Signalment:
Nine-year-old owl monkey (
Aotus sp.).Acute onset of weakness and depression with hypothermia, weight loss and anemia (PCV =18%). The animal responded slightly to palliative therapy but then was found dead.
Gross Description:
At necropsy the animal was in poor body condition. The scent gland was enlarged and both kidneys were pale, firm and pitted.
Histopathologic Description:
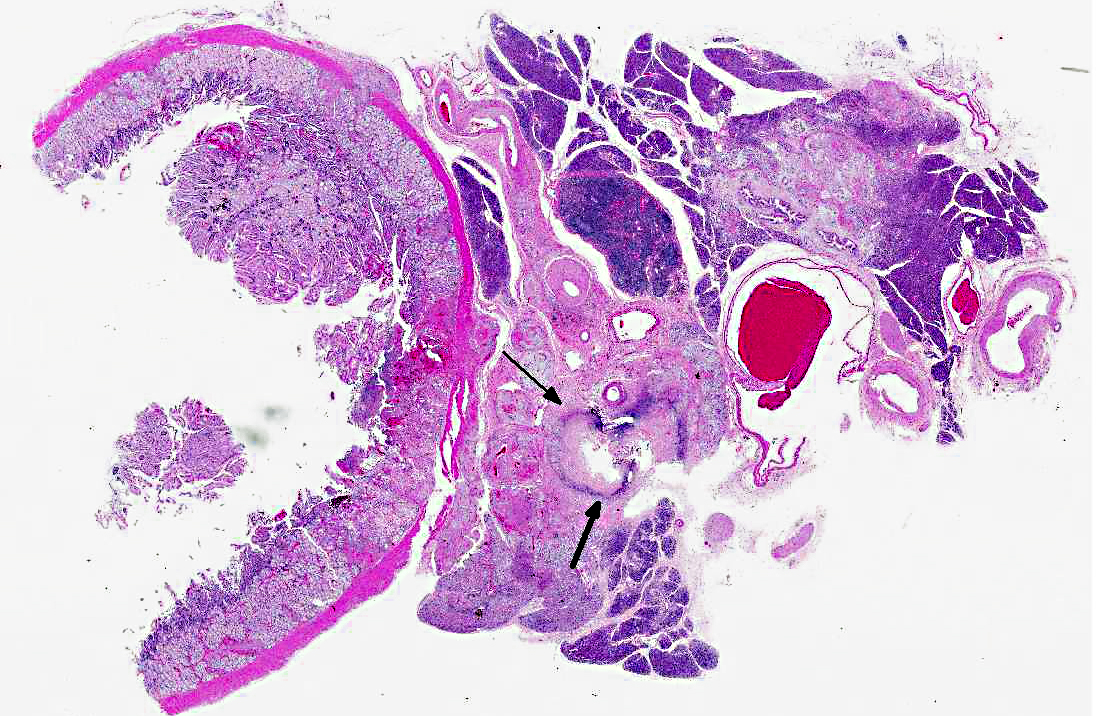
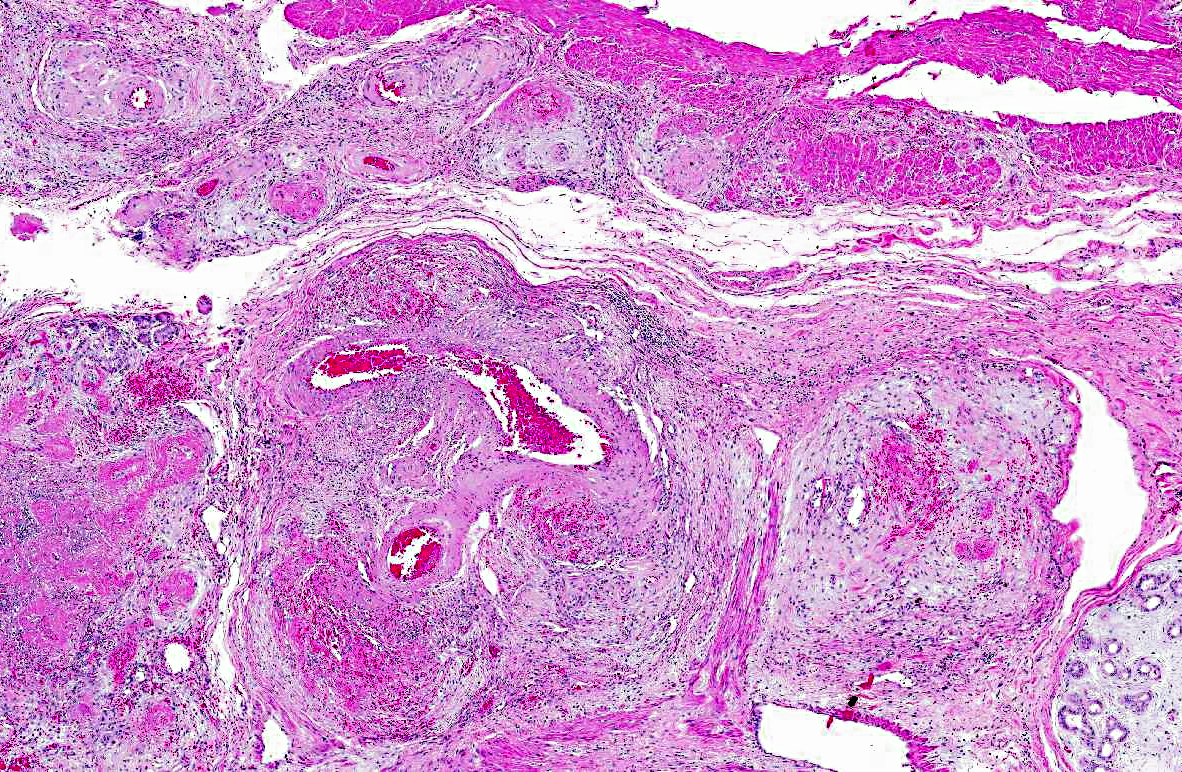
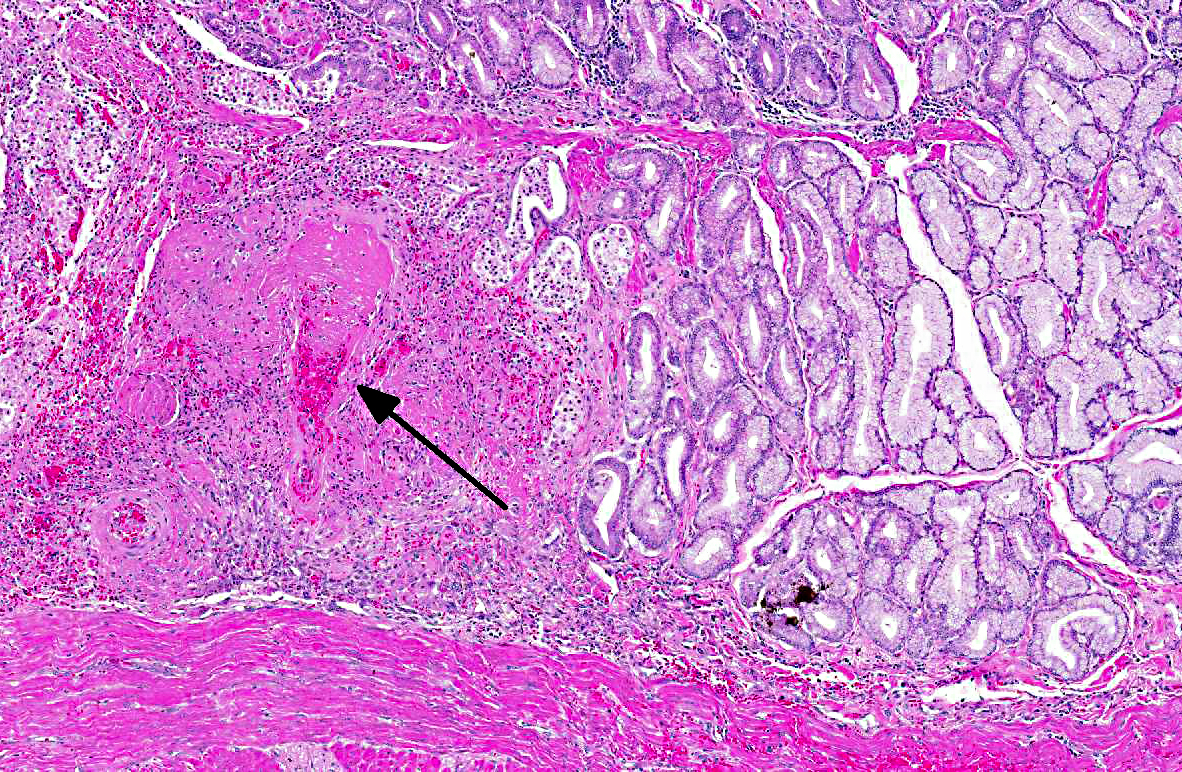
Small and medium sized arteries of the pancreas and duodenum are infiltrated and disrupted by low numbers of neutrophils, macrophage, and lymphocytes. Vessel walls are often necrotic and expanded by abundant fibrin and hemorrhage. Occasionally the entire vessel is replaced by fibrin and necrotic debris that completely occludes the vessel lumen (thrombus). Often the tunica intima is proliferative. The tunica adventitia and perivascular connective tissue contain moderate numbers of macrophages, fewer lymphocytes and fibroblasts. Both in the duodenum and more extensively in the pancreas there is coagulative necrosis. Multifocally pancreatic lobules contain tubules and ducts that are surrounded by a loosely arranged, proliferative, edematous stroma. Other tissues affected in this case with a necrotizing arteritis include the heart, gallbladder, jejunum, and adrenal gland.Â
Morphologic Diagnosis:
1. Pancreas; duodenum: Arteritis, fibrinonecrotic, multifocal, marked, with coagulative necrosis.
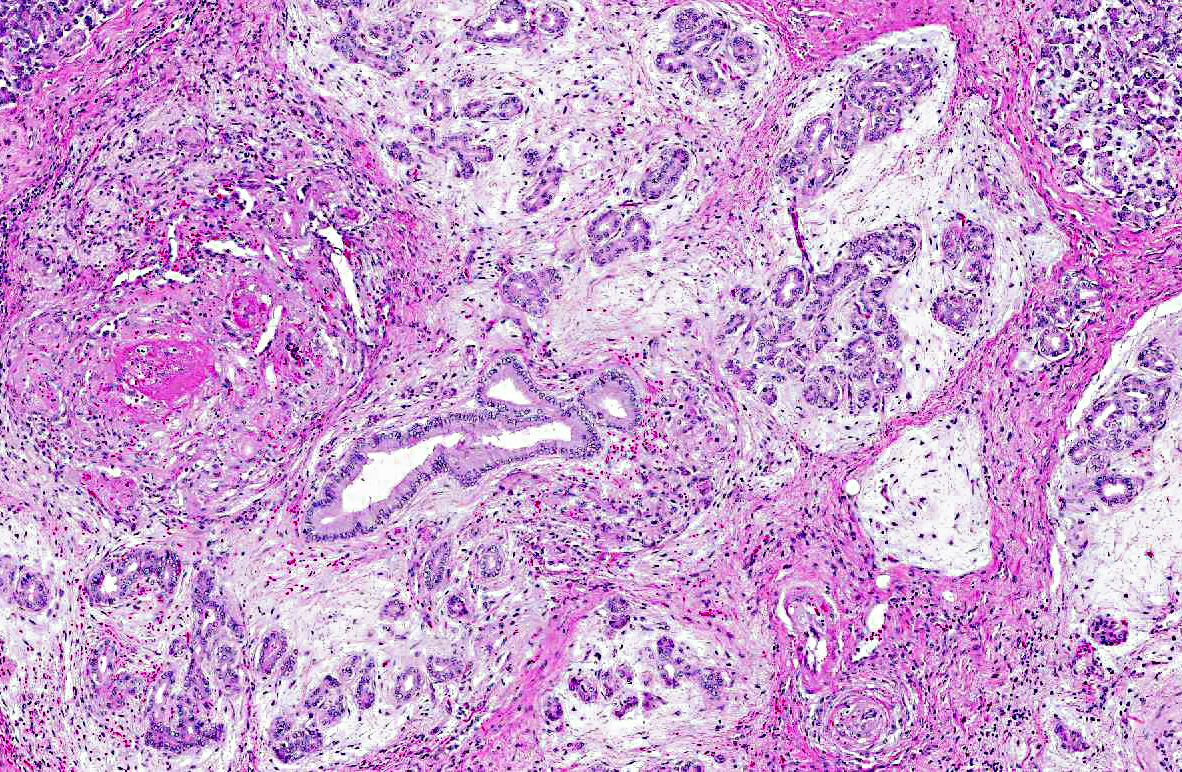
2. Pancreas: Mesenchyme proliferation, peritubular and periductal, multifocal, moderate.
Lab Results:
PCV= 18%.
Condition:
Polyarteritis nodosa
Contributor Comment:
In humans polyarteritis nodosa (PAN) is a systemic vasculitis of small and medium-sized arteries, most often affecting young adults. It typically involves renal and gastrointestinal vessels with a predilection for branching points but spares the lungs.(5) It has been reported in rats, mice, dogs, cats, pigs, and non-human primates, specifically cynomolgus macaque.(4) In rats it typically involves the mesentery, pancreas, and testis of aged Sprague-Dawley rats, with a higher incidence in males.(3) In dogs it is most often reported in beagles, referred to as beagle pain syndrome, affecting vessels of the mediastinum, cervical spinal cord, and heart.(2) To our knowledge, this is the first case involving an
Aotus monkey. The tissues affected and histologic characteristics of the lesion make it similar to the case previously reported in a cynomolgus.(4) The lesion typically involves a necrotizing vasculitis with transmural infiltration by neutrophils, eosinophils and fewer macrophages often with fibrinoid necrosis of the vessel wall. Eventually the vessel wall is replaced by a fibrous thickening. In humans renal involvement is often the cause of death. Interestingly approximately 30% of humans with PAN have hepatitis B antigen in the serum. Characteristically various stages of chronicity exist simultaneously, even within the same vessel.(5) The pathogenesis of PAN is unknown but is suspected to be immunological. Patients with PAN have elevations of serum interferon- gamma and interleukin-2. Immunohistochemistry showed inflammatory cell infiltrates are mainly macrophages and CD4+ T cells.(4) Clinical response to immunosuppressive therapy further supports an immunologic-based pathogenesis. In this case the histologic lesions in the pancreas and duodenum are typical of PAN. Within the pancreas, however, there are lobules that contain tubules, and to a greater extent ducts, that are surrounded by a loosely arranged, edematous
stroma. In more severely affected areas this stroma appears proliferative. To our knowledge this change has not been reported in association with PAN. We speculate that these areas are proliferative due to the many cytokines and growth factors being released in the local area from the extensive necrosis and inflammation. Why only peritubular and periductal areas are affected is not known and other possible explanations for this morphologic characteristic cannot be excluded.Â
JPC Diagnosis:
1. Duodenum: Arteritis, proliferative and necrotizing, chronic, multifocal, severe, with mucosal necrosis.
2. Pancreas: Arteritis, proliferative and necrotizing, chronic, multifocal, severe, with acinar necrosis, atrophy and loss and ductal hyperplasia.Â
Conference Comment:
The contributor provided an excellent review of polyarteritis nodosa. The conference moderator noted the presence of autolysis of the common bile duct, and cautioned students not to mistake this for a focus of lytic necrosis. Participants noted the areas of necrosis within the duodenum and pancreas correspond to areas of arteritis. The occurrence of abundant stroma within the pancreas that the contributor described and speculated to be due to cytokines and growth factors in areas of inflammation and necrosis led to discussion of the induction of mesenchymal tissue due to acinar loss. This phenomenon that occurs during acute and chronic pancreatitis is thought to be due to the release of TGF-β (which acts as the main stimulator), TNF-α, IL-1 and IL-6, and PDGF (which acts as the main mitogen), from acinar cells, inflammatory cells and platelets. These factors induce the activation and proliferation of periacinar myofibroblasts (aka pancreatic stellate cells) by inducing them to transform from their fat-storing phenotype to their matrix-producing phenotype, which plays a role in tissue repair and result in pancreatic fibrosis.(1)
References:
1. Bachem MG, Xhou Z, Zhou S, et al. Role of stellate cells in pancreatic fibrinogensis associated with ace and chronic pancreatitis.Â
J Gastroenterol and Hepatol. 2006; 21(s3):s92-s96.Â
2. Catharine J, Scott-Moncrieff R, Snyder PW, et al. Systemic necrotizing vasculitis in nine young Beagles.Â
JAVMA. 1992;201:1553-1557.Â
3. Percy DH, Barthold SW. In:
Pathology of Laboratory Rodents and Rabbits. 2nd ed. Ames, IA: lowa State Press;2001:153.
4. Porter BF, Frost P, Hubbard Gf. Polyarteritis nodosa in a cynomolgus macaque (
Macaca fascicularis).Â
Vet Pathol. 2003;40:570-573.
5. Schoen FJ. Blood vessels. In: Kumar V, Abbas AK, Fausto N, eds.Â
Robbins Pathologic Basis of Disease. 7th ed. Philadelphia, PA: Elsevier Inc; 2005:539.