Signalment:
Gross Description:
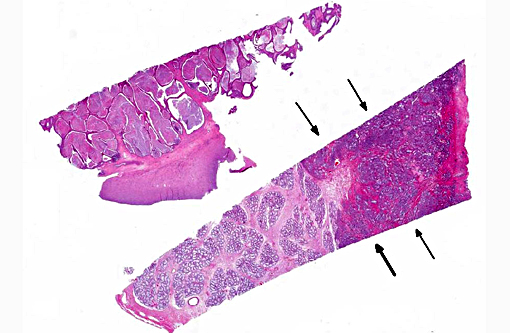
Histopathologic Description:
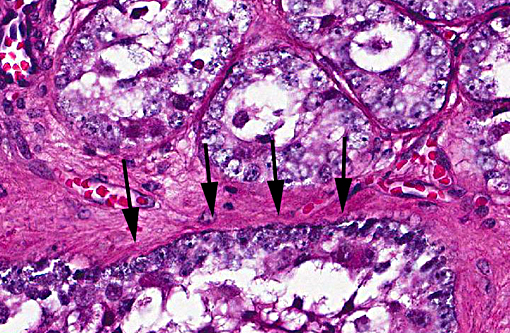
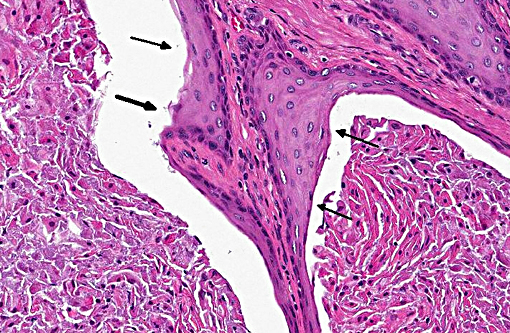
Prostate: Prostatic acini are diffusely and severely distended by abundant mixtures of keratinaceous debris, sloughed epithelial cells, granular debris, and acicular clefts. The normal glandular epithelium is diffusely replaced by a well-differentiated, 3-4 cell thick, stratified squamous epithelium (metaplasia). In some sections, scattered acini are ruptured and infiltrated by numerous foamy macrophages and neutrophils.
Morphologic Diagnosis:
Retained testicle: Sertoli cell tumor
Prostate: Squamous metaplasia, diffuse, severe
Condition:
Contributor Comment:
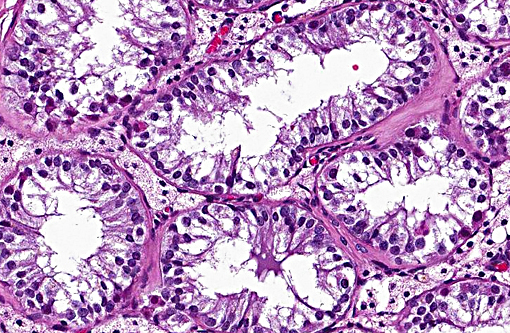
Growth of Sertoli cell tumors may be intra-tubular, as in this case, or diffuse. Microscopic characteristics of intratubular neoplasms include a dense collagenous stroma surrounding semi-niferous tubule-like structures that contain polygonal to elongate cells, with eosinophilic, foamy to vacuolated cytoplasm. In some areas, the neoplastic cells palisade along the stroma. Mitotic figures are few. Cells appear discrete and spherical in diffuse tumors and show little or no tendency to palisade. The dense stroma and palisading cells usually differentiates Sertoli cell tumors from seminomas or interstitial cell tumors. In well differentiated tumors, neoplastic cells resemble normal Sertoli cells, with basally located nuclei and frequent cytoplasmic lipid droplets and globules. Cells of less differentiated tumors exhibit disordered growth and increased pleomorphism.
The incidence of Sertoli cell tumors is 20 times higher in cryptorchid dogs and up to 30% of affected dogs manifest signs of hyperestrinism. Especially with larger tumors, production of excessive estrogen and inhibin result in feminization, including attraction of male dogs, reduced libido, testicular and penile atrophy, preputial swelling, perineal hernia, gyneco-mastia, redistribution of fat, and symmetrical, often ventral, alopecia. Squamous metaplasia of the prostate gland and suppurative prostatitis may lead to dysuria. Estrogenic depression of bone marrow can result in anemia, thrombocytopenia and granulocytopenia, predisposing the dog to hemorrhage and infection. Castration of affected dogs generally results in recovery and regression of associated changes. Most Sertoli cell tumors are benign, but metastasis can occur to regional lymph nodes and distant organs. Metastatic tumors can also be hormonally active.
JPC Diagnosis:
Testicle: Sertoli cell tumor. Testicle, seminiferous tubules and interstitial cells: Hypoplasia, diffuse, severe.
Prostate: Squamous metaplasia, diffuse, severe.
Conference Comment:
The feminizing effect of Sertoli cell tumors was also discussed during the conference and mentioned above by the contributor. Inhibin inhibits the release of GnRH from the hypothalamus, and ultimately the release of LH and FSH from the anterior pituitary, which affects the production of estrogen and testosterone, resulting in the feminizing effects.(1) In addition to squamous metaplasia of the prostate gland, glandular hyperplasia can also occur. The squamous metaplasia is not considered to be a pre-neoplastic change in this case, unlike in other locations with other causes, such as the lung of smokers. In metaplastic tissue, specialized epithelium is frequently replaced by less specialized epithelium, it is often a reversible change, and the mechanism varies with cause. Another example of squamous metaplasia occurs in the esophageal glands of various avian species in response to vitamin A deficiency, the exact mechanism of which is unclear.(6)
Following discussion of the neoplasm in this section, participants discussed the non-neoplastic portion of the testicle. The lack of developing spermatogonia, small volume of remaining seminiferous tubules and decreased numbers of interstitial cells were considered to be hypoplastic changes due to the abdominal location of the testis, although the hormonal contribution of the Sertoli cell tumor was considered as a contributing factor non-viability of the adjacent seminiferous tubules.
Slide variation was noted with some slides demonstrating more prominent acicular cholesterol clefts and degree of inflammation.
References:
1. Foster RA. Male reproductive system. In: McGavin MD, Zachary JF, eds. Pathologic Basis of Veterinary Disease. 5th ed. St. Louis, MO: Mosby Elsevier; 2012:1130-1145.
2. Foster RA. Male reproductive system. In: McGavin MD, Zachary JF, eds. Pathologic Basis of Veterinary Disease. 4th ed. St. Louis, MO: Mosby Elsevier; 2007:1317-1348.
3. Foster RA, Ladds PW. Male genital system. In: Maxie MG, ed. Jubb, Kennedy and Palmers Pathology of Domestic Animals. 5th ed. Philadelphia, PA: Elsevier Saunders; 2007:565-619.
4. Kennedy PC, Cullen JM, Edwards JF et al. World Health Organization Histological classification of tumors of the genital system of domestic animals. Washington DC: Armed Forces Institute of Pathology; 1998:15-18.
5. MacLachlan NJ, Kennedy PC. Tumors of the genital systems. In: Meuten DJ, ed. Tumors in Domestic Animals. 4th ed. Ames, IA: Blackwell Publishing; 2002:547-573.
6. Myers RK, McGavin DM, Zachary JF. Cellular adaptations, injury, and Death: Morphologic, biochemical and genetic bases. In: McGavin MD, Zachary JF, eds. Pathologic Basis of Veterinary Disease. 5th ed. St. Louis, MO: Mosby Elsevier; 2012:29-30.