Signalment:
Gross Description:
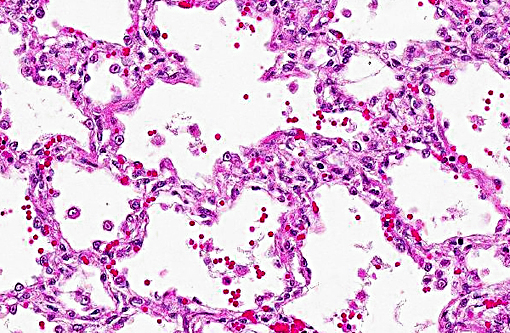
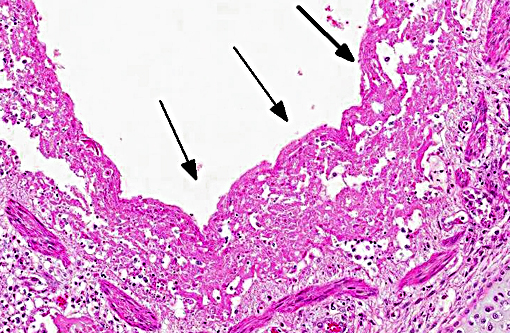
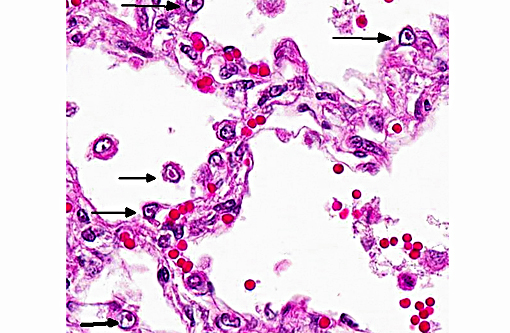
Histopathologic Description:
Morphologic Diagnosis:
Lab Results:
Condition:
Contributor Comment:
Infection can occur in utero, neonatally or in the 2-12 week age range when maternal immunity wanes. Virus is infectious on all mucous membranes but requires contact with nasal or ocular secretions, airborne spread only occurs over short distances.
Infections normally happen intranasal followed by a rapid cytolytic infection of nasal epithelium under an optimal replication-temperature of 37°C and then a secondary spread to conjunctival sac, oropharynx, trachea, bronchi and bronchioli. The typical incubation period is two to four days. Microscopically intranuclear inclusion bodies can be seen in epithelial cells of the described organs named above, but only from the second to seventh day after infection. Transient viremia may occur but is not a prominent feature. Latent infections develop in as many as 80% of infected cats, where the virus is distributed in several tissues of the head. The virus persists in the trigeminal nerve, ganglia of the optic nerve, olfactory bulb and cornea.
Latent carriers have been converted to active virus shedders by giving them corticosteroids for several days or by stress. Also parturition and lactation are stressful events that may trigger activation, which may be an important source of infection for kitten.
The classical rhinotracheitis occurs in kitten within an age range of 6-12 weeks. Severity of clinical signs varies greatly. The most consistent manifestation is rhinitis associated with sneezing and nasal exudation. Low-grade fever occurs but usually clinical signs disappear in 7-14 days. Some kitten can manifest rhinitis, pharyngitis, glossitis, tracheitis, pneumonia, high fever, depression, anorexia and drooling. Mortality occurs among this group of animals.
Besides the classical rhinotracheitis other clinical syndromes can be attributed to FeHV-1 infection. Those may belong to the complex of respiratory diseases as recurrent rhinitis in older cats as a result of reinfection or activation of a latent infection. Also a chronic sinusitis with opportunistic bacteria and mycoplasma that follows an acute upper respiratory infection of FeHV-1 with permanent turbinate and mucosal damage can be seen. Another manifestation can be a chronic conjunctivitis and keratitis that can last for months and is usually mild and bilateral but can become copious and purulent with time. Photophobia is particularly characteristic. Acute or chronic herpetic corneal ulcers can be a troublesome complication or herpesvirus ulcerative dermatitis. 1,3,6
Also problems in context of reproductive events can occur as abortion, which is probably a non-specific phenomenon, and neonatal disease associated with queens that fail to provide maternal immunity or infect their young at birth. There the kitten shows pneumonia without upper respiratory disease after a short period of fading away.1,6 In our case, the two week old kitten didnt show pathologic changes in the upper but in the lower respiratory tract.
| Alphaherpesvirinae: focal lesions in skin and mucosa of respiratory and genital tract; abortion; neonates: necrosis in multiple organs, latency in nerves |
Equine herpesvirus 1: Equine herpesviral abortion, rhinopneumonitis, neurologic disease | horse |
| Equine herpesvirus 3: Equine coital exanthema | horse | |
| Equine herpesvirus 4: Equine rhinopneumonitis | horse | |
| BHV-1: infectious bovine rhinotracheitis/infectious pustular vulvovaginitis/infectious balanoposthitis | cattle | |
| BHV-2: bovine mammilitis virus (bovine herpes mammilitis) | cattle | |
| BHV-5: bovine herpesvirus encephalitis | cattle | |
| SHV-1: Aujezky-¦s disease, Pseudorabies | pig>others | |
| Canine herpesvirus 1: | dog | |
| Feline herpesvirus 1: upper respiratory tract disease (rhinotracheitis) and conjunctivitis (ulcers) | cats | |
| Feline herpesvirus 1: feline herpesvirus ulcerative dermatitis | cats | |
| Gallid herpesvirus-1: Infectious laryngotracheitis (ILT) | chicken | |
| Gallid herpesvirus-2: Marek-¦s disease | chicken | |
| Psittacine herpesvirus: Pacheco's disease | psittacines | |
| Anatid herpesvirus-1: Duck plaque/Duck virus enteritis | ducks, geese, swan | |
| Simplexvirus: HSV-1, HSV-2, HBV, BHV-2 | ||
| Herpesvirus simplex, type 1/type 2 | human & nonhuman primates | |
| Herpesvirus simiae/Herpes B/Cercopithecine HV | rhesus macaques | |
| Simian varicella virus | macaques, AGM, Patas monkeys | |
| Betaherpesvirinae: no cell lysis, karyomegaly, latency in secretory glands, lymphoreticular organs, kidney |
HHV-5, HHV-6, MCMV-1 | humans |
| Porcine herpesvirus 2: porcine cytomegalovirus disease/Inclusion body rhinitis | porcine | |
| Cytomegalovirus | humans + nonhuman primates | |
| Gammaherpesvirinae: primates: lymphoproliferative disease, latency in lymphoid tissue |
EHV-2 | |
| EHV-5 | ||
| BHV-4: bovine herpes mammary pustular dermatitis | cattle | |
| OHV-2/AHV-1: malignant catarrhal fever | various ruminants | |
| Epstein-Barr virus (lymphocryptovirus-gamma 1) | primates | |
| Kaposi-sarcoma-associated herpesvirus/human herpesvirus-8 (KSHV/HHV8) (Rhadinovirus-gamma 2) | primates | |
| Deltaherpesvirinae | Anatid herpesvirus-1: duck plague | |
| SHV-2: Einschlussk+â-¦rperchenkrankheit | pig | |
| Karpfenpocken | fish | |
| ? | Koi-herpesvirus (KHV, carp nephritis and gill necrosis virus, CNGV, Cyprinid-Herpesvirus-3, CyHv-3) | fish |
JPC Diagnosis:
Conference Comment:
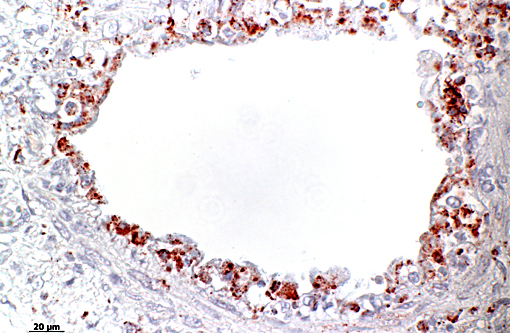
Differential diagnosis discussed for this lesion included feline calicivirus and high pathogenicity avian influenza, although viral inclusions are not a histologic feature of either entity. Feline calicivirus (FCV), in addition to causing rhinitis, stomatitis and conjunctivitis, can also cause an interstitial pneumonia and necrotizing bronchiolitis with secondary bacterial infection. The clinical and pathologic findings in FHV and FCV are similar in some aspects, and together account for the majority of feline respiratory diseases. FCV can also result in a transient lameness due to a self-limiting arthritic condition, termed limping kitten syndrome. There is a virulent form of FCV, described as a systemic hemorrhagic syndrome with high mortality(5), in which cats present clinically with diarrhea, pneumonia, edema and hemorrhage. Unlike the more virulent strain where viral anti-gen is present systemically in both endothelial cells and epithelial cells, in the less virulent more ubiquitous form, viral antigen is generally only present in oral mucosa and respiratory epithelium.(7)
Cats can also become infected with highly pathogenic avian influenza virus (HPAI) H5N1 from infected wild birds. The primary respiratory lesions in H5N1-infected cats includes bronchointerstitial pneumonia and bronchiolar necrosis, but significant disease outside the respiratory system, including multifocal hepatic necrosis, is also a noted and described finding.(4,8) To date, horizontal transmission between domestic cats has not been documented in the wild(4) but has been demonstrated experimentally with excretion from both the respiratory and digestive tracts.(8) Additionally, cats have also been shown to be susceptible to other forms of influenza virus such as H1N1 from humans as well as low pathogenicity avian influenza viruses.(2)
References:
1. Appel, MJG. Virus infections of carnivores. Amsterdam, The Netherlands: Elsevier Science Ltd; 1987.
2. Driskell EA, Jones CA, Berghaus RD, et al. Domestic cats are susceptible to infection with low pathogenic avian influenza viruses from shorebirds. Vet Pathol. 2012;50(1):39-45.
3. Gross TL, Ihrke PJ, Walder EJ, Affolter VK. Skin Diseases of the Dog and Cat. Clinical and Histopathological Diagnosis. 2nd ed. Ames, IA: Blackwell; 2005.
4. Klopfleisch R, Wolf PU, Uhl W et al. Distribution of Lesions and Antigen of Highly Pathogenic Avian Influenza Virus A/Swan/Germany/R65/06 (H5N1) in Domestic Cats after Presumptive Infection by Wild Birds. Vet Pathol. 2007;44:261-268.
5. Lopez A. Respiratory system, mediastinum, and pleurae. In: McGavin MD, Zachary JF, eds. Pathologic Basis of Veterinary Disease. 5th ed. St. Louis, MO: Mosby Elsevier; 2012:472-474.
6. Maxie MG. Jubb, Kennedy and Palmers Pathology of Domestic Animals. 5th ed. Philadelphia, PA: Elsevier Saunders; 2007.
7. Pesavento PA, Murphy BG. Common and emerging infectious disease in the animal shelter. Vet Pathol. 2014;51(2):478-491.
8. Rimmelzwaan GF, van Riel D, Baars M, et al. Influenza A virus (H5N1) infection in cats causes systemic disease with potential novel routes of virus spread within and between hosts. Am J Pathol. 2006;168(1):176-83.