Signalment:
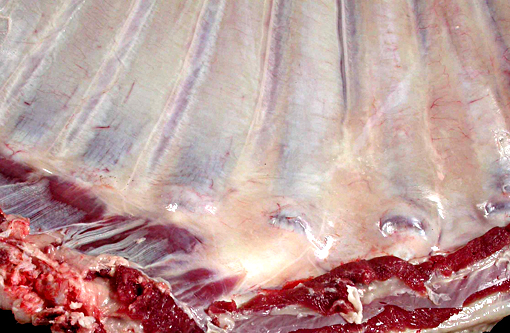
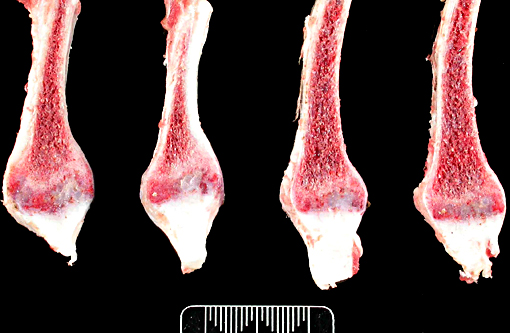
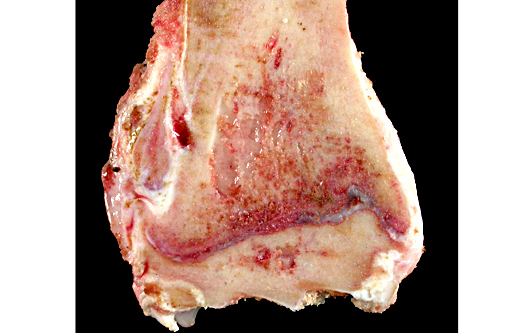
Gross Description:
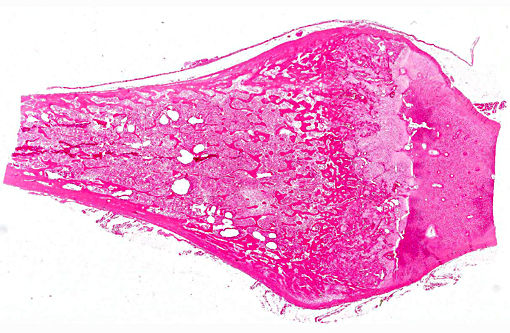
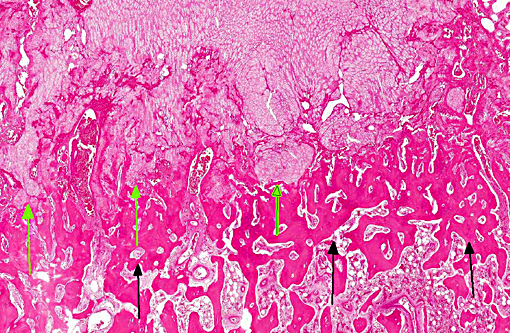
Histopathologic Description:
Morphologic Diagnosis:
Lab Results:
Calcium 1.97 mmol/L (2.0-2.7)
Phosphate: 0.73 mmol/L (1.3-2.7)
25-hydroxyvitamin D3: 36 nmol/L (no difference compared with control sheep)
1,25-dihydroxyvitamin D3: 154 pmol/L (no difference compared with control sheep)
Condition:
Contributor Comment:
In Corriedale sheep with autosomal recessive inherited rickets,(3,5) a novel nonsense mutation in the noncollagenous bone protein, dentin matrix protein 1 (DMP1), leading to a premature stop codon and truncation of the protein has been found (personal communication, K. Dittmer). The equivalent form of this disease in humans is called autosomal recessive hypophosphataemic rickets I. The mutation in DMP1 leads to increased serum fibroblast growth factor 23 (FGF23) concentration. FGF23 inhibits the renal NPT-2a cotransporter (a Na-P cotransporter) and CYP27B1 (1α-hydroxylase) activity in the kidney, altering phosphate reabsorption in the renal tubules, and inhibiting 1,25(OH)2D3 production. Consequently, humans with this condition have hypophosphataemia, phosphaturia, inappropriately normal serum 1,25(OH)2D3 concentrations and rickets.(6)
The thickened cortices seen in the long bones of affected sheep(2) and enthesiophytes are a feature of X-linked hypophosphataemic rickets in humans.(9) The pathogenesis of this is unclear. However in the sheep, we hypothesize that the enthesiophytes are the result of strain on ligament/tendon attachments to weakened, poorly mineralised bone. Likewise, the thickened cortices are a consequence of bone being deposited at sites of strain in order to decrease deformation associated with mechanical loading, as described by Wolffs law or the mechanostat model.(7,8)
This inherited disease has the potential to be widespread in Corriedale sheep worldwide. A test for heterozygous animals has been developed.
JPC Diagnosis:
Conference Comment:
The lesions of rickets result from failure of mineralization, which includes both impaired endochondral ossification and failed mineralization of osteoid. This results in the presence of excess osteoid as well as the prominent nodular thickenings of cartilage which are apparent grossly. The enlarged irregular physis is composed of increased numbers of disorganized chondrocytes, as seen in this case. The metaphyses are flared secondary to impaired osteoclast removal of cartilage and unmineralized bone (osteoid) from the cutback zones. Osteoclasts cannot bind unmineralized matrix, impairing remodeling and resulting in accumulation of unmineralized osteoid and cartilage.(1) Naturally occurring rickets is uncommon in sheep, cattle, horses, dogs, and cats; llamas and alpacas are highly susceptible and pigs are susceptible when they dont receive adequate feed supplementation. There is also a genetic form in pigs which is similar to a condition that occurs in people.(4) Like rickets, the pathogenesis of osteomalacia also involves defective mineralization, but is a condition that occurs in adult animals after the growth plates have closed and therefore does not involve growth plates. Osteo-malacia results in increased amounts of unmineralized osteoid at sites of pressure or stress.
Vitamin D3 is formed in the skin and can also be absorbed in the diet, as can vitamin D2. It is stored in fat or transported to the liver where it must undergo the first step of activation, hydroxylation. Once hydroxyl-lated in the liver, 25-hydroxycholecalciferol [25(OH)D3], which is the primary form of vitamin D in circulation, must again be hydroxylated in the kidney by 1α-hydroxylase, to form1, 25-dihydrox-ycholecalciferol [1,25(OH)2D3], the metabolically active form of vitamin D. Formation of this metabolically active form in the kidney is regulated by serum phosphorus and calcium concentrations and parathyroid hormone. The metabolically active form of vitamin D acts to mobilize calcium from bone in order to maintain serum calcium concentration, by maturation and activation of osteoclasts. The precise mechanism by which vitamin D influences bone growth is not completely understood, but may be indirectly through calcium and phosphorus concentration, or through direct interaction with osteoblasts and chondrocytes.(4)
References:
1. Carlson CS, Weisbrode SE. Bones, joints, tendons and ligaments. In: McGavin MD, Zachary JF, eds. Pathologic Basis of Veterinary Disease. 5th ed. St. Louis, MO: Mosby Elsevier; 2012:946-949.
2. Dittmer KE, Firth EC, Thompson KG, Marshall JC, Blair HT. Changes in bone structure of Corriedale sheep with inherited rickets: A peripheral quantitative computed tomography assessment. The Veterinary Journal. 2011;187: 369-373.
3. Dittmer KE, Howe L, Thompson KG, Stowell KM, Blair HT, Cockrem JF. Normal vitamin D receptor functions with increased expression of 25-hydroxyvitamin D3-24-hydroxylase in Corriedale sheep with inherited rickets. Res Vet Sci. 2011;91(3):362-369.
4. Dittmer KE, Thompson KG. Vitamin D Metabolism and Rickets in Domestic Animals: A Review. Vet Pathol. 2011;48:389-407.
5. Dittmer KE, Thompson KG, Blair HT. Pathology of inherited rickets in Corriedale sheep. J Comp Path. 2009;141:147-155.
6. Lorenz-Depiereux B, Bastepe M, Benet-Pages A, Amyere M, Wagenstaller J, Muller-Barth U, Badenhoop K, Kaiser SM, Rittmaster RS, Shlossberg AH, Olivares JL, Loris C, Ramos FJ, Glorieux F, Vikkula M, Juppner H, Strom TM. DMP1 mutations in autosomal recessive hypophosphatemia implicate a bone matrix protein in the regulation of phosphate homeostasis. Nature Genetics. 2006;38:1248-1250.
7. Pearson OM, Lieberman DE. The aging of Wolff's "law": Ontogeny and responses to mechanical loading in cortical bone. Yearbook of Physical Anthropology. 2004;47: 63-99.
8. Rauch F. Material matters. A mechanostat-based perspective on bone development in osteogenesis imperfecta and hypophosphatemic rickets. Journal of Musculoskeletal and Neuronal Interactions. 2006;6:142-146.
9. Reid IR, Hardy DC, Murphy WA, Teitelbaum SL, Bergfeld MA, Whyte MP. X-linked hypophosphatemia: A clinical, biochemical, and histopathologic assessment of morbidity in adults. Medicine. 1989;68:336-352.
10. Thompson KG. Bones and joints. In: Maxie MG, ed. Jubb, Kennedy, and Palmer's Pathology of Domestic Animals. 5th ed. Vol1. Philadelphia, PA: Elsevier Saunders; 2007:1-184.