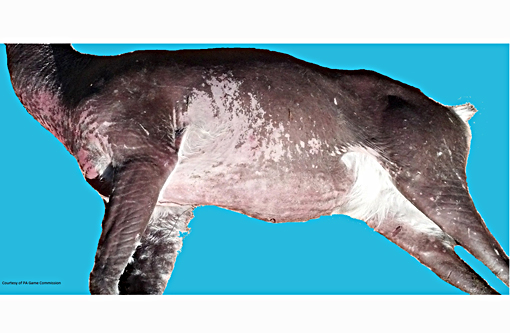
Signalment:
Gross Description:
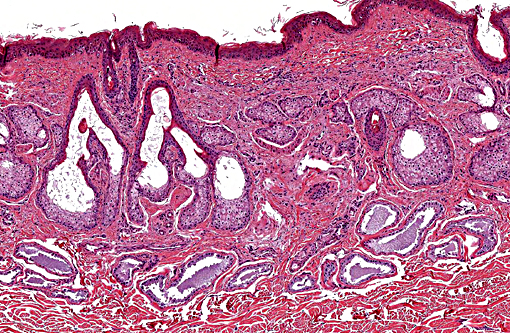
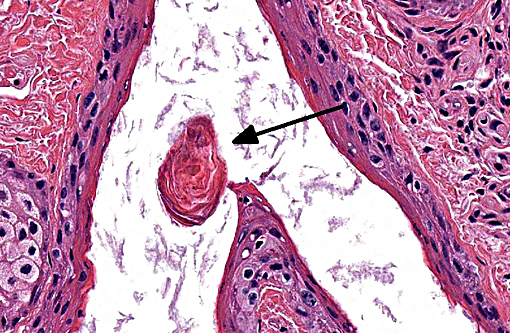
Histopathologic Description:
Morphologic Diagnosis:
Condition:
Contributor Comment:
This condition calls into question the distinction between follicular dystrophy and dysplasia. A true follicular dystrophy suggests a degenerative process and possible association with -Ç-ÿmalnutrition of hair follicle cells.(7) The result is defective and impaired development of hair in spite of a structurally normal hair follicle. In contrast, follicular dysplasias not only feature abnormal hairs but are also accompanied by abnormal hair follicles.(5) While this entity has been classified as a form of follicular dystrophy in the past, we consider a diagnosis of follicular dysplasia appropriate in this case given the occasional abnormal morphology of hair bulbs, and their often aberrant location within the superficial dermis. In prior cases of toothpaste hair disease, a direct link with malnutrition, though suspected, has not been confirmed. A nutrient/mineral assay was not performed on tissues from the present case.
Follicular dysplasias are not uncommon in veterinary medicine. Color dilution alopecia and black hair follicular dysplasia are well described conditions in various breeds of dog and cattle.(1) The former has also been reported in the horse.(4) Non-color dependent follicular dysplasias have also been reported for various dog breeds including Siberian huskies, Irish water spaniels, and Portugese water dogs amongst others. The hairlessness trait of the Sphynx cat and Chinese crested and Mexican hairless dogs is another widely recognized form of follicular dysplasia (congenital hypotrichosis) brought about by the intentional propagation of spontaneous genetic mutations.(1) Hair cycle disorders (including cyclic flank alopecia and follicular arrest) are also classified by some to be follicular dysplasias.
Follicular dystrophies are comparatively quite rare. There are several well characterized follicular dystrophies in mice. Recently, a spontaneous autosomal recessive mutation was discovered on mouse chromosome 2, termed follicular dystrophy (fold), affecting the P/J mouse strain.(3) A primary follicular dystrophy has also been described in a substrain of B6 mice.(11) The phenotype is one of focal alopecia progressing to ulcerative dermatitis and scarring, and is attributed to polymorphism in alcohol dehydrogenase (Adh4) and differential expression of epithelial retinol dehydrogenase (DHRS9), leading to the impaired removal of excess retinol. Yet another follicular dystrophy phenotype has been defined in B6.C mice, resulting from the Angora mouse mutation brought about by a deletion in fibroblast growth factor 5 (Fgf5) gene.(10)
Humans are also subject to follicular dystrophies. An entity known as acquired progressive kinking of hair is an androgen dependent disorder that causes affected hairs of the scalp to resemble pubic hair in morphology.(9) A subset of follicular dystrophies are known to stem from deficiencies in one of various nutrients, including copper (Menkes kinky hair syndrome), sulfur (trichothiodystrophy), and amino acids (Nethertons syndrome).(5)
JPC Diagnosis:
Conference Comment:
Although uncommon, a similar condition has been reported previously in white tailed deer. In the other reported case, hair follicle density was normal, follicles were ectatic and either empty or contained keratin debris and hair shaft fragments, and apocrine ducts were dilated and hair bulbs were abnormal, similar to what was seen in this case. In that case there was normal hair present on the ventral thorax and sebaceous gland hypertrophy and hyperplasia was variably present. There was also mild epidermal hyperplasia and hyperpigmentation, similar to what is seen in this case. The authors of that manuscript went on to discuss the types of congenital hypotrichosis described in cattle including forms which are lethal, forms associated with dental abnormalities and viable hypotrichosis, which shares many similarities with this case.(12) Viable hypotrichosis is reported to affect Guernsey, Jersey, Holstein and Hereford cattle with an autosomal recessive mode of inheritance resulting in dysplastic hair follicles that dont produce hair shafts, generalized alopecia and cystic apocrine glands. Ectodermal defects resulting in congenital alopecia can be restricted to the hair follicle, or also be associated with other ectoderm derived tissues (i.e. teeth, nails).(6)
Formation of a hair follicle and hair shaft involves complex molecular signaling pathways that begin with formation placode, a condensed mesodermal structure which lies just below an epidermal invagination. The placode grows down into the mesenchyme which is followed by differentiation of the follicular mesenchyme, and formation of the dermal papilla and connective tissue sheath, which leads to formation of the hair bulb. The hair bulb is responsible for formation of the hair shaft. When the mesenchymal cells of the dermal papilla become enclosed by keratinocytes, formation of the hair shaft begins. Hair shaft formation is accomplished by the matrix keratinocytes of the hair bulb. Hair shafts are composed of a cortex, which is covered by a cuticle protecting the hair from damage, and many hair shafts have a pigmented medulla. Surrounding the hair shaft is the inner root sheath, which disappears at the level of the follicular infundibulum. The outer root sheath forms at the same time as the inner root sheath and hair follicle, but is not derived from matrix keratinocytes. Downgrowth of the outer root sheath pushes the hair bulb toward the subcutis while matrix keratinocytes are producing the hair shaft and inner root sheath, which grow toward the skin surface. As mentioned above by the contributor, hair follicle dysplasias, which involve defects in the hair follicle and shaft, are often differentiated from the alopecic conditions where the hair follicle appears normal, but the shaft itself is abnormal. Additionally, it is important to differentiate between alopecic conditions with a decreased number of relatively normal hair follicles, vs. alopecic conditions with a normal number of abnormal follicles. Congenital forms of alopecia may involve the epithelial and mesenchymal cells of the hair follicle, but may also involve follicular melanocytes, derived from neuroectoderm, and include color dilution alopecia and black hair follicular dysplasia in dogs.(6)
References:
1. Ginn PE, Mansell JEKL, Rakich PM. Skin and appendages. In: Maxie MG, ed. Jubb, Kennedy and Palmers Pathology of Domestic Animals. Vol 1. 5th ed. Philadelphia, PA: Elsevier Limited; 2007:582-585.
2. Gross, Ihrke, Walder, and Affolter. Skin diseases of the dog and cat. 2nd ed. Ames, IA: Blackwell Science; 2005:518-530.
3. Harris BS, Ward-Bailey PF, Johnson KR, Bronson RT. Follicular dystrophy: a new skin and hair mutation on mouse Chromosome 2. The Jackson Laboratory. Mouse mutant resource. 2013.
4. Henson FMD and Stidworthy MF. Alopecia due to colour-dilute follicular dysplasia in a horse. Equine Veterinary Education. 2003; 15(6): 288-290.
5. Joint Pathology Center. Wednesday Slide Conference; No. 26. April 7, 1999. http://www.askjpc.org/wsco/wsc/wsc98/98wsc26.htm. Accessed April 10, 2015.
6. Mecklenburg L. An overview on congenital alopecia in domestic animals. Vet Dermatol. 2006;17(6):393-410.
7. Mecklenburg L, Linek M, Tobin, DJ, eds. Hair loss disorders in domestic animals. Wiley-Blackwell; 2009:83-85.
8. Miller WH, Griffin CE, Campbell KL. Muller and Kirks small animal dermatology. 7th ed. St. Louis, MO: Elsevier; 2013:597-599.
9. Mortimer PS, Gummer C, English J, Dawber RP. Acquired progressive kinking of hair. Report of six cases and review of literature. Arch Dermatol. 1985; 121(8):1031-3.
10. Sundberg JP, Rourk MH, Boggess D, Hogan ME. Angora mouse mutation: altered hair cycle, follicular dystrophy, phenotypic maintenance of skin grafts, and changes in keratin expression. Vet Pathol. 1997; 34(3):171-9.
11. Sundberg JP, Taylor D, Lorch G, Miller J, et al. Primary follicular dystrophy with scarring dermatitis in C57BL/6 mouse substrains resembles central centrifugal cicatricial alopecia in humans. Vet Pathol. 2011; 48(2): 513-524.
12. Zimmerman TJ, Jenks JA, Holler LD, Jacques CN, Morlock WW. Congenital hypotrichosis in a white-tailed deer fawn from South Dakota. J Wildl Dis. 2004;40(1):145-149.