Signalment:
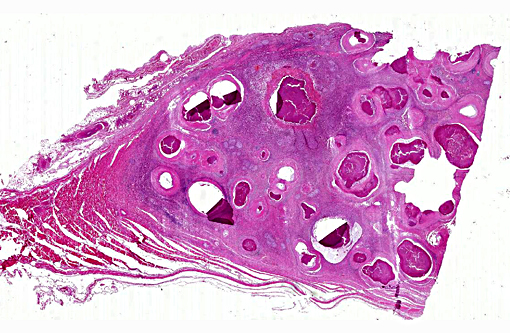
Gross Description:
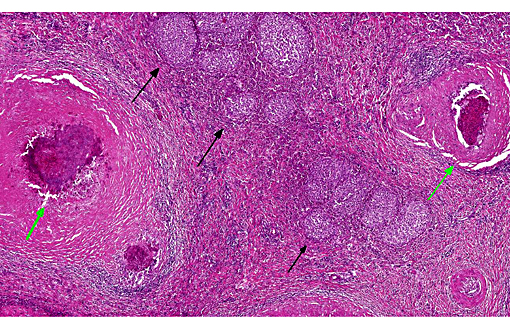
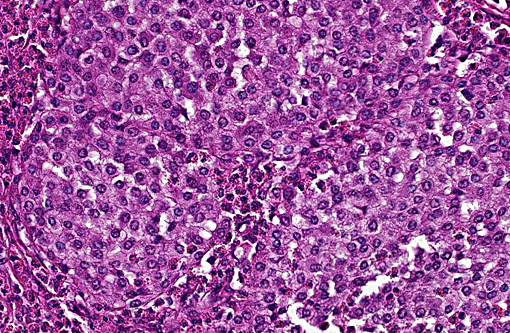
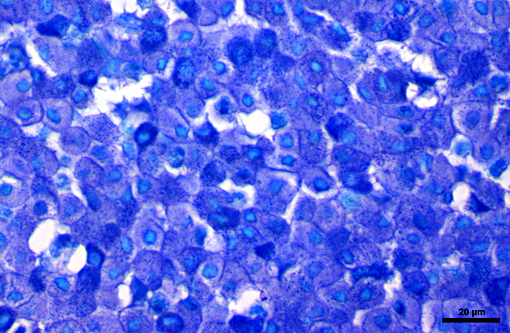
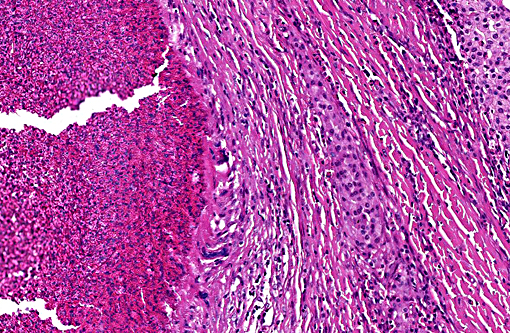
Histopathologic Description:
Morphologic Diagnosis:
Condition:
Contributor Comment:
Mast cells are involved in numerous inflammatory conditions, especially type I hypersensitivity. After binding of IgE to membrane receptors and cross-linking of immunoglobulins on these receptors by specific antigens, degranulation of stored primary mediators and de novo synthesis of secondary mediators occurs. Granules contain, among others, primary mediators like biogenic amines (histamine, adenosine and serotonin), chemotactic factors (eosinophil chemotactic factor and neutrophil chemotactic factor) and multiple enzymes. Secondary mediators include leukotrienes, prostaglandins, platelet activating factor and several cytokines (TNF-alpha, IL-1, 3, 4, 5 and 6). In summary, these mediators generate edema, mucus secretion, smooth muscle spasm, and the recruitment of additional leukocytes, especially eosinophils.
In the dog, mast cell tumors are common, accounting for 15 20 % of all skin tumors, being additional the most frequent malignant or potentially malignant cutaneous neoplasia.2 Well differentiated neoplastic cutaneous mast cells typically form diffuse loose sheets or densely packed cords with intermingled mature eosinophils. They are never encapsulated or even well demarcated. Ulceration may occur in large tumors after traumatization due to pruritus triggered by mediators degranulated from mast cells. The proto-oncogene c-KIT has been discussed as a possible prognostic factor in canine cutaneous mast cell tumors with mutations or aberrant expression of c-KIT turned out to be poor predictors.(13)
In horses, mast cell tumors are relatively rare, covering only 3 7% of cutaneous and muco-cutaneous neoplasms.(11) Commonly affected horses are male without an apparent breed predilection and the mean age is 7 - 11 years with a range from 1 to 30 years.(1, 5,6) In a recent large study Arabians were affected more frequently.(1) Tumors typically present as solitary immovable nodules on the head, trunk, neck, and limbs, where they are often found close to joints.(11) The tumors may be hyperpigmented, alopecic or ulcerated.(3) Nevertheless, most tumors are neither painful nor pruritic.(3) Usually, mast cell tumors in the horse behave benign and excision is curative as recurrence is uncommon.(5)
Histologically, cutaneous mast cell tumors in horses often differ from their counterparts in the dog. Foci of necrosis, severe eosinophilic infiltration with subsequent collagenolysis, reactive fibrosis and dystrophic mineralization are often present. In this respect mast cell tumors may resemble equine collagenolytic granulomas or verminous dermatitis due to onchocerciasis or habronemiasis.(2) Typically, neoplastic mast cells are well-differentiated and arranged in multifocal small nests or coalescing nodules.(11) Malignant tumors are rare, presenting with loss of cellular differentiation and high mitotic index.(8) Local but not visceral metastasis has been documented.(8,9) In contrast to canine mast cell tumors, neither changed c-KIT staining pattern nor histologic features were associated with an unfavorable clinical course.(1)
JPC Diagnosis:
Conference Comment:
In canine mast cell tumors, the proto-oncogene c-kit encodes for the transmembrane receptor tyrosine kinase (RTK) KIT, which normally plays a role in transcription of genes that control mast cell growth and survival, but also plays a role in tumor development in some tumors. A percentage of canine mast cell tumors demonstrate mutations in c-kit, including internal tandem duplication (ITD) mutations in exon 11, which result in activation of KIT in the absence of ligand binding, as well as point mutations in c-kit extracellular domains at exons 8 and 9.
Specific mutations in the gene that encodes the KIT RTK results in constitutive activation of the RTK (via tyrosine phosphorylation) and cell proliferation in the absence of growth factor stimulation. In general tumors with ITD mutations in c-kit exon 11 are known to behave more aggressively.(7) However, the presence of these specific mutations also results in a more favorable response to treatment with tyrosine kinase inhibitors (TKIs) as compared to mast cell tumors without these specific mutations. Additionally, it has also been demonstrated that c-kit mutations are conserved between the primary tumor and lymph node metastasis, indicating that both can be used for mutational testing to select patients that will respond to favorably to TKIs.(4) Mutations in KIT genes may also result in changes in cellular proteins involved in motility and the cytoskeleton.(10) To our knowledge, analogous studies on equine mast cell tumors and their response to TKI therapy have not been published at the time of this conference. Additionally, another study of canine mast cell tumors showed that in addition to KIT, cellular expression patterns of the RTK vascular endothelial growth factor receptor 2 (VEGFR2) may also be predictive of a poorer prognosis.(12)
References:
1. Clarke L, Simon A, Ehrhart EJ, Mulick J, Charles B, Powers B, Duncan C. Histologic characteristics and KIT staining patterns of equine cutaneous mast cell tumors. Vet Pathol. 2014;51: 560-562.
2. Ginn PE, Mensett JEKL, Rukich PM. In: Maxie GM, ed. Jubb, Kennedy, and Palmer's Pathology of Domestic Animals. 5th ed. Edinburgh: Elsevier Saunders; 2007:553-781.
3. Mair TS, Krudewig C. Mast cell tumours (mastocytosis) in the horse: A review of the literature and report of 11 cases. Equine Veterinary Education. 2008; 20:177-182.
4. Marconato L, Zorzan E, Giantin S, et al. Concordance of c-kit mutational status in matched primary and metastatic cutaneous canine mast cell tumors at baseline. J Vet Intern Med.2014;28:547-553.
5. McEntee MF: Equine cutaneous mastocytoma: morphology, biological behavior and evolution of the lesion. J. Comp Path. 1991;104:171-178.
6. Millward LM, Hamberg A, Mathews J, Machado-Parrula C, Premanandan C, Hurcombe SDA, Radin MJ, Wellman ML: Multicentric mast cell tumors in a horse. Vet Clin Pathol. 2010;39:365-370.
7. Letard S, Yang Y, Hanssens K, et al. Gain-of-function mutations in the extracellular domain of KIT are common in canine mast cell tumors. Mol Cancer Res. 2008;6(7): 1137-1145.
8. Reppas GP, Canfield PJ. Malignant mast cell neoplasia with local metastasis in a horse. New Zealand Veterinary Journal. 1996;44: 22-25.
9. Riley CB, Yovich JV, Howell JM. Malignant mast cell tumors in horses. Australian Veterinary Journal. 1991;68:346-347.
10. Schlieben P, Meyer A, Weise C, et al. Tandem duplication of KIT exon 11 influences the proteome of canine mast cell tumours. J Comp Path. 2013;148:318-322.
11. Scott D, Miller W. Equine Dermatology. St. Louis, MO: Elsevier;2003.
12. Thompson JJ, Morrison JA, Pearl DL, et al. Receptor tyrosine kinase expression profiles in canine cutaneous and subcutaneous mast cell tumors. Vet Pathol. Oct 12, 2015; online first:1-14.
13. Webster JD, Yuzbasiyan-Gurkan V, Miller RA, Kaneene JB, Kiupel M. Cellular proliferation in canine cutaneous mast cell tumors: Associations with c-KIT and its role in prognostication. Vet Pathol. 2007; 44: 298-308.