WSC 2022-2023
Conference 19
Case IV:
Signalment:
1 year-old, intact female, Cavalier King Charles Spaniel, dog (Canis familiaris)
History:
This dog was a normal active puppy up until 4 weeks prior when the owner noted the puppy acting lethargic and had labored breathing. The puppy was brought to the vet and was diagnosed with a respiratory infection. Fungal cultures were negative. The puppy was started on an antibiotic, prednisone, and another medication (unknown). Clinical signs improved but then worsened and the puppy quickly died. All other puppies in the litter and dogs in the home were all normal with no clinical signs.
Gross Pathology:
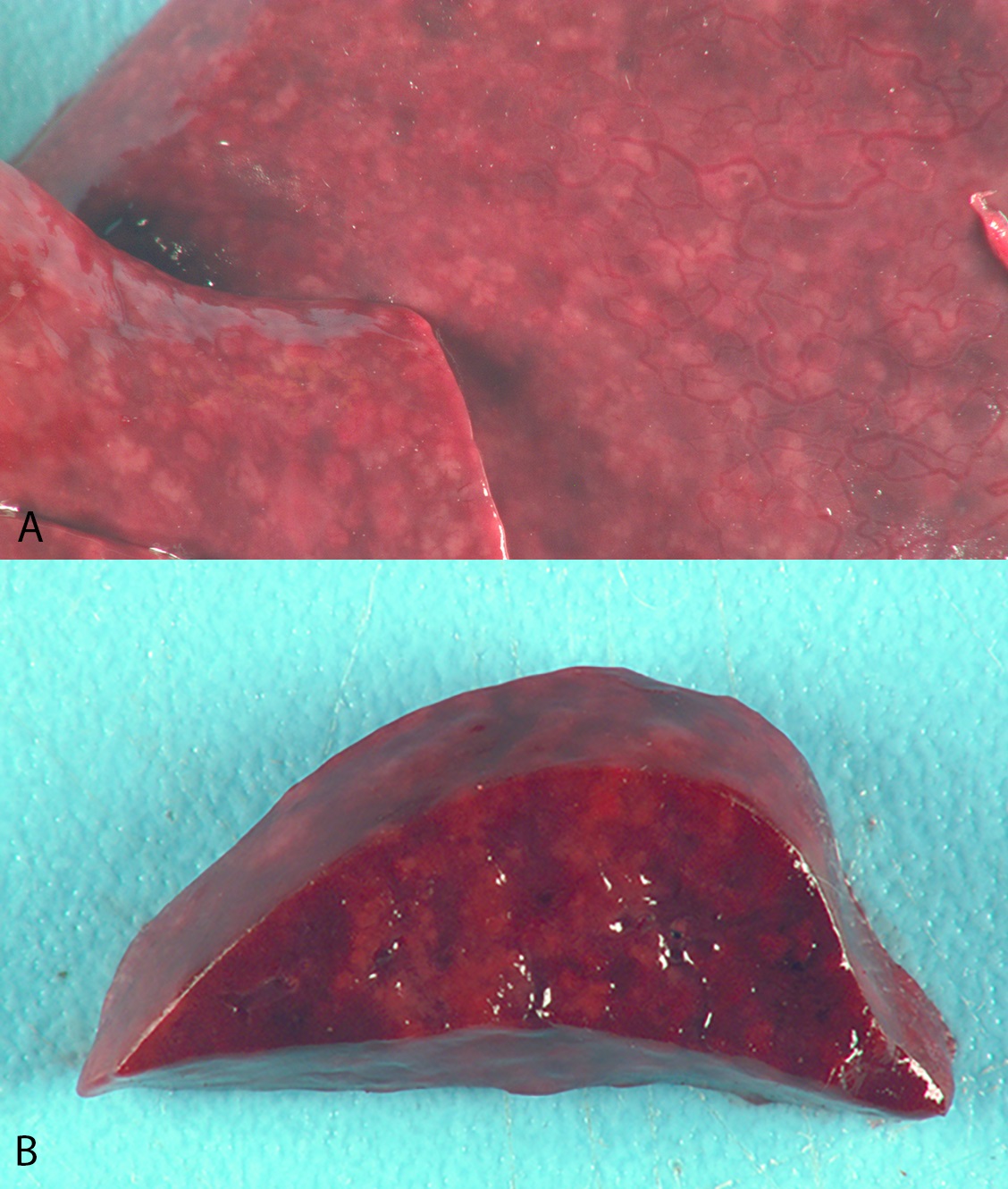
The pulmonary parenchyma was diffusely dark red, rubbery, and did not collapse. There were hundreds of randomly scattered, 1-2 mm in diameter, tan to white, firm raised foci throughout all lung lobes.
Laboratory Results:
No laboratory results reported.
Microscopic Description:
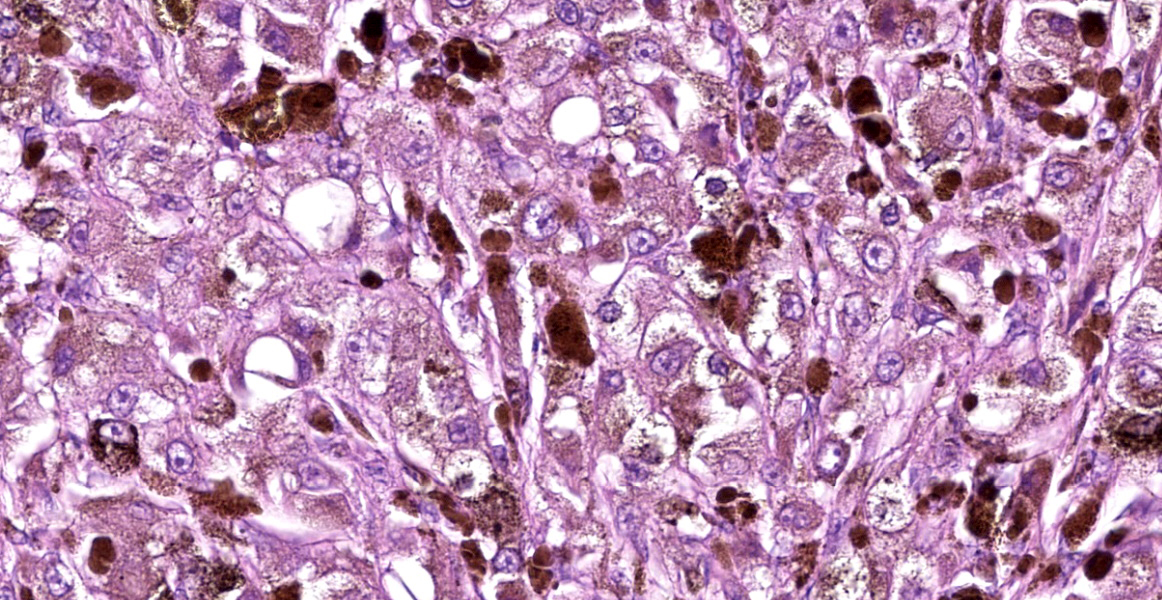
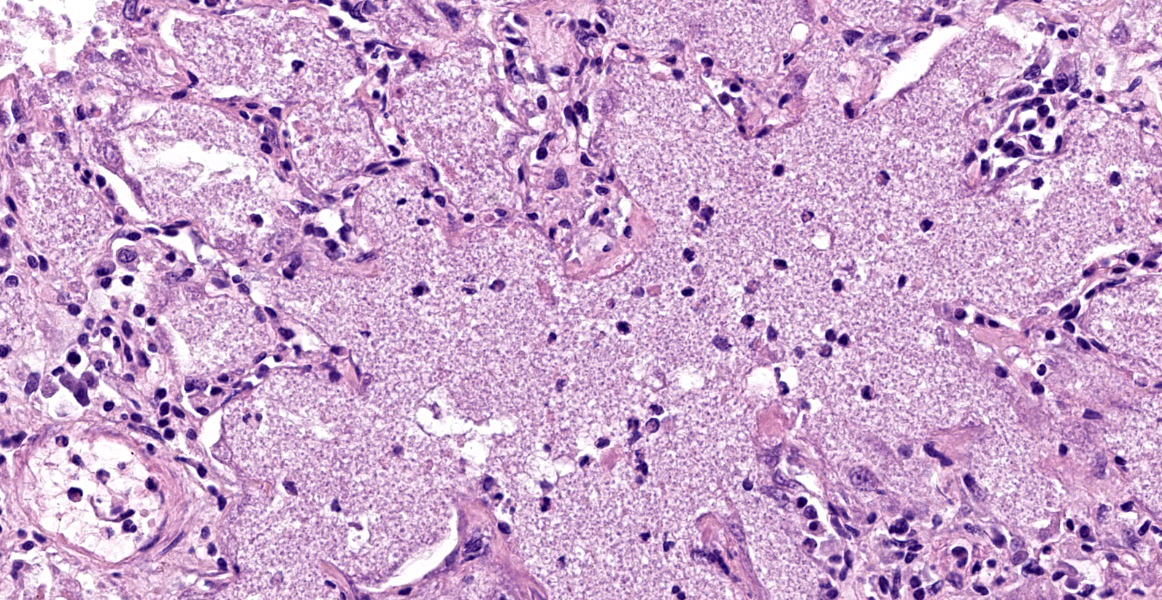
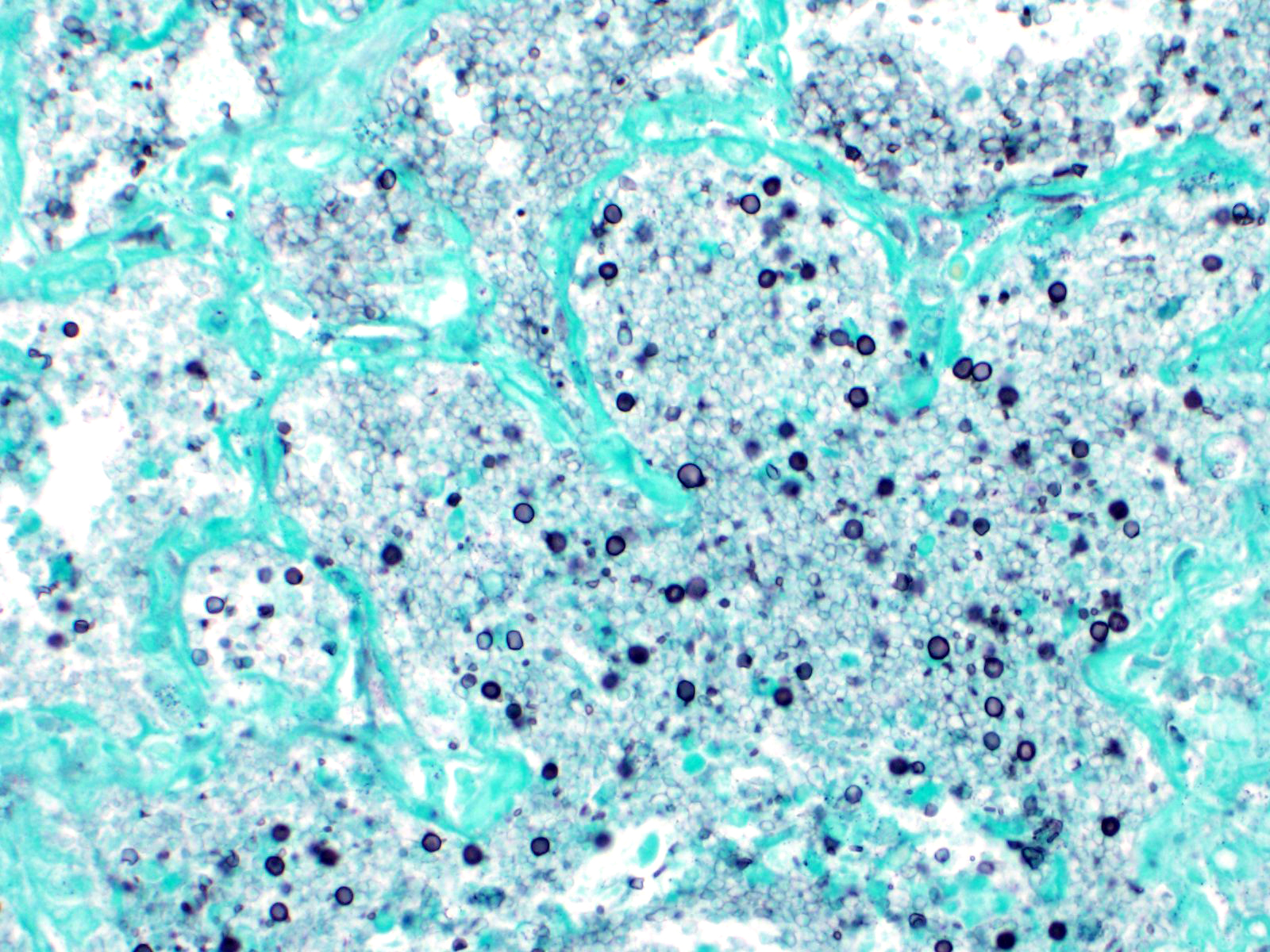
Approximately 90% of alveolar spaces are multifocally expanded by eosinophilic, foamy, amorphous material (Figure 2) with numerous approximately 7µm in diameter, teardrop-shaped, yeast structures that have variably thick walls highlighted by a Grocott’s Methenamine Silver (GMS) stain (Figure 3). Moderate numbers of histiocytes are associated with these regions. The adjacent alveoli are lined by plump cuboidal type II pneumocytes. The remaining pulmonary parenchyma is extensively autolytic and contains multifocal mats of cadaver bacilli.
Contributor’s Morphologic Diagnoses:
Lung: Severe, multifocal, histiocytic alveolitis with intralesional yeasts and type 2 pneumocyte hyperplasia
Contributor’s Comment:
Pneumocystis sp., is a commensal yeast-like fungal organism, in the class Pneumocystidomycetes, found in the lungs of adult animals. Initial colonization with Pneumocystis sp. is thought to occur during the birthing process but can also be horizontally transmitted between the same species as an airborne organism5,8. Essentially every mammal has a unique strain of Pneumocystis sp. and interspecies transmission is not thought to occur. There are five host-specific Pneumocystis sp. that have been formally described in the literature and include: P. carinii/wakefeldeii in rats, P. murina in mice, P. oryctolagi in rabbits, and P. jirovecii in humans1. Pneumocystis pneumonia (PCP) can occur in any species of animal, especially if immunocompromised. In humans, PCP and secondary airway obstruction are serious consequences in immunocompromised individuals, especially in those individuals that are infected with human immunodeficiency virus (HIV). Pneumocystis sp. are fastidious organisms and cannot be grown in continuous culture, leading researchers to heavily rely on animal models of disease2,5,8. P. carnii infections in rats were the original study models; however, due to host-specific genotypic and phenotypic variations, it was difficult to make comparisons to P. jiroveccii infections in humans2. Additionally, due to the inability to recreate an HIV model in rats, rat models to study PCP infections were not ideal. Studies utilizing macaques infected with simian immunodeficiency virus (SIV) then became the most relevant animal model not only due to macaques developing clinical signs similar to those in humans with AIDS, but, P. carinii derived from humans and non-human primates are evolutionarily more related than P. carinii derived from rats2.
Pneumocystis pneumonia, suspected to be caused by Pneumocystis carinii f sp. canis6 is uncommon in dogs. Young to middle-aged Cavalier King Charles Spaniels and Miniature Dachshunds are overrepresented and may have inheritable immunodeficiency predisposing them to over colonization3,9. In a study that evaluated IgG, IgM, and IgA serum concentration in Cavalier King Charles Spaniels diagnosed with Pneumocystis pneumonia compared to breed and age-matched nonaffected dogs, it was found that affected dogs had significantly lower IgG concentrations9. These dogs tend to respond to medical treatment if treated early in the course of the disease.
Pneumocystis infects hosts by direct attachment to the alveolar epithelium via surface antigen glycoprotein A (gpA). The life cycle is composed of two morphologically distinguished forms, a trophic form and a cystic form which undergo binary fission and sexual replication, respectively. The trophic form is the predominant form during infection and is characterized by 1-4µm, thin-walled, uninucleate organisms8. The cystic form is 5-8 µm in diameter, thick-walled, and contains 8 intracystic nuclei/bodies. The intracystic bodies are released, attach to type I pneumocytes, and are thought to develop into the trophic form4.
Pneumocystis can evade host immunity due to its cell wall composed of mannose-rich polysaccharides and heavily glycosylated surface antigens4. gpA is the key surface antigen responsible for attachment to the host epithelium as well as evasion of host immunity via interactions with macrophages, surfactant proteins, and fibronectin4. Multiple genes encode this protein; however, during infection only one isoform is expressed3. The expression of this isoform is unique to the associated organism adapted to each host species. Pneumocystis causes disease by expanding alveoli as well as possibly altering surfactant homeostasis. Infection is controlled by macrophages and cell-mediated immunity4.
Gross lesions can be multifocal to diffuse and are often characterized by red to brown areas of consolidation that may be variably nodular4. Histologically, lesions are characterized by foamy eosinophilic material and variable numbers of foamy macrophages that fills alveolar spaces. Organisms are difficult to identify on routine hematoxylin and eosin staining. Special staining with methenamine sliver highlights the walls of the organism. Secondary, variably prominent findings include, increased interstitial lymphocytes, plasma cells, and type II pneumocyte hyperplasia4. The number of organisms within alveolar spaces can greatly vary, thus PCR is the most sensitive detection method.
Contributing Institution:
Michigan State University Veterinary Diagnostic Laboratory. Department of Pathobiology and Diagnostic Investigation - https://cvm.msu.edu/vdl
JPC Diagnosis:
Lung: Pneumonia, interstitial, histiocytic and necrotizing, diffuse, marked, with type II pneumocyte hyperplasia and many intra-alveolar yeasts.
JPC Comment:
The contributor provides a great overview of this ubiquitous fungus which can infect a wide variety of hosts. A recent meta-analysis evaluated the clinical and diagnostic features of 43 dogs with pneumocystosis.10 Miniature dachshunds and cavalier King Charles spaniels were predisposed.10 Clinical signs were nonspecific and included weight loss, cough, dyspnea, and cyanosis, but, interestingly, most dogs had normal body temperature. 10 Hemogram changes include leukocytosis and neutrophilia.10 The authors found that hypogammaglobulinemia was useful in the diagnosis and reflects the inability of infected dogs to class-switch from IgM to IgG production.10 On necropsy, the lungs were firm or rubbery and discolored or pale. 10 Pneumothorax was uncommonly seen in these dogs but is a common finding in human infections.10 Cytologic examination of bronchoalevolar lavage fluid, the mainstay of diagnosis in humans, was insensitive and unreliable in this report; more sensitive antemortem diagnostics included fine needle aspirates of the lung and cytologic examination of tracheal swabs.10 Histologic findings included lymphohistiocytic interstitial pneumonia with type II pneumocyte hyperplasia and foamy to granular eosinophilic material filling alveolar lumens containing poorly-staining extracellular and intracellular trophozoites and cysts.10 GMS and PAS highlighted the cyst walls and confirmed the diagnosis.10 Advanced diagnostics that were useful in the meta-analysis include PCR, electron microscopy, and immunohistiochemistry.10
An uncommon complication of pneumocystis infection is disseminated spread, which was recently reported in a young adult toy poodle.7 In addition to the alveolar lumen, the fungus was found in the pulmonary interstitium and had spread to multiple lymph nodes, the liver, heart, spleen, pancreas, kidneys, and gastrointestinal tract.7 There was marked lymphoid depletion in multiple lymph nodes and the spleen.7 Based on the distribution pattern, the authors speculated that the fungus spread through lymphatics.7 The dog may have been immunodeficient due to a prolonged course of corticosteroids or due to a underlying immunodeficiency.7
Conference participants discussed the gross photograph from this case; the pale patchy discoloration combined in an immunosuppressed animal (i.e. in a severe combined immunodeficiency in an Arab foal, or an NHP infected with SIV) is strongly suggestive of Pneumocystis pneumonia.
References:
- Alanio A, Bretagne S. Pneumocystis jirovecii detection in asymptomatic patients: what does its natural history tell us? F1000 Research. 2017; 6.
- Board KF, Patil S, Lebedeva I, et al. Experimental Pneumocystis carinii pneumonia in simian immunodeficiency virus–infected rhesus macaques. J Infect Dis. 2003;187(4): 576-588.
- Farrow, BRH, Watson, ADJ, Hartley, WJ, and Huxtable, CRR. Pneumocystis pneumonia in the dog. J Comp Path. 1972; 82(4): 447-453.
- Maxie, MG. Jubb, Kennedy & Palmer’s Pathology of Domestic Animals. Vol 2. Elsevier. 2015; 535-536.
- Morris A, Sciurba FC, Norris KA. Pneumocystis: a novel pathogen in chronic obstructive pulmonary disease? COPD. 2008;(1)5: 43-51.
- Ralph E, Reppas G, Halliday C, Krockenberge M, Malik R. Pneumocystis canis pneumonia in dogs. Microbiology Australia. 2015; 36:79-82.
- Sakashita T, Kaneko Y, Izzati U z, et al. Disseminated Pneumocystosis in a Toy Poodle. J Comp Pathol. 2020; 175: 85-89.
- Thomas C, Limper A. Current insights into the biology and pathogenesis of Pneumocystis pneumonia. Nat Rev Microbiol. 2007;5: 298–308.
- Watson PJ, Wotton P, Eastwood J, Swift ST, Jones B, Day MJ. Immunoglobulin deficiency in Cavalier King Charles spaniels with Pneumocystis pneumonia. J Vet Intern Med. 2006; 20(3): 523-527.
- Weissenbacher-Lang C, Fuchs-Baumgartiner A, Guija-De-Arespocachaga, Klang A, Weissenbock H, Kunzel F. Pneumocystosis in dogs: meta-analysis of 43 published cases including clinical signs, diagnostic procedures, and treatment. J Vet Diagn Invest. 2018; 30(1): 26-35.