses by immunohistochemical and in-situ hybridization techniques. J Comp Path 129:147-153, 2003