Signalment:
Gross Description:
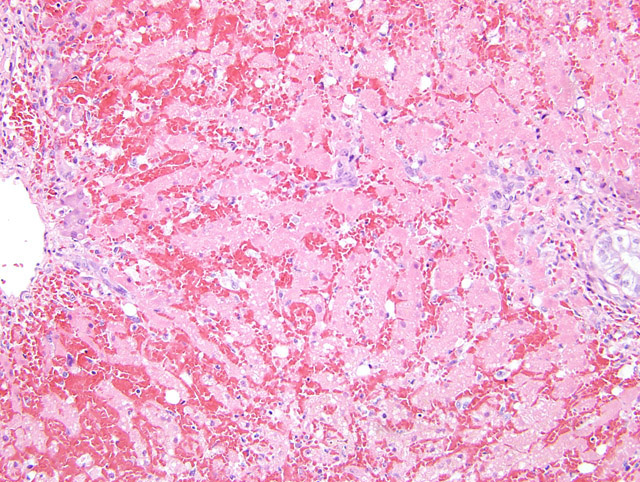
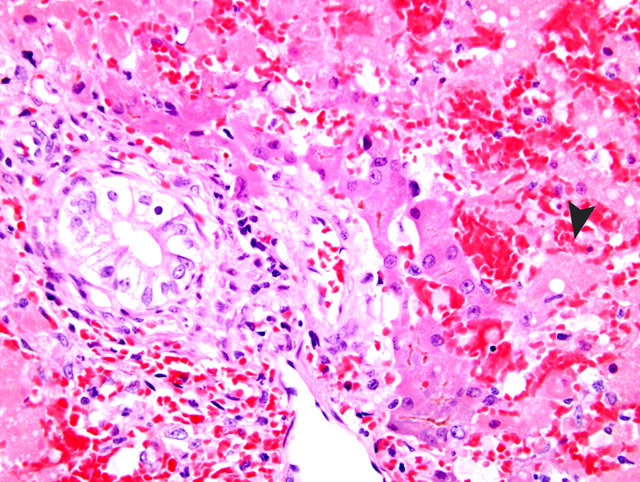
Histopathologic Description:
Morphologic Diagnosis:
Condition:
Contributor Comment:
JPC Diagnosis:
Conference Comment:
Hepatotoxic agents can be divided into two broad categories based on their predicted activity. Predictable hepatotoxins are those that produce a generally consistent activity in the majority of the animals that are exposed. The extent of injury produced in an individual animal by a predictable hepatotoxin may differ depending on various factors including age, sex, diet, and endocrine function. Idiosyncratic drug reactions are caused by those agents that produce an effect in a small minority of the animals exposed, such as carprofen occasionally causing acute hepatic necrosis in Labrador retrievers and diazepam causing acute fatal hepatic injury in some, but not all, cats.2
Hepatotoxic agents can be classified into six different categories based on their cellular target.2 1. Production of toxic metabolites by the cytochrome p450 system is the most common form of hepatocellular injury. The enzymes of this system are located in the smooth endoplasmic reticulum and are found in the highest concentration of centrilobular hepatocytes. They function to metabolize lipid-soluble chemicals into water-soluble compounds for excretion.Â
2. Drugs and cellular enzymes may combine together to form neoantigens. When transported to the cell surface and presented as antigens, these neoantigens may stimulate both cellular and humoral immune responses resulting in either direct cellular cytotoxicity or antibody-dependent cellular cytotoxicity. (halothane)
3. Some toxins may directly initiate apoptosis by stimulating proapoptotic pathways within hepatocytes. (hydrophobic bile acids)
4. Certain toxins may directly damage cellular membranes disabling calcium homeostasis and resulting in cell death. (carbon tetrachloride)
5. There are chemicals that will bind and disrupt the canalicular pumps that normally secrete bile into the canaliculi. This disruption results in cholestasis. (estrogen, erythromycin)
6. Direct damage to mitochondria decreases production of adenosine triphosphate as well as resulting in the release of cytochrome-c leading to apoptosis or necrosis. (antiviral nucleosides, intravenous tetracycline)
Certain toxic compounds may affect cells other than hepatocytes.2 Damage to biliary epithelium may be caused by trimethoprim-sulfa or sporidesmin, while damage to Kupffer cells can be caused by endotoxin. Arsenicals damage endothelial cells of the liver, and vitamin A excess causes activation of hepatic stellate cells.Â
Selected hepatotoxins extracted from Cullen2
| Category | Members | Mechanism of action | Remarks |
| Blue-green algae | Anabaena, Aphanizomenon, Microcystis | Microcystin LR (cyclic heptapeptide) | More closely related to bacteria |
| Pyrrolizidine alkaloids | Senecio, Cynoglossum, Crotalaria, Heliotropium | Ingested alkaloids converted to pyrrolic esters by cytochrome p450 enzymes | Esters are alkylating agents that act on cytosolic and nuclear proteins. Megalocytes due to antimitotic effect. |
| Aflatoxin | Aspergillus flavus | Aflatoxin B1 (toxic intermediates produced by cytochrome p450 enzymes) | Toxin and carcinogen. Sheep more resistant. |
| Sporidesmin | Pithomyces chartarum (fungus growing on dead rye grass) | Necrosis of the epithelium of large intrahepatic and extrahepatic biliary ducts | Results in cholestasis with failure to excrete phylloerythrin leading to photosensitization |
| Mushroom | Amanita sp. |
|
|
Equine serum hepatitis is an idiopathic condition most closely associated with administration of equine-origin biologics.1,3 It is generally reported 41-60 days following administration of a biologic product, and is characterized by acute hepatic centrilobular necrosis.1
References:
2. Cullen JM: Liver, biliary system, and exocrine pancreas. In: Pathologic Basis of Veterinary Disease, eds. McGavin MD, Zachary JF, 4th ed., pp. 440-447. Elsevier, St. Louis, MO, 2007
Divers TJ: Liver disease and liver failure in horses, in Proceedings. Am Assoc Equine Pract
3. Guglick MA, MacAllister CG, Ely RW, Edwards W: Hepatic disease associated with administration of tetanus antitoxin in eight horses. J Am Vet Med Assoc 11:1737-1740, 1995
4. Stalker MJ, Hayes MA: Liver and biliary system. In: Jubb, Kennedy, and Palmers Pathology of Domestic Animals, ed. Maxie MG, 5th ed., vol. 2, pp. 322-333. Elsevier Limited, St. Louis, MO, 2007
5. Toshach K, Jackson MW, Dubielzig RR: Hepatocellular necrosis associated with the subcutaneous injection of an intranasal Bordetella bronchiseptica-canine parainfluenza vaccine. J Am Anim Hosp Assoc 33:126-128, 1997