Signalment:
Gross Description:
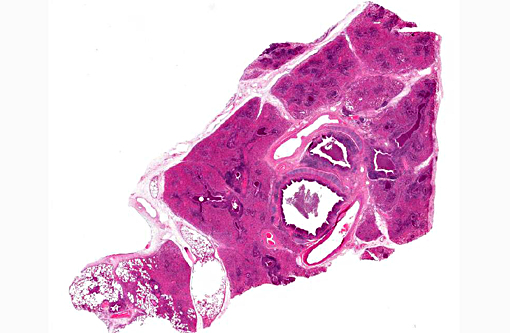
Affected lung did not float in formalin. Tracheal mucosa was diffusely reddened and contained intraluminal mucopurulent exudate. Tracheobronchial and mediastinal lymph nodes were moderately enlarged and edematous.
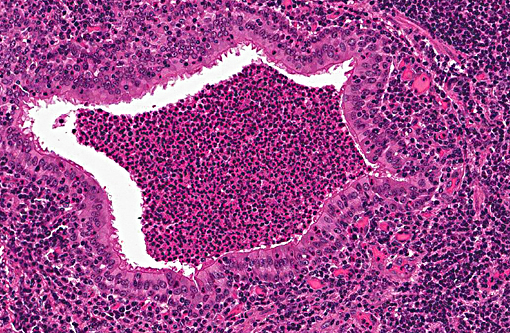
Histopathologic Description:
Morphologic Diagnosis:
Lab Results:
Condition:
Contributor Comment:
P. multocida has traditionally been classified into 5 capsular groups (A,B,D,E and F). Worldwide, serogroup A isolates are major causes of bovine respiratory disease. Serogroups A, and to a lesser extent D, cause fowl cholera in birds. Serogroups B and E are associated with hemorrhagic septicemia in cattle and water buffaloes in tropical regions of Africa and Asia. Serogroups B and E are rarely isolated from cattle in North America. In pigs, atrophic rhinitis and pneumonia are associated with toxigenic strains of serogroups D and A, respectively. Serogroup F strains are predominately isolated from diseased poultry.(4)
Pasteurellosis has been recorded in many species of deer including axis, black-tailed deer, mule deer, fallow, red, roe, sambar, reindeer, elk and white-tailed deer.(4,5,7) Disease generally presents as either hemorrhagic septicemia or pneumonia. Hemorrhagic septicemia has been responsible for outbreaks with high mortality in elk and fallow deer.(4,5) The pneumonic form, as seen in this case, is characterized by fibrinosuppurative bronchopneumonia, with or without necrosis and hemorrhage.
While Mannheimia haemolytica (formerly P. haemolytica) can cause severe, acute fibrinonecrotic pneumonia and is an important component of the bovine respiratory disease complex, P. multocida is associated with less fulminating fibrinous to fibrinopurulent bronchopneumonia with limited lung necrosis.(2)
JPC Diagnosis:
Conference Comment:
Compared to M. hemolytica, bacterial patho-genicity is reduced in P. multocida infection resulting in a slower onset of disease with absence of necrosis, vasculitis and exudation of abundant fibrin. In P. multocida, injury to cells of the respiratory system results from infiltration of inflammatory cells and release of their mediators and enzymes.(9) In contrast, M. haemolytica causes injury by release of bacterial toxins, such as leukotoxin, in addition to the effects of inflammatory mediators released from leukocytes, which results in a more necrotizing and fibrinous lesion. The leukotoxin, which is a member of the RTX group of toxins, is M. hemolyticas most important virulence factor, and can be directly toxic to cells as well as enhances the overall acute inflammatory response through activation of leukocytes and the complement system. Additionally, M. haemolytica has other virulence determinants such as polysaccharide in the capsule which can facilitate bacterial adherence and colonization, and inhibit their phagocytosis.(9) Environmental stressors and concurrent viral infection with agents such as bovine respiratory syncytial virus, bovine viral diarrhea, and infectious bovine rhinotracheitis virus result in increased susceptibility to infection by the bacterial agents.
P. multocida hemorrhagic septicemia is a reportable condition which occurs in cattle and water buffalo in Asia and Africa, and uncommonly in wild ruminants in the United States as previously mentioned by the contributor.(4,5) In this condition, the bacteria disseminate hematogenously to multiple organs resulting in congestion and hemorrhages in the respiratory, gastrointestinal and urinary tracts.(1) It is a high mortality condition with death being rapid,(6) and infection is thought to occur by inhalation or ingestion. Natural infections also occur in goats, but this species is considered more resistant to the infection.(8)
References:
1. Annas S, Zamri-Saad M, Jesse FF, Zunita Z. Comparative clinicopathological changes in buffalo and cattle following infection by Pasteurella multocida B:2. Microb Pathog. 2015;88:94-102.
2. Confer AW: Update on bacterial pathogenesis in BRD. Anim Health Res Rev. 2009;10:145-148.
3. Dabo SM, Taylor JD, Confer AW: Pasteurella multocida and bovine respiratory disease. Anim Health Res Rev. 2008;8:129-150.
4. Eriksen L, Aalbaek B, Leifsson PS, Basse A, Christiansen T, Eriksen E, Rimler RB: Hemorrhagic septicemia in fallow deer (Dama dama) caused by Pasteurella multocida multocida. J Zoo Wild Med. 1999;30:285-292.
5. Franson JC, Smith BL: Septicemic pasteurellosis in elk (Cervus elaphus) on the United States National Elk Refuge, Wyoming. J Wild Dis. 1988; 24:715-717.
6. Lopez A. Respiratory system, mediastinum and pleurae. In: McGavin MD, Zachary JF, eds. Pathologic Basis of Veterinary Disease. 5th ed. St. Louis, MO: Mosby Elsevier; 2012:500-520.
7. Mackintosh C, Haigh JC, Griffin F: Bacterial diseases of farmed deer and bison. Rev Sci Tech Off Int Epiz. 2002;21:249-263.
8. Shafarin MS, Zamri-Saad M, Khairani BS, Saharee AA. Pathological Changes in the Respiratory Tract of Goats infected by Pasteurella multocida B:2. J Comp Path. 2009;140:194-197.
9. Zachary JF. Mechanisms of microbial infections. In: McGavin MD, Zachary JF, eds. Pathologic Basis of Veterinary Disease. 5th ed. St. Louis, MO: Mosby Elsevier; 2012:170-180