CASE 3: 15-2890 (1-4) (4084553-00)
Signalment: Twelve-month-old, intact female NOD.Cg-Prkdcscid Il2rgtm1Wjl/SzJ (NSG) mouse, Mus musculus.
History: The animal was part of a cohort of NSG retired breeders experimentally infested with Syphacia obvelata. Health records indicated that the animal was clinically normal for 4-6 months post-infection. Subsequently, decreased appetite and marked weight loss were noted, along with lethargy and dehydration. The animal was euthanized and submitted for necropsy.
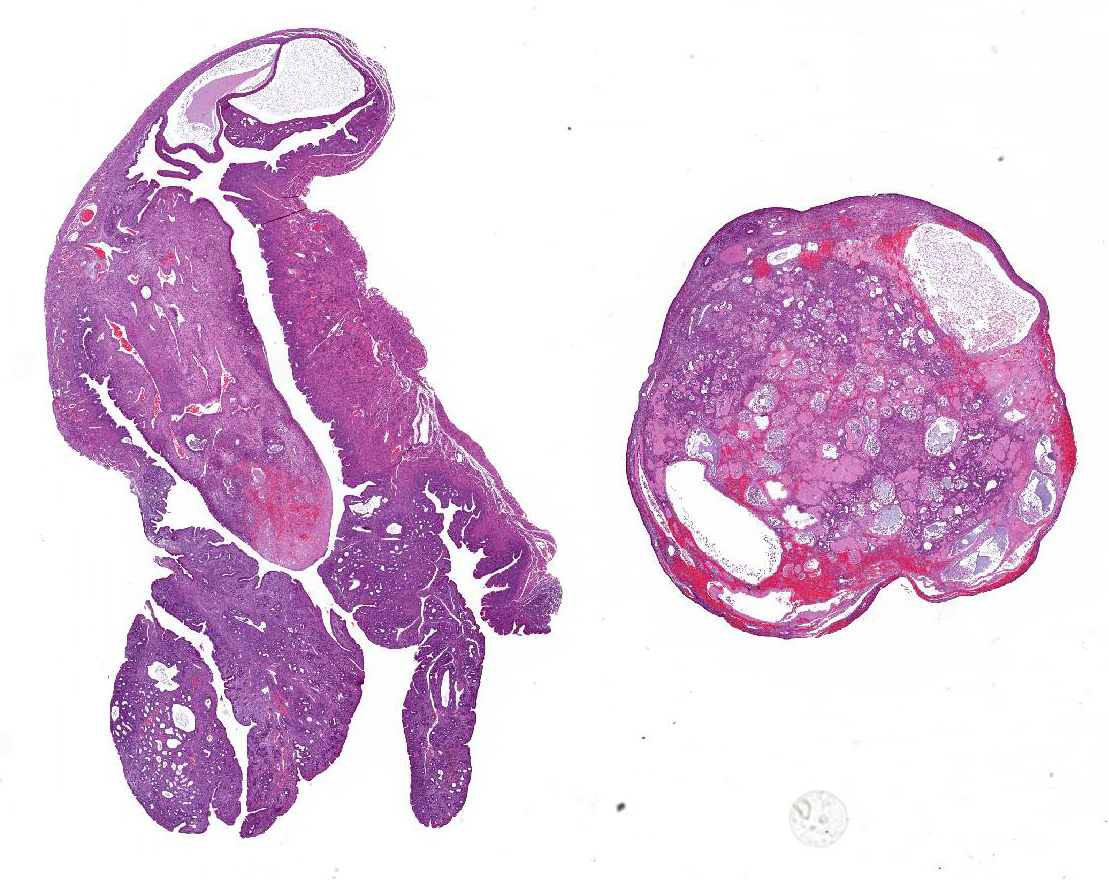
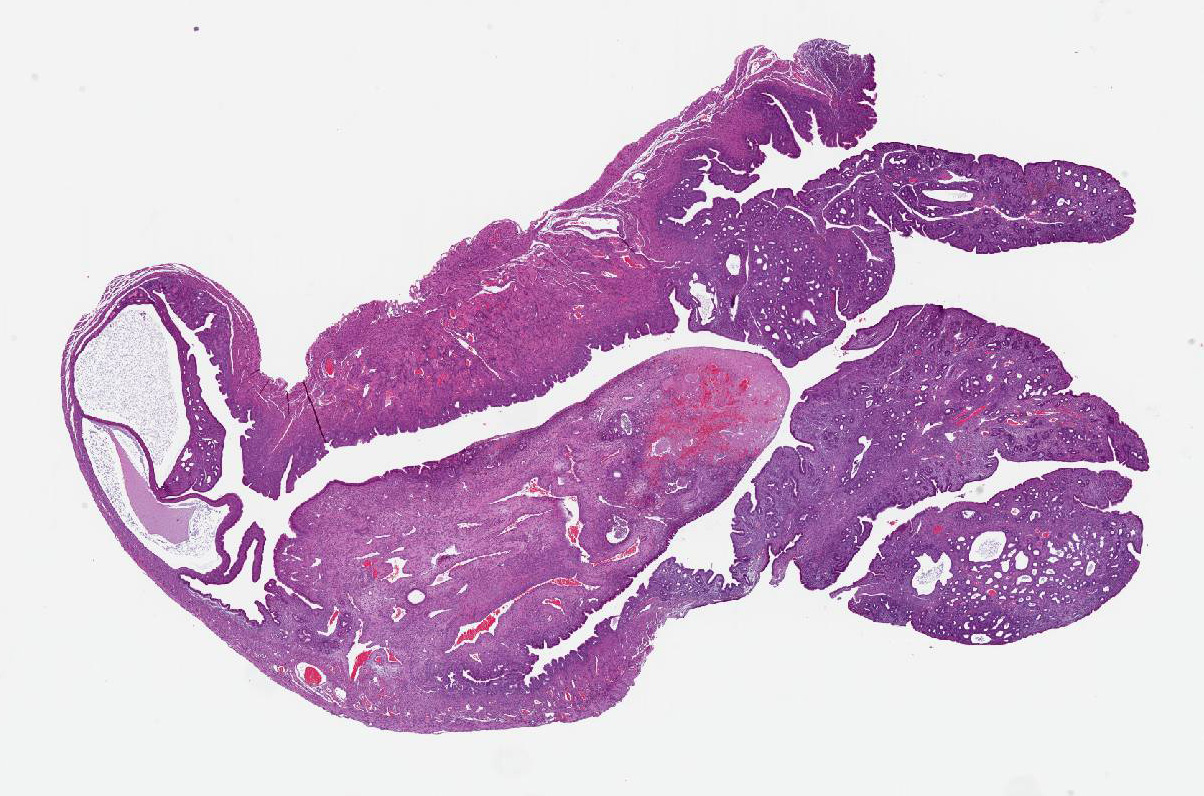
Gross Pathology: The uterine horns were diffusely thickened and slightly dilated and measured 5 to 6 mm in diameter. Upon opening, the uterine lumen was partially filled with a moderate amount of clear yellow viscous material.
Laboratory results: A swab of the uterine mucoid content was submitted for aerobic and anaerobic cultures, and no bacteria were isolated upon microbiology.
Microscopic description:
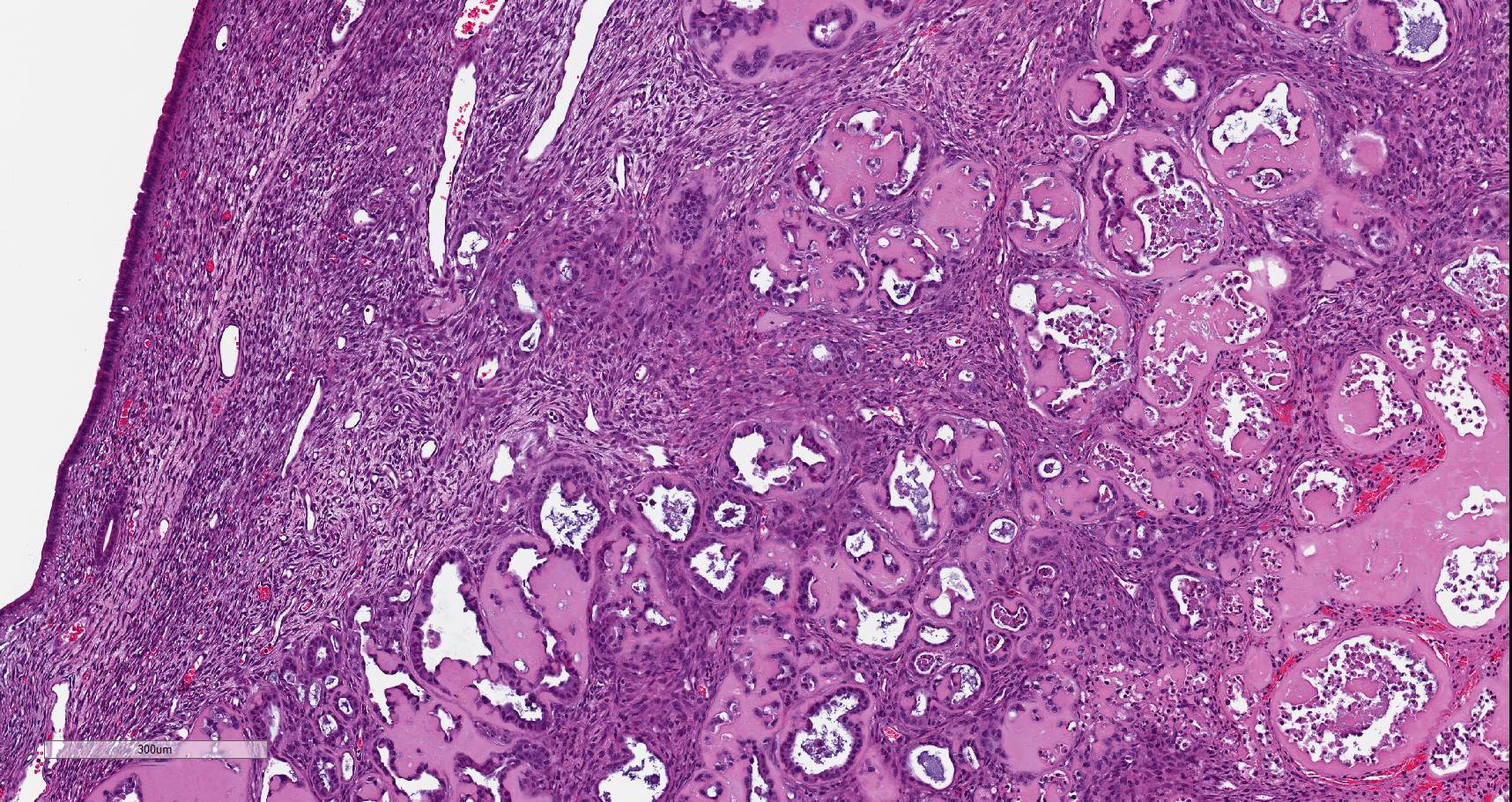
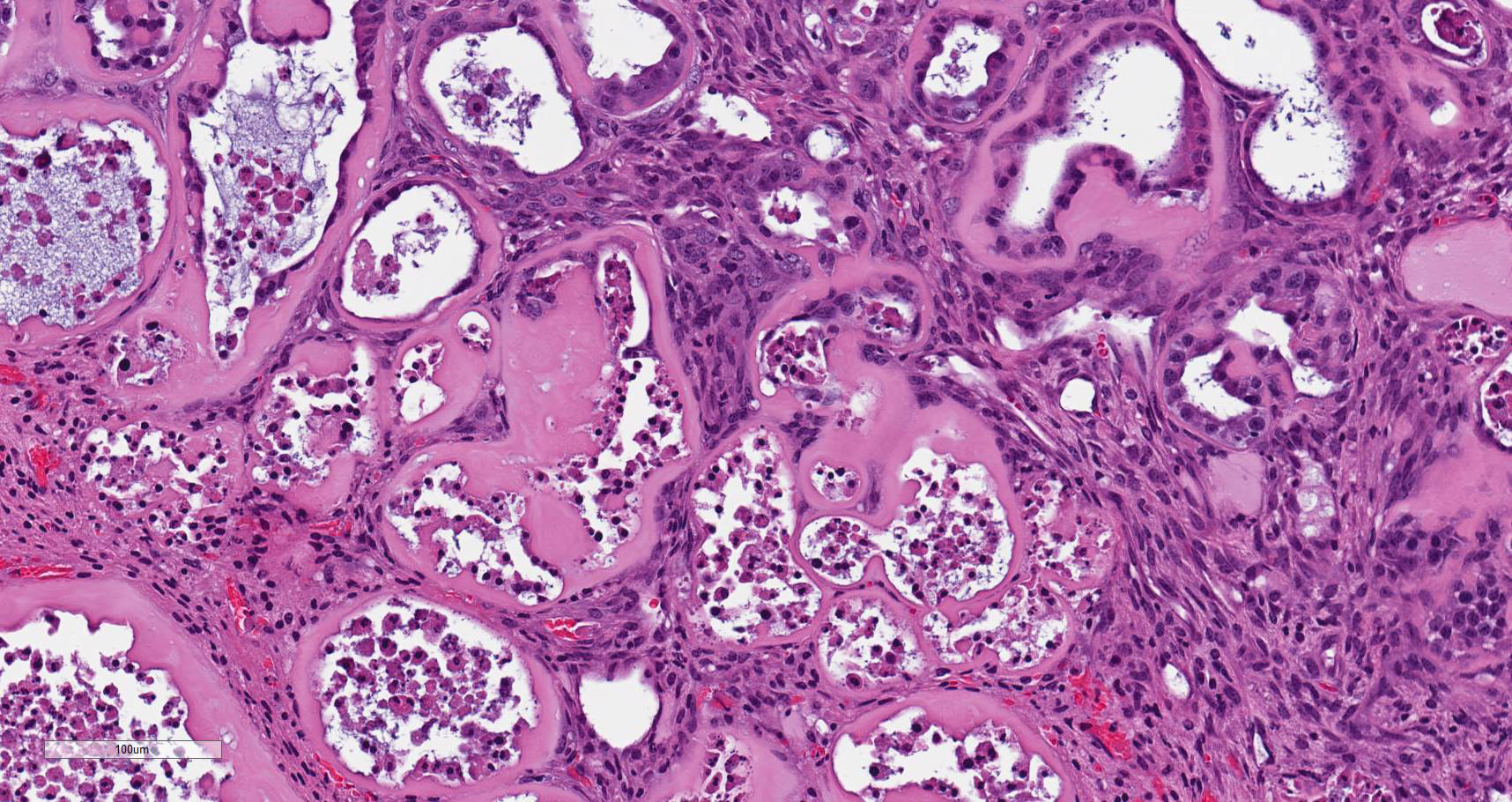
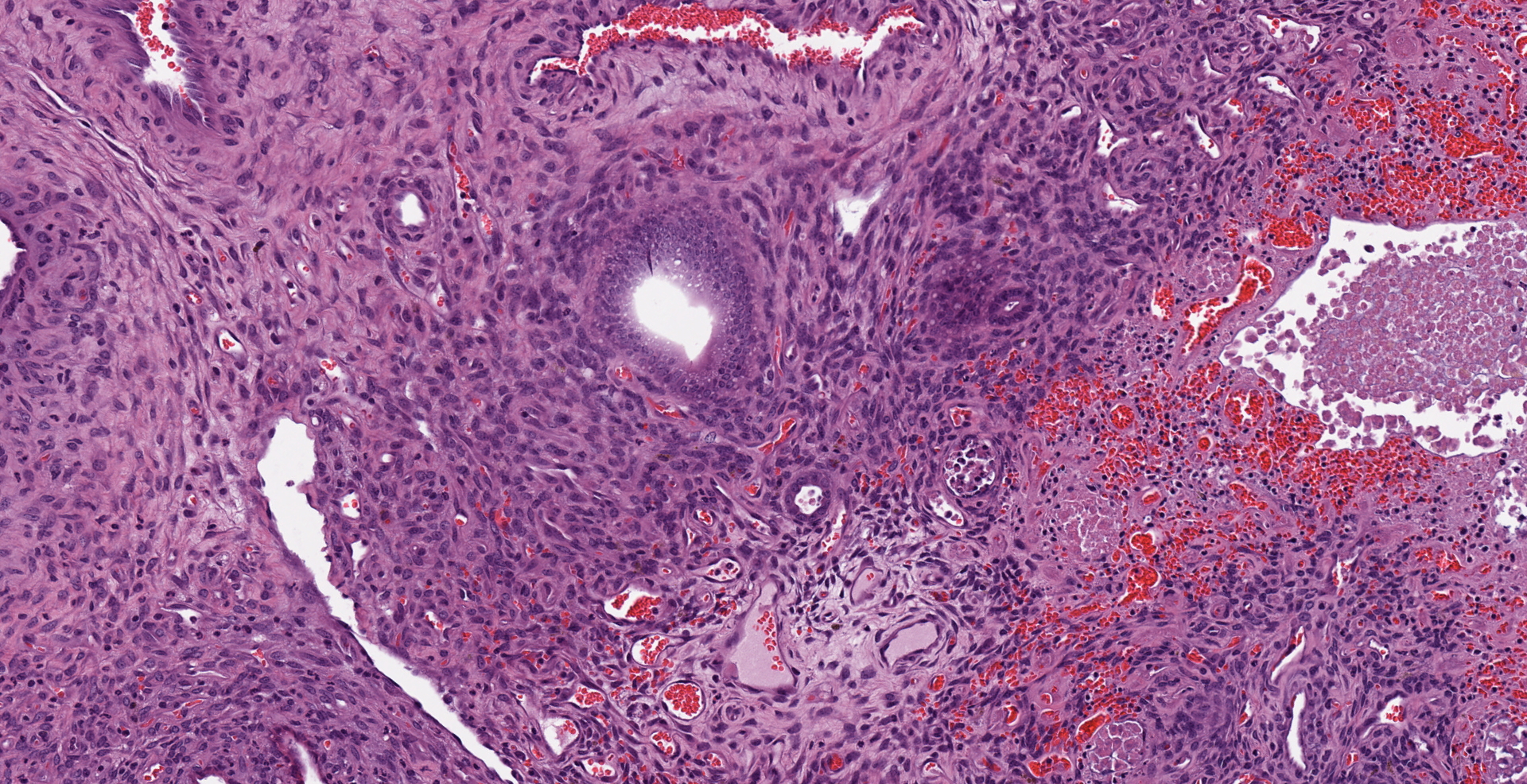
Uterus. Multifocally infiltrating the endometrium and subjacent myometrium and filling the lumen, there is a poorly demarcated, unencapsulated, scarcely cellular epithelial neoplasm composed of acini lined by a simple columnar epithelium, nests and ribbons. The cells are associated with an abundant extracellular eosinophilic hyaline material. Cells are cuboidal and polygonal, with distinct borders and a moderate amount of amphophilic and occasionally vacuolated cytoplasm. Nuclei are round to oval, with finely granular to vesicular chromatin and one or multiple prominent magenta nucleoli. There is moderate anisocytosis and anisokaryosis. The mitotic index is 15 (mitoses per 10 high power fields 40x). Scattered throughout the neoplasm, there are multifocal areas of coagulative necrosis and hemorrhages. In the adjacent section of more preserved uterus, there is moderate multifocal cystic dilation of endometrial glands, without increased mitotic activity of evidence of proliferative changes of the epithelium. There is also a focal endometrial polyp composed of fibrovascular stroma without glands and infiltrated by a small numbers of hemosiderin laden macrophages. The surface of the polyp is lined by a simple columnar epithelium, with focal ulceration associated with a neutrophilic and fibrinous exudate.
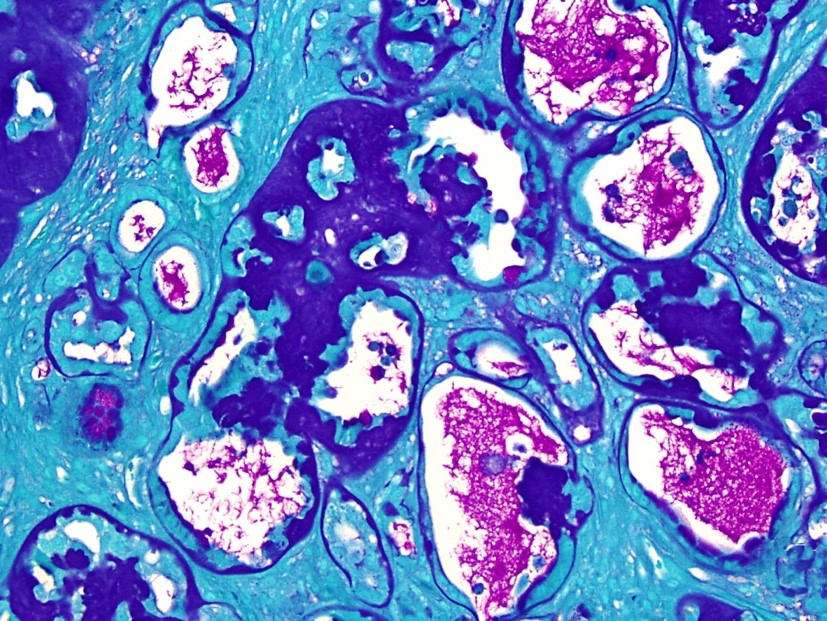
Special stains (Periodic acid-Schiff, Masson's trichrome and Congo red stains): The hyaline ground substance is negative for Congo red and positive for PAS. This material also stained blue with Masson's trichrome. Additionally, the neoplastic cells occasionally contain PAS-positive intracytoplasmic granules.
Immunohistochemical stain (laminin):
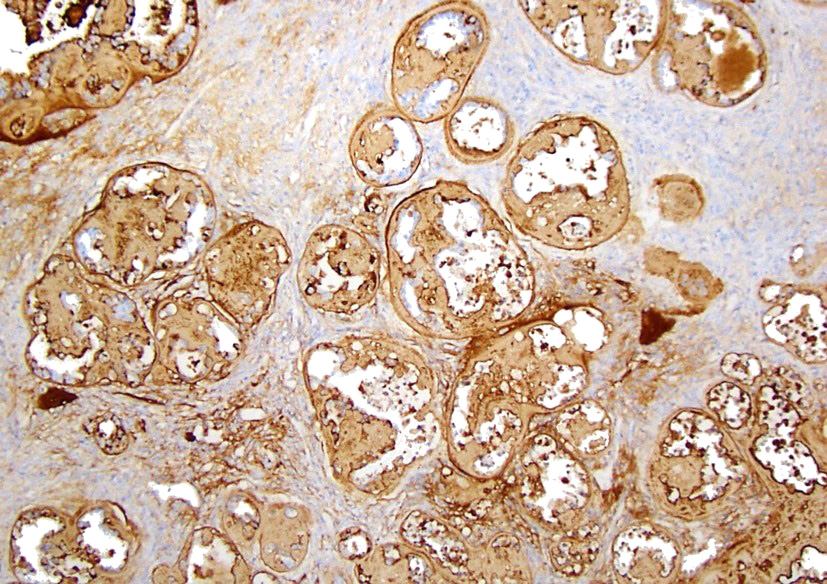
The hyaline ground substance is positive for laminin. Additionally, the neoplastic cells exhibited strong cytoplasmic positivity.
Contributor's morphologic diagnosis:
Uterus: Yolk sac carcinoma, parietal.
Uterus: Endometrial glandular cystic dilation, multifocal, moderate.
Uterus: Endometrial stromal polyp.
Contributor's comment: Yolk sac carcinomas, also known as malignant tumors of the endodermal sinus, represent a rare spontaneous neoplastic variant of germ cell tumors3 and have been previously reported in experimentally manipulated or control rodents.1,5,9,17,18 Spontaneous yolk sac carcinomas may be more commonly seen in BDII/Han rats,3 and have been found at the level of the reproductive system both in the ovaries and the uterus. Eight cases of primary uterine yolk sac carcinomas have been described in mice.18 Yolk sac carcinomas can be also experimentally induced in both rats and mice through implantation of an extraembryonic tissues or whole egg,10,15 or through intraperitoneal injection of neoplastic ascitic fluid.10 Their spontaneous occurrence in the testis has been reported in the SD rat and in a Swiss albino mouse.2,6
In humans, primary yolk sac tumors of the endometrium are extremely rare.7 Only nine cases have been reported in the literature, among which seven cases are pure yolk sac tumors and only two cases are in coexistence with endometrial carcinoma.11,12 On the contrary, they are mostly discovered in the gonadal tissues infants and adolescents (median age, 19 years), although 10% to 15% of the cases may arise in a variety of midline extragonadal sites with an axial distribution pattern.13 Yolk sacs of rodents and humans display several morphologic differences which give rise to the peculiar appearance of those tumors in the two species.18 Diagnostic feature of yolk sac carcinomas in humans include Schiller-Duval bodies and hyaline globules that stained positively for alpha fetoprotein.
In rodents, both spontaneous and induced yolk sac tumors have histologic patterns mimicking one or two layers of fetal membranes which are represented by the parietal and the visceral yolk sac. It has been reported that inducible yolk sac carcinoma is composed of endodermal cells and contains mesenchymal, trophoblastic and mesodermal cells.17
Parietal yolk sac tumor cells produce an abundant, eosinophilic, PAS-positive matrix in which nests and cords of neoplastic cells are embedded. The neoplastic cells often form rosettes, cords or papillary structures, and intracytoplasmic PAS-positive granules are also present.3,9,18 Visceral yolk sac tumor cells are large, cylindrical, lack of PAS-positive droplets and are positive for alpha fetoprotein.3,6,14
The hyaline ground substance characteristic of parietal yolk sac tumors is reminiscent of Reichert's membrane, a basement membrane that separates the visceral and parietal layers of the rodent yolk sac.9 Reichert's membrane is a specialized basement membrane that is not present in the human placenta. It surrounds the embryo, connects trophoblast cell layer on the maternal side and the parietal endodermal cells on the embryonic side, and passively filters nutrients; therefore, proper functioning of Reichert's membrane plays a crucial role in normal embryo development. Several methods have been reported to better characterize this hyaline substance, including silver impregnation, mucicarmine, PAS, PTAH, and staining for collagen.17 Both the neoplastic cells and the matrix stain positive for laminin,3,14,18 as documented here.
Presenting clinical signs in affected rodents include abdominal distension, ascites, and intraabdominal palpable masses. Metastatic spread of yolk sac carcinomas has been reported both in mice and rats, with neoplastic foci detected at the level of the peritoneal cavity, mediastinum and lungs.8,18 In the present case, no metastatic spread was evidenced in any of the organ examined.
The pathogenesis of these tumors remains poorly understood, as yolk sac tumors have been reported both in virgin animals,8,18 and in animals that have been mated.10 In the present case, the mouse was a retired breeder with a long term history of pregnancies and parturitions; however, the animal has not been mated since 6 months prior.
Contributing Institution:
Laboratory of Comparative Pathology
Hospital for Special Surgery
Memorial Sloan Kettering Cancer Center
The Rockefeller University, Weill Cornell Medicine
https://www.mskcc.org/research-areas/programs-centers/comparative-medicine-pathology
JPC diagnosis:
1. Uterus: Yolk sac carcinoma, mouse, rodent.
2. Uterus: Endometrial stromal polyp.
3. Uterus, endometrium: Cystic endometrial hyperplasia, diffuse, mild.
JPC comment: The contributor succinctly summarized the current knowledge about yolk sac carcinomas. This is a rare diagnosis to make, and much more commonly diagnosed in young human boys.
While this is a rare entity, it presents a good opportunity to review embryology and the development of the reproductive system. Unlike non-mammalian species, the mammalian embryo does not rely on stored yolk, though the structure of the yolk sac is present. The yolk sac is a bilaminar structure with an outer splanchnic mesoderm and inner extraembryonic endoderm. The yolk sac is continuous with the embryonic midgut.
All primordial germ cells, male and female, are derived from yolk sac endoderm. Development is initiated in the early epiblast, where factors such as BMP-4 transform the endodermal cells prior to their migration to the yolk sac. The cells cluster in the caudal surface of the yolk sac mesoderm, close to the primitive hindgut. From that position, they migrate to the wall of the hindgut through the allantoic stalk. Finally, the cells migrate through the dorsal mesentery to reach the genital ridges. It generally takes about three weeks for the cells to make it to the genital ridges, and about 2000 primordial cells ultimately survive the migration. Improper migration is extragonadal regions can result in teratomas. The extent of migration for these primordial germs cells is surpassed only by the migration of neural crest cells.8
While not of germ cell lineage, another common condition seen in a laboratory animal, specifically most commonly in the Fischer F344 rat, are endometrial stromal polyps. These occur singularly or as multiples, are usually pale tan or mottled red and tan, and are sessile with a long stalk. They are composed predominantly of loosely organized endometrial stromal cells, vascular supply, and few entrapped glands. The surface epithelium may either be similar to normal endometrial epithelium and low cuboidal or may exhibit squamous metaplasia. When there is an adenomatous component, the terms glandular or adenomatous polyp has been used.4 These must be distinguished from endometrial stromal sarcoma, which may also be polypoid, but exhibits invasion, poor differentiation of spindle cells, numerous mitotic figures, and high cellular pleomorphism.3
Cystic endometrial hyperplasia is one of the most common changes seen in intact female animals, including domestic and laboratory animals, humans, and exotic and wildlife species. It has been reported in nondomestic swine, red wolves, African wild dogs, raccoons, Asian elephants, and wild rabbits. While there is hyperplasia of components of the endometrium, there is prominent hyperplasia of glandular structures, with glands becoming cystic and often detectable macroscopically. A variant also exists where endometrial glands are not cystic and not grossly visible, but there is proliferation of glands (noncystic endometrial hyperplasia).16 Prolonged and excessive exposure to estrogen is responsible in some species but may also be caused by ingestions of certain plants, or iatrogenic exposure to estrogen containing creams.
References:
1. Alison RH and Morgan KT. Ovarian neoplasms in F344 rats and B6C3F1 mice. Environ Health Perspect 1987; 73:91-106.
2. Creasy D, Bube A, De Rijk E, Kandori H, Kuwahara M, Masson R, Nolte T, Reams R, Regan K, Rehm S, Rogerson P, Whitney K. Proliferative and nonproliferative lesions of the rat and mouse male reproductive system. Tox Pathol 2012; 40:40S-121S.
3. Dixon D, Alison R, Bach U, et al. Nonproliferative and Proliferative Lesions of the Rat and Mouse Female Reproductive System. J Toxicol Pathol 2014;27(3&4 Suppl):1S?107S.
4. Dixon D, Vidal JD, Leininger JR, Jokinen MP. Oviduct, Uterus, and Vagina. In: Sutttie AW Ed. Boorman's Pathology of the Rat, 2nd Ed. San Diego, CA: Elsevier. 2018:548.
5. Frith CS, Evans MG. Spontaneous ovarian choriocarcinoma, yolk sac carcinoma, and teratoma in B6C3F1 mice: a case Report. Tox Pathol 1993; 21 (1): 91-98.
6. Jamadagni SB, Jamadagni PS, Lacy Sh, Williams B, Upadhyay SN, Gaidhani SN, Hazra J. Spontaneous nonmetastatic choriocarcinoma, yolk sac carcinoma, embryonal carcinoma, and teratoma in the testes of a Swiss albino mouse. Tox Pathol 2013; 41:532-536.
7. Ji M, Lu Y, Guo L, Feng F, Wan X, Xiang Y. Endometrial carcinoma with yolk sac tumor-like differentiation and elevated serum ?-hCG: a case report and literature review. Onco Targets Ther 2013; 6:1515?1522.
8. Kumar MSA. Clinically oriented anatomy of the dog and cat. Ronkonkoma, NY: Linus Publications. 2012.
9. Majeedr SK, Alisong H, Boorman A, Gopinath C. Ovarian yolk sac carcinoma in mice. Vet Pathol 1986; 23: 776-778.
10. Nakahara W, Tokuzen R, Fukuoka F. A transplantable hyalinogenic tumor of the mouse. Gan 1967:59(5):475-47.
11. Oguri H, Sumitomo R, Maeda N, Fukaya T, Moriki T. Primary yolk sac tumor concomitant with carcinosarcoma originating from the endometrium: case report. Gynecol Oncol 2006; 103(1):368?371.
12. Patsner B. Primary endodermal sinus tumor of the endometrium presenting as ?recurrent? endometrial adenocarcinoma. Gynecol Oncol 2001; 80(1):93?95.
13. Rossi R, Stacchiotti D, Bernardini MG, Calvieri G, Lo Voi R. Primary yolk sac tumor of the endometrium: a case report and review of the literature. Am J Obstet Gynecol 2011; 204(4):e3-4.
14. Sakamoto A, Yamaguchi Y, Yamakaw S, Nagatani M, Tamura K. Highly metastatic ovarian yolk sac carcinoma in a rat. J Toxicol Pathol 2011; 24: 81?85.
15. Sakashita S, Tsukada Y, Nakamura K, Tsuji I, Hirai H. (1977). Experimental yolk-sac tumors produced by fetectomy without virus infection in rats. Int J Cancer 20, 83?86.
16. Schlafer DH, Foster RA. Female Genital System. In: Maxie MG ed. Jubb, Kennedy, and Palmer's Pathology of Domestic Animals, 6th Ed. Vol 3. St Louis, MO: Elsevier. 2016:383.
17. Sobis H, Van Hove L, Vandeputte M. Trophoblastic and mesenchymal structures in rat yolk sac carcinoma. Int J Cancer 1982; 29: 181-186.
18. Steward HL, Saas B, Deringer MK, Dunn, TB, Liotta LA, Togo S. J Natl Cancer Inst 1984; 73(1):115-122. Pure yolk sac carcinoma of the mouse uterus: report of 8 cases.