Signalment:
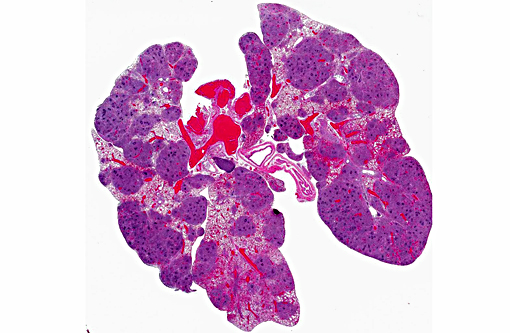
Gross Description:
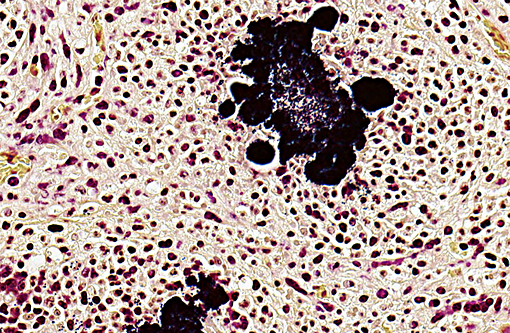
Histopathologic Description:
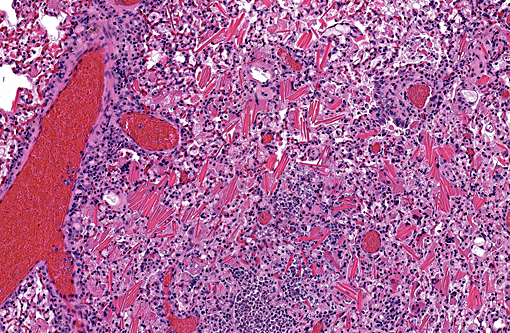
Gram Stain: Lung: Bacterial cocci are gram positive, consistent with Staphylococcus sp.
Morphologic Diagnosis:
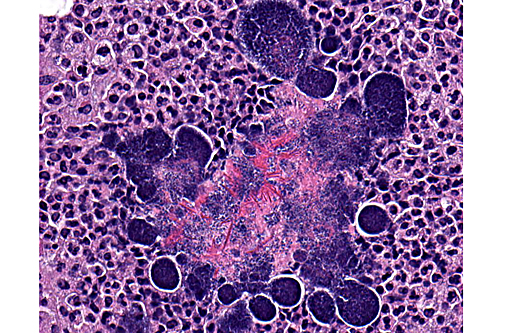
Lung: Diffuse, chronic-active, severe, pyogranulomatous pneumonia with intralesional bacteria (pulmonary botryomycosis).
Lung: Multifocal, subacute to chronic, moderate, eosinophilic crystalline pneumonia.
Condition:
Contributor Comment:
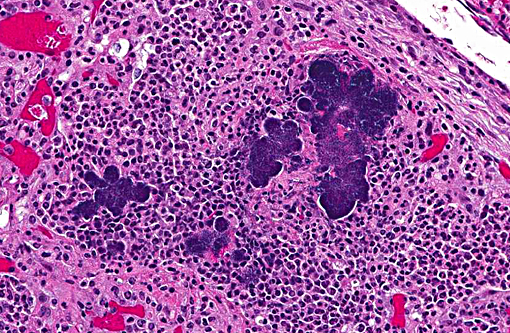
Eosinophilic crystalline pneumonia, sometimes called acidophilic macrophage pneumonia or simply intracellular eosinophilic crystals is an idiopathic granulomatous pneumonia that occurs spontaneously in many strains of mice, namely the C57BL/6 and strains on a C57BL/6 background.(5,10) This condition is also common among Swiss mice, B6C3F1 mice used by the National Institutes of Health and the National Toxicology Program and 129 strains, particularly the 129S4/SvJae strain in which severe cases resulting in respiratory distress or death have been reported.(1,5,8) Largely considered a background lesion, eosinophilic crystalline pneumonia can range from subclinical to fatal and is characterized by focal to diffuse accumulation of eosinophilic crystals composed predominantly of Ym1 protein, a chitinase-like protein expressed in neutrophil granule products and active macrophages. As in this case, crystals are readily apparent within macrophages, multi-nucleated giant cells, alveolar spaces and airways.(1,5,8,10) The function of Ym1 protein is not completely understood, but may play a role in host immune defense, tissue repair and hematopoiesis.(5,10)
The Cybb transgenic mouse was designed as a model for developing new treatments for chronic granulomatous disease (CGD); JAX developed this particular transgenic strain targeting the gene involved in X-linked CGD. As a result, male hemizygotes tend to have an increased susceptibility to infection with S. aureus due to a lack of phagocyte superoxide production.(7) Given the strain specific increased susceptibility to bacterial infection and apparent pulmonary botryomycosis, staphylococcal infection is the most likely etiology in this case. Large colonies of Gram-positive cocci are consistent with Staphylococcus sp., most likely S. aureus or S. xylosis. We were unable to definitively identify the bacterial agent since bacterial culture was not performed. As previously mentioned, eosinophilic crystalline pneumonia is a common spontaneous lesion in the C57BL/6 mouse, which was the background strain used to manufacture this particular transgenic. However, since this condition may also occur in conjunction with other diseases, it is difficult to determine whether the eosinophilic crystalline pneumonia developed prior to the pulmonary botryomycosis, as a spontaneous disease possibly predisposing the animal to infection, or developed secondary to, or in conjunction with, the pulmonary botryomycosis.
JPC Diagnosis:
1. Lung: Pneumonia, pyogranulomatous, multifocal to coalescing, severe, with Splendore-Hoeppli material and numerous colonies of cocci.
2. Lung: Pneumonia, histiocytic, diffuse, moderate with extracellular and intra-histiocytic eosinophilic protein crystals.
Conference Comment:
The differential diagnosis for a nodular mass composed of pyogranulomatous inflammation includes filamentous bacterial or fungal infections, which result in the formation of a mycetoma.(4) The organisms are not always readily visualized on H&E stained sections and therefore may require special histochemical stains for differentiation. Botryomycosis may also occur in the mammary gland of ruminants associated with staphylococcal mastitis, and in other large animals secondary to post castration staphylococcal infection.(2)
The conference description was very similar to that provided by the contributor. Additional features described and discussed include: multifocal expansion of alveolar septa by fibrous connective tissue; an exudate composed of neutrophils and histiocytes within larger airways, which may represent reflux from alveolar spaces; and the presence of Splendore-Hoeppli phenomenon within the center, rather than the periphery, of the bacterial colonies. Medullary sinuses of a hilar node contain numerous plasma cells, with few neutrophils and macrophages, which attest to the longstanding nature of this infection. There was discussion regarding the route of infection in this case; most participants favored an airway route, as the bronchi and bronchioles are particularly affected by the inflammatory process and the vessels are relatively spared.
References:
1. Dixon D, Herbert RA, Sills RC, Boorman GA. Lung, Pleura, Mediastinum. In: Maronpot RR, ed. Pathology of the Mouse. Vienna, IL:Cache River Press; 1999:318.
2. Foster RA. Male reproductive system. In: McGavin MD, Zachary JF, eds. Pathologic Basis of Veterinary Disease. 5th ed. St. Louis, MO: Mosby Elsevier; 2012:1144.
3. Ginn PE, Mansell JEKL, Rakich PM. Skin and appendages. In: McGavin MD and Zachary JF, eds. Pathologic Basis of Veterinary Disease. 4th ed. Mosby Elsevier, St. Louis, MO; 2007:691.
4. Hargis AM, Ginn PE. The Integument. In: McGavin MD, Zachary JF, eds. Pathologic Basis of Veterinary Disease. 5th ed. St. Louis, MO: Mosby Elsevier; 2012:1034.
5. Hoenerhoff MJ, Starost MF, Ward JM. Eosinophilic Crystalline Pneumonia as a Major Cause of Death in 129S4/SvJae Mice. Vet Pathol. 2006; 43:682-688.
6. I-B02 - Botryomycosis - fibrovascular tissue rat; Veterinary Systemic Pathology Online (VSPO); The Joint Pathology Center (JPC); http://www.askjpc.org/vspo/show_page.php?id=285; 2014; accessed: February 6, 2016.
7. Mouse strain datasheet, Strain Name: B6129S-Cybbtm1Din/J; JAX Mouse Database. The Jackson Laboratory; http://jaxmice.jax.org/strain/002365.html; 2016; accessed: February 6, 2016.
8. Percy DH and Barthold SW. Mouse: Acidophilic Macrophage Pneumonia/Epithelial Hyalinosis. In: Percy DH and Barthold SW, eds. Pathology of Laboratory Rodents and Rabbits. 3rd ed. Ames, IA: Blackwell Publishing; 2007:95.
9. Percy DH and Barthold SW. Mouse: Staphylococcal Infection. In: Percy DH and Barthold SW, eds. Pathology of Laboratory Rodents and Rabbits. 3rd ed. Ames, IA: Blackwell Publishing; 2007:74-76.
10. Renne R, Brix A, Harkema J, Herbert R, Kittel B, Lewis D, March T, Nagano K, Pino M, Rittinghausen S, Rosenbruch M, Tellier P, Wohrmann T. Proliferative and Nonproliferative Lesions of the Rat and Mouse Respiratory Tract. Tox Pathol. 2009; 37:5S-73S.
11. Valentine BA, McGavin MD. Skeletal muscle. In: McGavin MD, Zachary JF, eds. Pathologic Basis of Veterinary Disease. 5th ed. St. Louis, MO: Mosby Elsevier; 2012:892.