Signalment:
Gross Description:
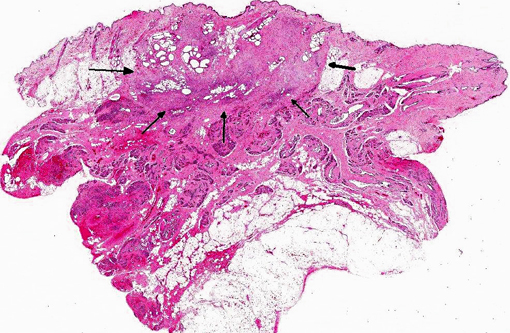
Histopathologic Description:
Morphologic Diagnosis:
Condition:
Contributor Comment:
The present case is a 7-year-old Maltese bitch with a history of ovariohysterectomy performed a year before the tumor occurrence. No other external abnormalities were observed. Lumpectomy rather than mastectomy was performed. This bitch was still alive at the time this manuscript was prepared (8 months post-surgery) despite the malignant features observed on histopathology. Continuous follow up for two year survival will be interesting.
Classification of canine mammary tumors has been complicated and debatable. The earliest classification scheme for canine and feline mammary tumors was seen in 1961, in Moultons Tumor in Domestic Animals. The classification at that time was fairly simple; however, over the years, the classification scheme has further advanced to more detailed and complex ones, including the 1974 WHO edition of International Histological Classification of Tumors of Domestic Animals, and the 1999 Armed Forces Institute of Pathologys classification. In 2011, a new classification scheme was proposed.(2)
JPC Diagnosis:
Conference Comment:
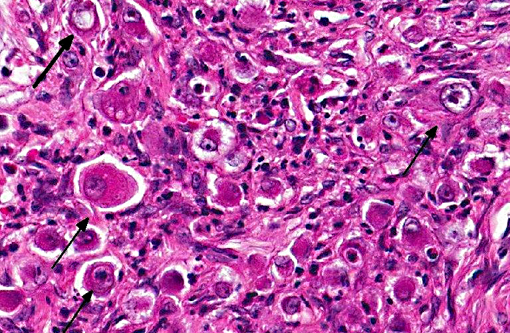
As the name implies, neoplastic cells are pleomorphic, round to polygonal, with moderate to abundant eosinophilic cytoplasm, and round to oval nuclei which are sometimes indented. Neoplastic cells are often individualized or grouped in small nests. Features of malignancy include frequent multiple prominent nuclei, severe anisokaryosis and anisocytosis and a high mitotic rate; occasional multinucleated neoplastic cells are also present.(1)
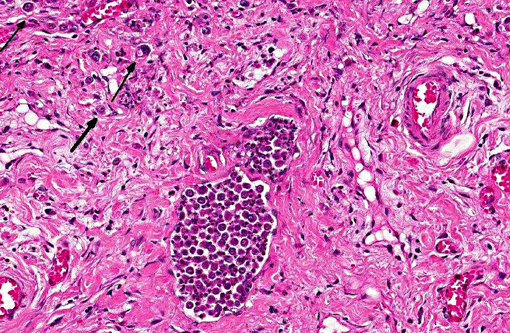
Conference participants discussed use of the term inflammatory carcinoma, which should not be used as a morphologic diagnosis, as it refers to a clinical entity that can be associated with several types of malignant mammary carcinomas, including anaplastic carcinomas. The hallmark histologic lesion for this condition is invasion of neoplastic emboli into dermal lymphatics2 and despite the name, and the clinical appearance of a reddened mammary neoplasm, which is warm to the touch, there is often little to no microscopic inflammation associated with the neoplasm.Â
In addition, inflammatory carcinomas are highly angiogenic.(1) Tumor angiogenesis can be accomplished via several mechanisms, including production of vessels through endothelial precursor cells from the bone marrow or from endothelial cells in preexisting vessels, by sprouting angiogenesis, by intussusceptive angiogenesis, and by vessel co-option. More recently, a new mechanism, vasculogenic mimicry (VM) has been identified. Vasculogenic mimicry consists of the de novo generation of microvascular channels by genetically deregulated aggressive tumors cells without participation of endothelial cells. The resulting channels are not true blood vessels, although they function to distribute plasma and blood cells to the neoplasm, and are thought to play a role in metastasis. VM has been identified in several types of malignant tumors in humans, including inflammatory breast carcinoma and ductal breast carcinoma, as well as in several types of inflammatory mammary carcinomas in dogs, with its occurrence seen most frequently in anaplastic carcinomas. Highly malignant neoplastic cells in canine mammary cancer have been observed to resemble endothelial cells that histologically, immunohistochemically, and ultrastructurally resembled VM as described in human tumors. Specifically, the endothelial-like cells (ELCs) in VM are positive for epithelial markers and for the same markers used for the rest of the tumor cells may be positive for vimentin, but negative for smooth muscle actin and desmin, and show absence of specific immunoreaction with endothelial markers. Ultrastructurally, ELCs lack Weibel-Palade bodies (which are characteristic of endothelial cells) and have desmosomes (the type of junctions between epithelial cells) instead of fascia occludens (endothelial cell- to-cell junctions). Tumor and/or blood cells contained in the channels formed by ELCs are not inside a vacuole as in emperipolesis or phagocytosis; instead, they are inside an actual space formed by the cytoplasmic membranes of ECLs. It is thought that ECLs can also form channels that mimic lymph vessels rather than blood vessels; however, further studies are needed to identify the mechanisms of cancer cells developing channels of VM as well as confirm the presence of lymphatic VM.(1)
References:
2. Goldschmidt M, Pen+�-�a L, Rasotto R, Zappulli V. Classification and grading of canine mammary tumors. Vet Pathol. 2011:48(1):117-131.Â