Signalment:
Three-week-old, male, Mox-2 Cre transgenic mouse (
Mus musculus).A mass was noted on left front leg.
Gross Description:
A firm mass encompassed the left front leg from the shoulder to
the paw (~2 cm in diameter).
Histopathologic Description:
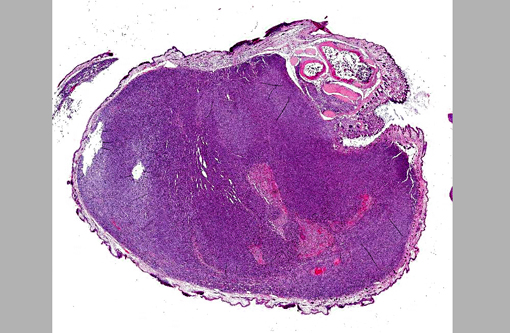
A cross-section of the left forelimb has an unencapsulated,
expansile, and infiltrative mass expanding the dermis, extending into the subcutis, and replacing
skeletal muscle. The mass is composed of densely-packed neoplastic cells haphazardly arranged
as long and short interweaving streams and bundles within a minimal fibrovascular matrix.
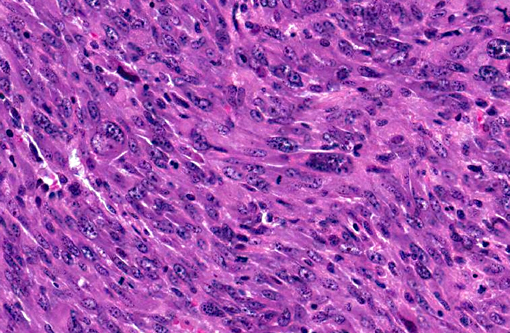
Neoplastic cells are predominantly spindle-shaped with some ovoid profiles; have abundant
granular to fibrillar amphophilic cytoplasm; and prominent round to oval nuclei with
marginalized chromatin and multiple magenta nucleoli. Many of these cells are quite long with a
fairly consistent width, and fusiform nuclei (presumed strap cells). Neoplastic cells exhibit
marked anisocytosis, anisokaryosis, and pleomorphism. Mitoses are numerous, varying between
~2 to ~10 per high-power field (40x). Multinucleated giant cells with abundant eosinophilic
cytoplasm and 2 to 5 nuclei clustered at the periphery are frequently present (racket cells).
Multifocal areas of the neoplasm are necrotic with hemorrhage and copious cytoplasmic and
karyorrhectic debris. Small bundles of skeletal muscle entrapped by neoplastic cells are present
at the periphery of the mass adjacent to sections of cortical bone. Mild edema (clear spaces) and
moderate aggregates of lymphocytes and plasma cells multifocally expand the superficial dermis.
The overlying epidermis has multiple variably sized ulcers with replacement by moderate
amounts of degenerate neutrophils and eosinophilic debris. Small pockets of degenerate
neutrophils are occasionally apparent within the stratum corneum.
Morphologic Diagnosis:
Left forelimb, skeletal muscle: Rhabdomyosarcoma.
Condition:
Rhabdomyosarcoma
Contributor Comment:
Spontaneous rhabdomyosarcomas in mice are rare with those of
skeletal muscle occurring more often than those of the heart.(6) Rhabdomyosarcomas are typically
induced experimentally via exposure to a variety of viruses, metals, and/or chemical
carcinogens.(6) An investigation at the Jackson Laboratory identified 14 spontaneous welldifferentiated
rhabdomyosarcomas out of 10,000 mice, approximately 4 months old.(7) Landau et
al(3) found a higher incidence of rhabdomyosarcomas (34% of controls); however, these mice
were approximately 14 months old.
The mouse currently described was transgenic for the Mox-2 gene, which is an important
regulator of vertebrate limb myogenesis.(4,8) Mox-2 is part of a cohort of genes important to
normal myogenic differentiation. Historically, mice homozygous for a null mutation of Mox-2
have a developmental defect of the limb musculature, characterized by an overall reduction in
muscle mass and elimination of specific muscles (www.jax.org).
Identification of strap cells may be difficult by light microscopy; however, phosphotungstic acidhematoxylin
(PTAH) stain is useful for identification of cross-striations.(6,5) Malignant fibrous
histiocytoma and leiomyosarcoma are differential diagnoses for rhabdomyosarcoma.(6)
Immunolabels useful for differentiating these neoplasms include pan myosin, sarcomeric actin,
desmin, actin, myosin, and smooth muscle actin.(7) The most useful antibodies are those that react
with sarcomeric or smooth muscle actin.
JPC Diagnosis:
Skeletal muscle, left forelimb: Rhabdomyosarcoma.
Conference Comment:
Rhabdomyosarcomas (RMS) occur infrequently in domestic animals,
as they do in mice. A recent publication reviewed the classification and pathogenesis of this
diverse group of rare tumors, comparing canine rhabdomyosarcomas with those that occur in
humans and with other canine soft tissue sarcomas.(1) Although in veterinary medicine
rhabdomyosarcomas are often categorized as high grade soft tissue sarcomas, they are excluded
from the soft tissue sarcoma grading scheme as recently proposed by Dennis et al.(2)
Diagnosis and classification is difficult due to their variation in phenotype, cellular morphology
and age of onset. It is likely that some RMS are diagnosed as undifferentiated sarcomas,
anaplastic sarcomas or poorly differentiated sarcomas, since skeletal muscle differentiation
is not always evident by light microscopy. Therefore, immunohistochemistry can aid in the
diagnosis. In addition to the immunolabels discussed by the contributor, MyoD1 and myogenin
(early embryological transcription factors involved in mesoderm cell differentiation into
myoblasts, myoblast proliferation and myoblast differentiation into myotubules) are associated
with RMS of more undifferentiated cells.
Transmission electron microscopy can also aid in the diagnosis; however, EM is not helpful in
classification, as several subtypes exhibit similar subcellular structures, including Z-lines,
numerous mitochondria, myofilament tangles, and myosin-ribosome complexes.(1)
Canine classification of RMS is similar to the human classification of RMS, with the following
subclasses:
- Embryonal, in which cells occur in different stages of development (from myoblast to
myotubular) on a mucinous stroma. These occur in both juveniles and adults, and occur
more frequently on the face, skull, within masticatory muscles, the oropharynx, trachea,
axilla, scapula, perirenal, tongue, flank, leg, mammary gland, and hard palate. In the
myotubular variant of embryonal RMS myotubule forms predominate; whereas large
myoblast cells predominate in the rhabdomyoblastic variant and streams of plump spindle
cells predominate in the spindle cell variant.(1)
- Botryoid RMS have a characteristic submucosal location and gross appearance that
resembels grape-like masses. Histologically they appear as mixed round, undifferentiated
myoblast cells and multinucleated myotubular cells on a mucinous stroma. These tend to
occur in the urinary bladder or uterus in juveniles.(1)
- Alveolar RMS occur in juveniles and are usually found in the hip, maxilla, greater
omentum or uterus. The classic variant is characterized by fibrous bands that divide
small round cells into clusters and loose aggregates, while the solid variant is composed
of closely packed cells with or without a thin fibrous septa.(1)
- Pleomorphic RMS typically occur in adult skeletal muscle. They are characterized by
haphazardly arranged plump spindle cells with marked anisocytosis and anisokaryosis
and bizarre mitotic figures.(1)
More studies are needed to determine if these classifications have prognostic significance in
veterinary medicine.1
References:
1. Caserto BG. A comparative review of canine and human rhabdomyosarcoma with emphasis on classification and pathogenesis.
Vet Pathol Online First. Published online 25 February 2013. Accessed online 2 March 2013.
2. Dennis MM, McSporran KD, Bacon NJ, Schulman FY, Foster RA, Powers BE. Prognostic factors for cutaneous and subcutaneous soft tissue sarcomas in Dogs.
Vet Pathol. 2011;48(1):73-84.
3. Landau JM, Wang ZY, Yang GY, Ding W, Yang CS. Inhibition of spontaneous formation of lung tumors and rhabdomyosarcomas in A/J mice by black and green tea.
Carcinogenesis.
1998;19:501-507.
4. Mankoo BS, Collins NS, Ashby P, Grigorieva E, Pevny LH, Candia A, et al. Mox2 is a component of the genetic hierarchy controlling limb muscle development.
Nature. 1990;400:69-
73.
5. Leininger JR. Skeletal muscle. In: Maronpot RR, ed.
Pathology of the Mouse. St. Louis, MO; Cache River Press. 1999;640-642.
6. Mohr U, ed. International Classification of Rodent Tumors,
The Mouse. Berlin, Heidelberg: Springer-Verlag. 2001;385, 386.
7. Sundberg JP, Adkison DL, Bedigian HG. Skeletal muscle rhabdomyosarcomas in inbred laboratory mice.
Veterinary Pathology. 1991;28:200-206.
8. Tiffin N, Williams, RD, Robertson D, Hill S, Shipley J, Pritchard-Jones K. WT1 expression does not disrupt myogenic differentiation in C2C12 murine myoblasts or in human
rhabdomyosarcoma.
Experimental Cell Research. 2003;287:155165.