Joint Pathology Center
Veterinary Pathology Services
Wednesday Slide Conference
2018-2019
Conference 9
1 November 2018
CASE III: H17-1982 (JPC 4105936-00).
Signalment: 16 day-old Holstein-Friesian male (Bos taurus).
History: The calf became acutely dull and lethargic, with subnormal body temperature, despite being placed under a heating lamp. The animal did not feed and died overnight.
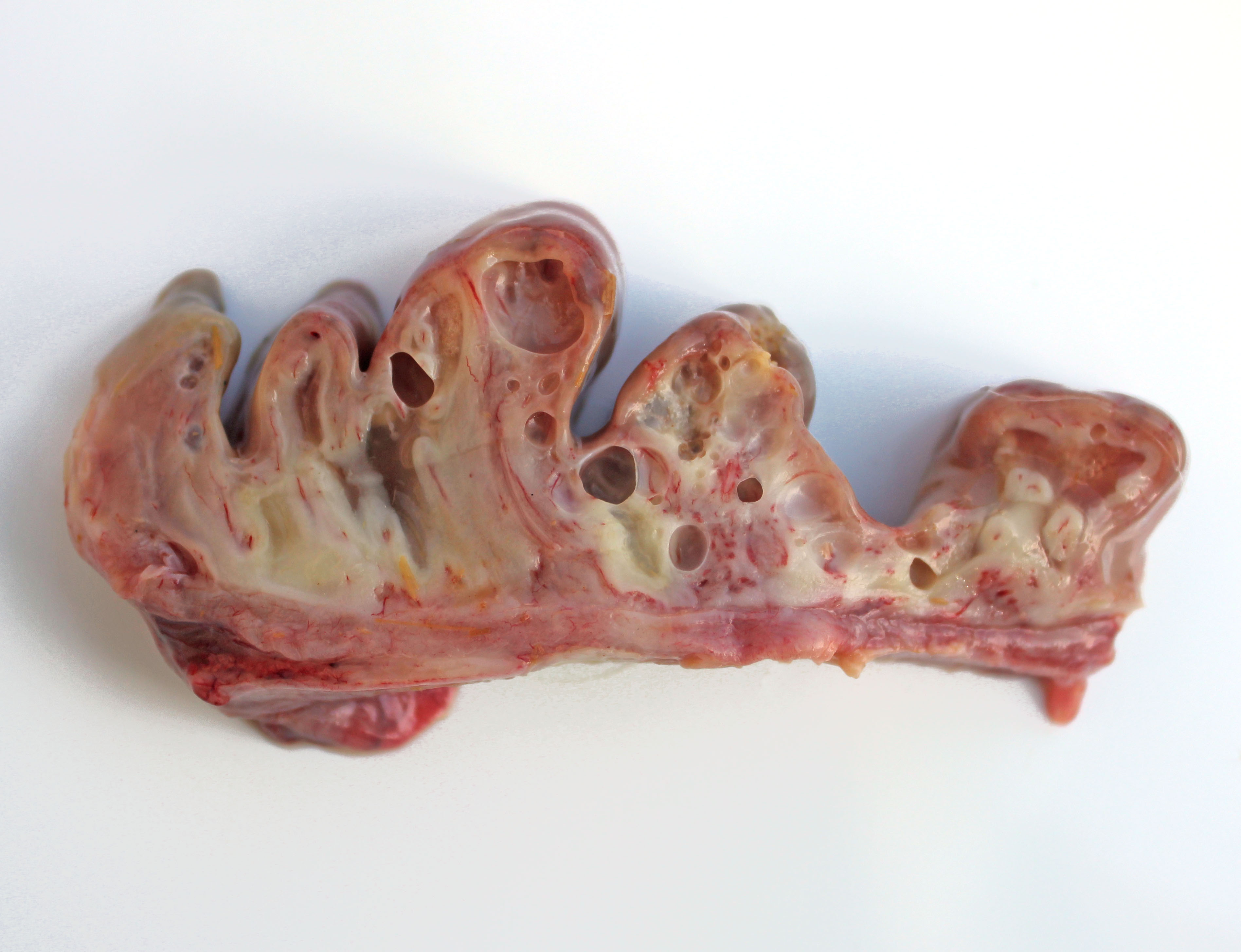
Gross Pathology: The calf was in reasonable body condition. Bilaterally, sunken eyeballs within sockets. Abundant serofibrinous fluid in abdominal cavity with fibrin adherent to a large (4 cm diameter) non-penetrating serosal tear along the greater curvature of the abomasum. Within the abomasum, the mucosa was diffusely congested, with two large (approximately 5 cm diameter) areas of ulceration. Diffusely, there was marked thickening, edema and emphysema of the abomasal folds.
Laboratory results:
Abomasal tissue Clostridium sordellii FAT – negative
Abomasal tissue Clostridium septicum FAT – negative
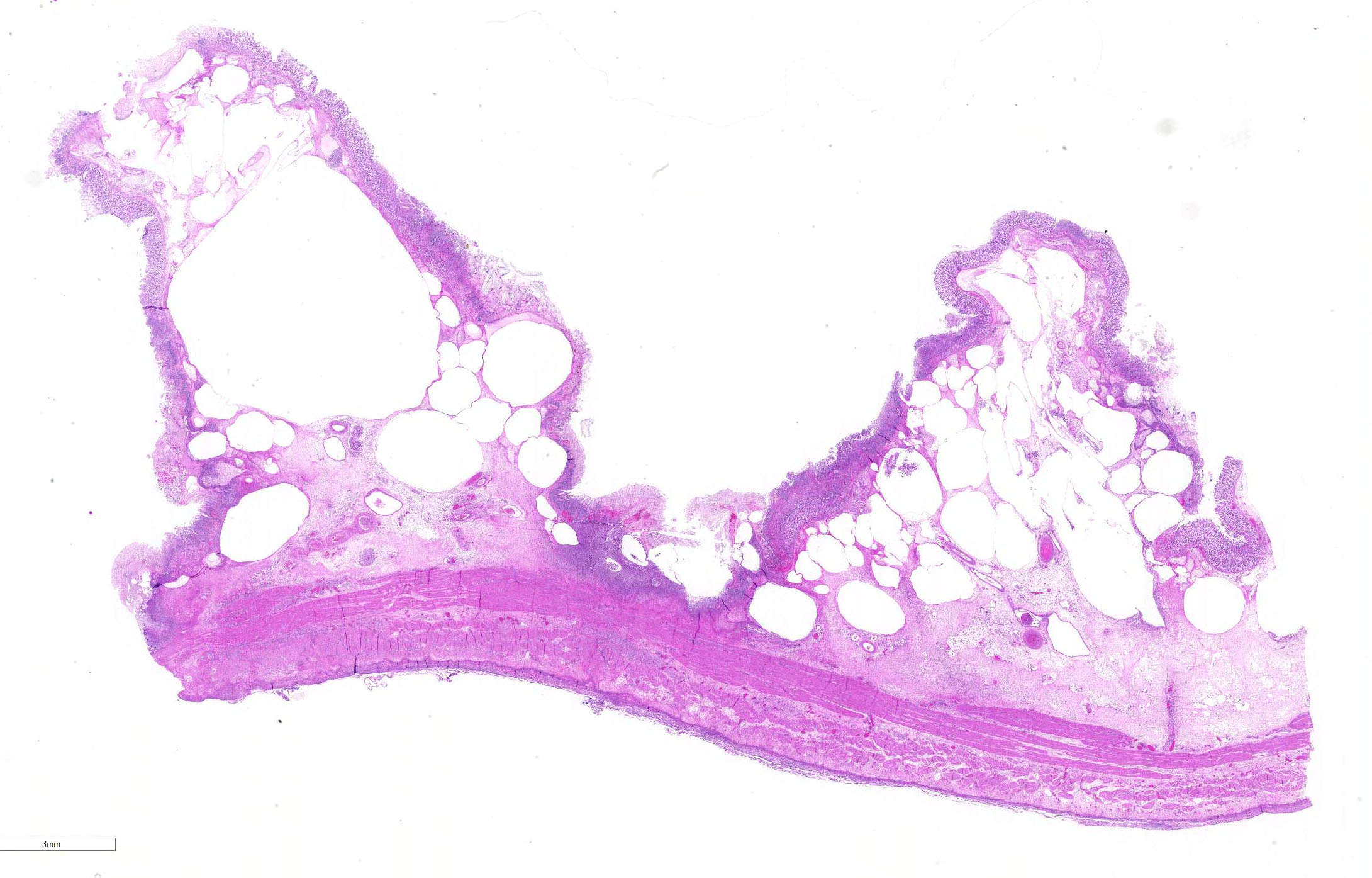
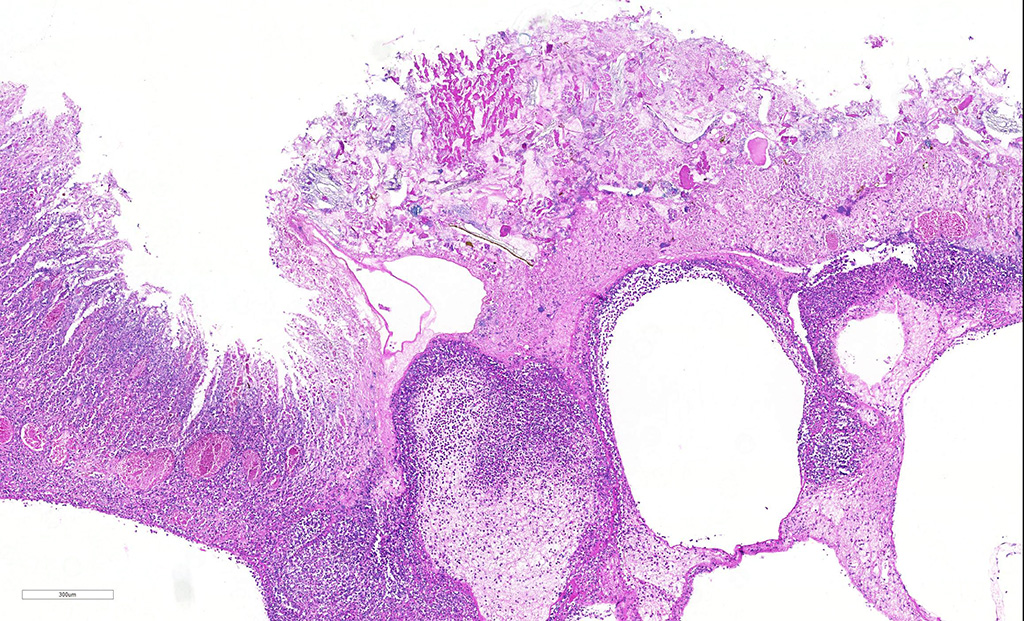
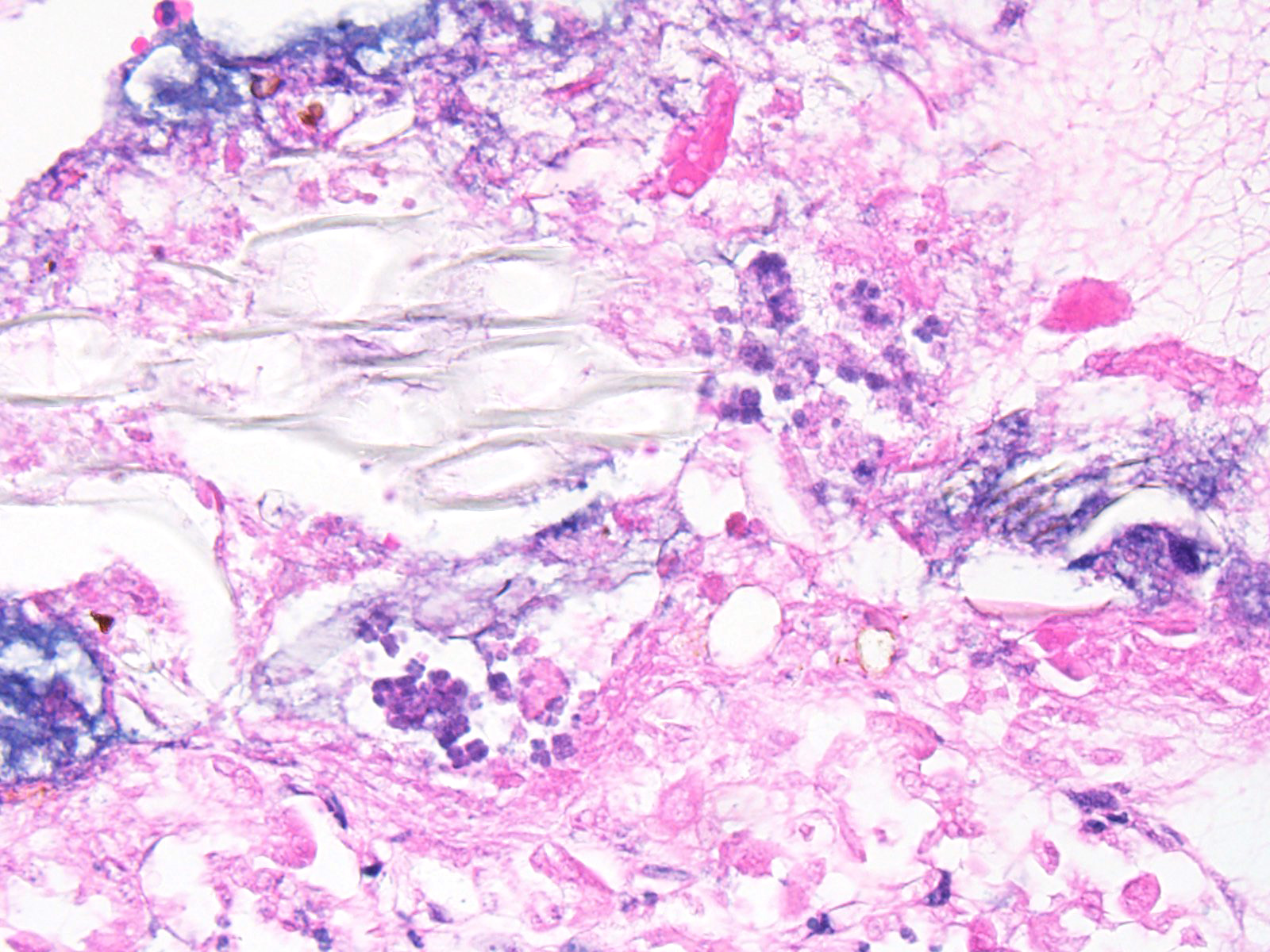
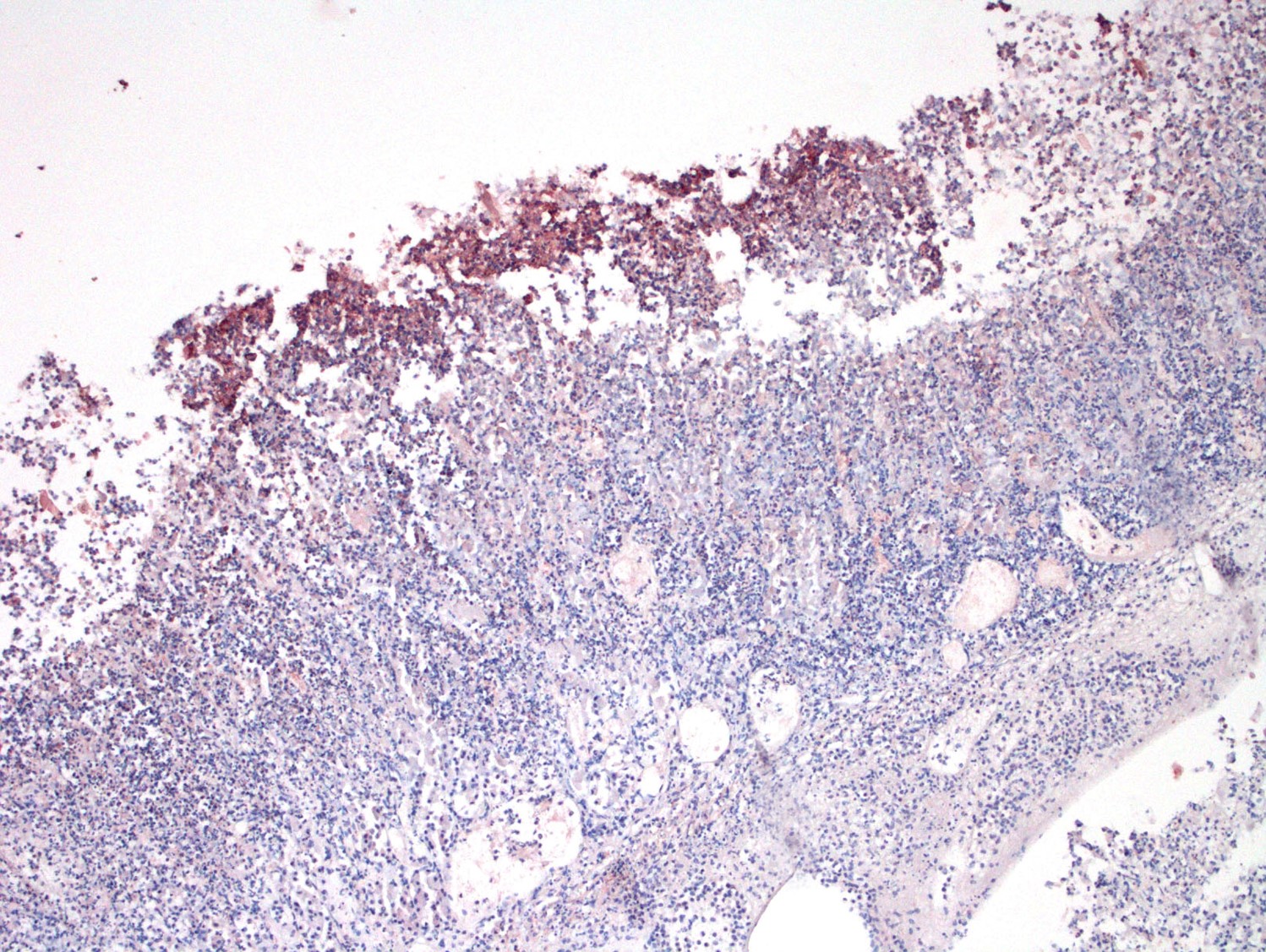
Microscopic Description: Abomasum: There are marked multifocal areas of degeneration, necrosis and loss of the glandular mucosa and gastric pits. Necrotic areas contain amorphous eosinophilic cellular debris, basophilic karyorrhectic debris and are infiltrated by large numbers of viable and degenerate neutrophils and some macrophages. Occasionally both within and overlying necrotic areas there are tetrad packets of basophilic cuboidal 2-3 um bacteria (Sarcina spp.). Diffusely, the abomasal leaves are thickened with severe submucosal expansion by large (up to 6mm) multifocal-coalescing clear spaces (emphysema), abundant finely fibrillar eosinophilic mesh (fibrinous exudate) and dilated lymphatics. There is marked submucosal infiltration of numerous neutrophils. The neutrophilic infiltrate and fibrinous oedema obscures the muscularis mucosa and extends through the submucosa, muscularis externa and serosa. The external serosa is covered by a thick mat of fibrin and neutrophils.
Contributor’s Morphologic Diagnoses:
Abomasum: Severe, acute, diffuse, necrosuppurative and emphysematous abomasitis with Sarcina spp.
Etiology: Sarcina spp. (putative)
Contributor’s Comment: The association of Sarcina spp. with fatal emphysematous abomasitis, abomasal bloat and abomasal ulceration in both young calves and lambs has been reported.2,3 The current case demonstrates a number of typical features; submucosal emphysematous bullae, fibrinous exudate, neutrophilic infiltrates, foci of mucosal necrosis and the characteristic colonies of Sarcina spp. bacteria2,5. Sarcina spp. are gram-positive anaerobic cocci, are carbohydrate fermenters and can grow in an acid environment (as low as pH 1.0). They have a characteristic microscopic morphology: basophilic, cuboidal bacteria with flattened walls occurring in tetrad packets. They are not invasive and are typically found along the mucosal surface.1 The main products of Sarcina carbohydrate fermentation are ethanol, acetaldehyde, carbon dioxide and hydrogen. In human gastritis associated with Sarcina ventriculi, it is postulated that the mucosal damage is the result of alcohol production, as the lesions are reminiscent of acute alcohol poisoning1. In animals it is postulated that production of large volumes of carbon dioxide induce abomasal bloat and emphysema2 with the distension in turn inducing ulceration, edema and haemorrhage5. Sarcina spp. are present in the soil and environment. When ingested by pre-ruminant calves the milk diet provides a rich fermentative substrate, and the low pH environment favoured by the bacteria, enabling them to outgrow the commensal abomasal flora and cause gas accumulation. Although Sarcina spp. are difficult to culture under routine conditions, their histological features are quite characteristic/pathognomonic.
Contributing Institution:
UCD School of Veterinary Medicine, University College Dublin, Belfield, Dublin 4, Ireland
http://www.ucd.ie/vetmed/
JPC Diagnosis: Abomasum: Abomasitis, necrotizing and neutrophilic, transmural, diffuse, severe, with marked submucosal emphysema and edema, and numerous bacterial tetrads and colonies of bacilli.
JPC Comment: Sarcina ventriculi is an interesting presumptive pathogen of young ruminants, and occasionally in humans. Since its original association with abomasal bloat in goat kids and lambs in the 1990s, it has been incriminated as a cause of various syndromes of abomasal bloat, tympany, and necrotizing abomasitis. It is a relatively fastidious anaerobe and notoriously difficult to culture, rendering inoculation studies difficult at best, and the additional observation that it may be found in the stomachs of healthy as well as diseased animals has led to great consternation for pathologist trying to add it to the established pathogens Clostridium septicum and Clostridum perfringens type A in the development of braxy or braxy-like syndromes in young ruminants. The concurrent identification of various clostridial species, notably C. sordelli, in affected animals has further clouded the diagnostic picture5; hence, the role of this microorganism in abomasitis of calves has, however, not been definitely demonstrated and it is possible that Sarcina is part of a multifactorial etiology.3,4
To complicate the etiology of the lesion in this individual even more, several rods identified in the lumen of this abomasum were immunopositive for C. perfringens IHC at the San Bernardino laboratory of the California Animal Health and Food Safety System.
In humans, S. ventriculi was first identified as a gastric pathogen by Dr. John Goodsir in 1842, while only 19 cases of gastritis due to S. ventriculi infection have been confirmed since, with more than half of these cases occurring in patients with a history of previous gastric surgery resulting in outflow impairment. The term “sarcinous vomiting” referring to a characteristic “obstinate, frothy” vomit has been described in these patients as well as others with chronic gastritis.1
Sarcina is a genus of three species of gram-positive coccobacteria in the family Clostridiaceae which are known commensals of human skin and GI tract, and synthesize microbial cellulose. It derives its name from “sarcina” from the Latin meaning a pack or bundle (similar to that worn by Roman legionaries). The bacteria grow in two planes, resulting in characteristic tetrads, with cell wall compression against adjacent bacilli giving them a cuboidal appearance. (A similar tetrad appearance may also be seen with the much smaller Micrococcus.) Interestingly, they are purportedly refractile (not demonstrated in this slide), mimicking plant material. 1
Sarcina sp. are naturally present in the soil and is likely ingested with feedstuffs. Sarcina sp.
possess an exclusive carbohydrate fermentative metabolism, producing ethanol, acetaldehyde, carbon dioxide and hydrogen. Theories of pathogenesis include the production of large amounts of carbon dioxide resulting in bloat and emphysema, with production of ethanol and acetaldehyde resulting in mucosal necrosis. Abomasal tympany may additionally result in secondary damage to the overstretched mucosa.2
References:
- Al Rasheed M, Senseng CG. Sarcina ventriculi: Review of the Literature. Arch Pathol Lab Med; 2016 140: 1441-1445.
- Edwards GT, Woodger NGA, et al. Sarcina-like bacteria associated with bloat in young lambs and calves. Vet Rec; 2008: 163: 391-393, 2008.
- Panciera RJ, Boileau MJ, Step DL. Tympany, acidosis, and mural emphysema of the stomach in calves: report of cases and experimental induction. J Vet Diagn Invest; 2007 19(4):392-5.
- John F. Prescott, Paula I. Menzies and Russell S. Fraser. Clostridial abomasitis. In: Uzal FA, Songer WG, Prescott J, Popoff M eds. Clostridial Diseases of animals. Wiley Blackwell, Ames, IA, 2016.
- Vatn S, Tranulis MA, Hofshagen M. Sarcina-like bacteria, Clostridium fallax and Clostridium sordellii in lambs with abomasal bloat, haemorrhage and ulcers. J Comp Path 1999; 122: 193-200.