CASE 3: N19-187 (4152932-00)
Signalment:
Adult male Virginia opossum (Didelphis virginiana)
History:
The patient was a wild opossum and presented to the wildlife clinic by a concerned member of the community for ill-thrift. On physical examination, the patient was severely dehydrated and emaciated. On full body radiographs, numerous radiolucent, pinpoint foci were identified throughout all bones in the axial and appendicular skeleton (Radiograph 1; Radiograph 2). The patient was euthanized due to poor prognosis.
Gross Pathology:
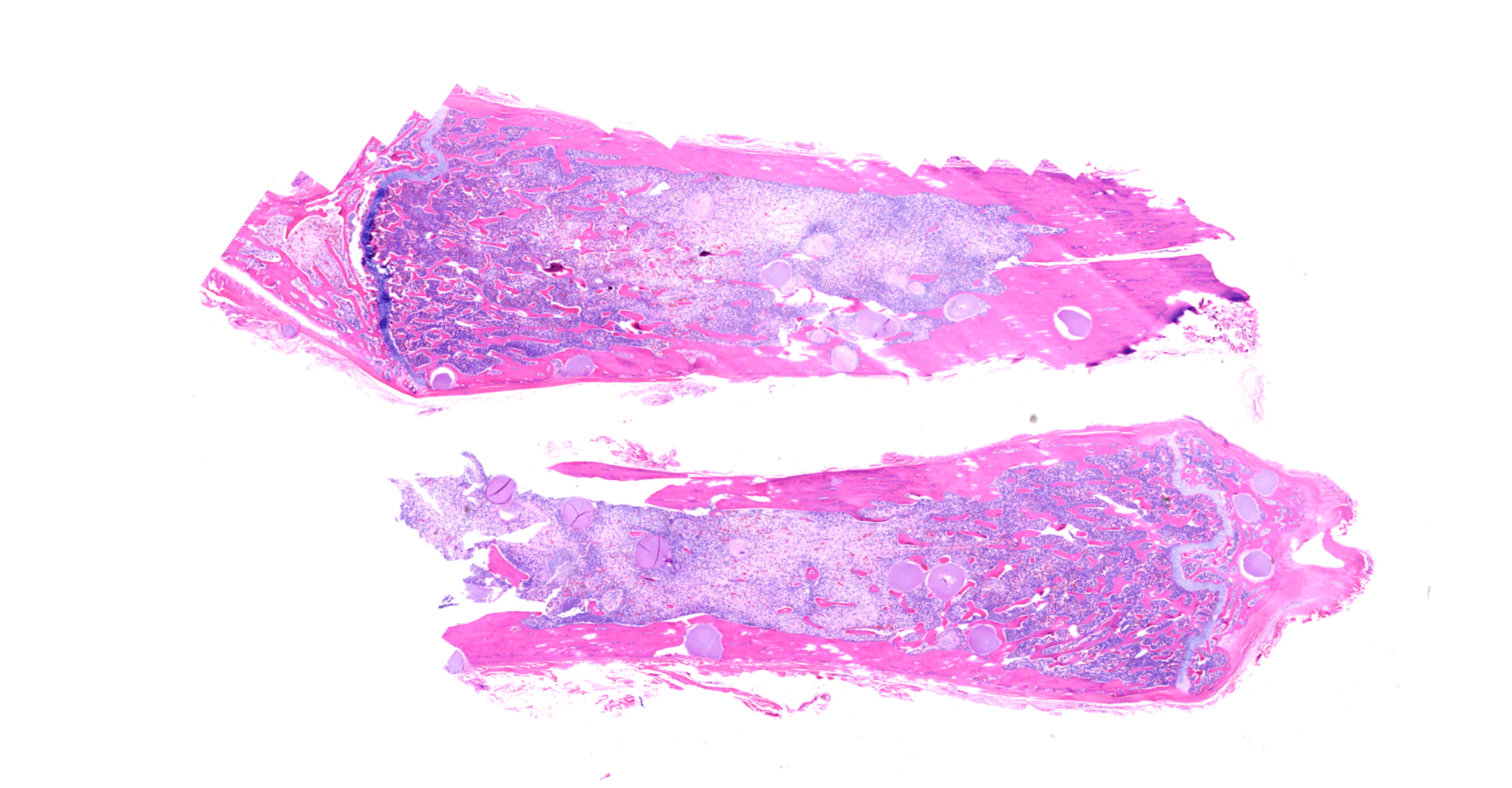
On gross postmortem examination, the patient was severely emaciated and dehydrated. The pericardial and bone marrow pericardial adipose were translucent, gelatinous, and pale yellow (serous atrophy of fat). Randomly distributed throughout the skeletal muscle, visceral organs, axial and appendicular skeleton, eyes, and within the lumen of large veins were thousands of pinpoint, spherical, firm, off-white foci.
The stomach was void of ingesta, but contained 30 to 40 round, off-white, 1.25 cm long and 0.1 cm in diameter nematodes with tapered ends.
Laboratory results:
None.
Microscopic description:
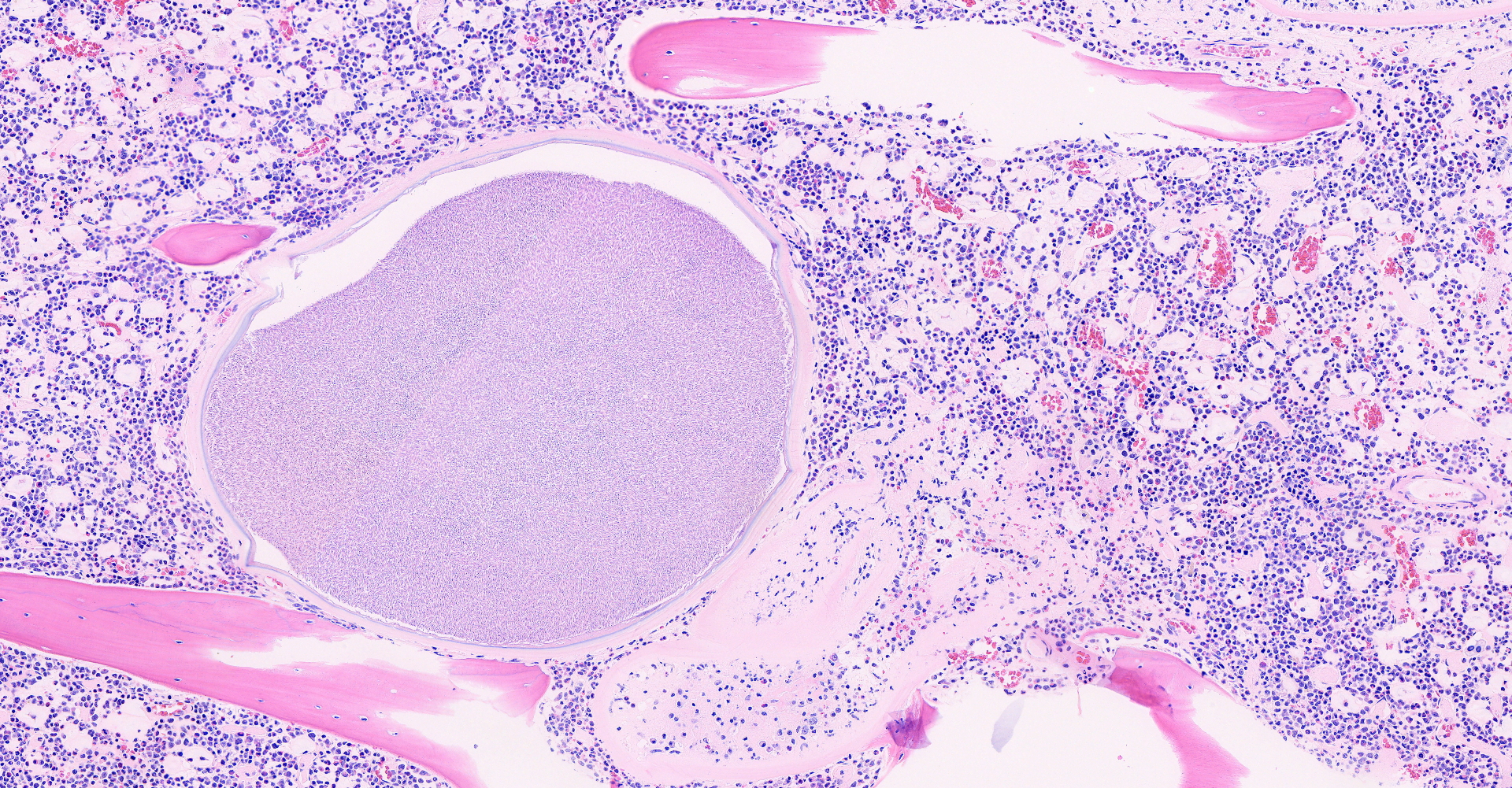
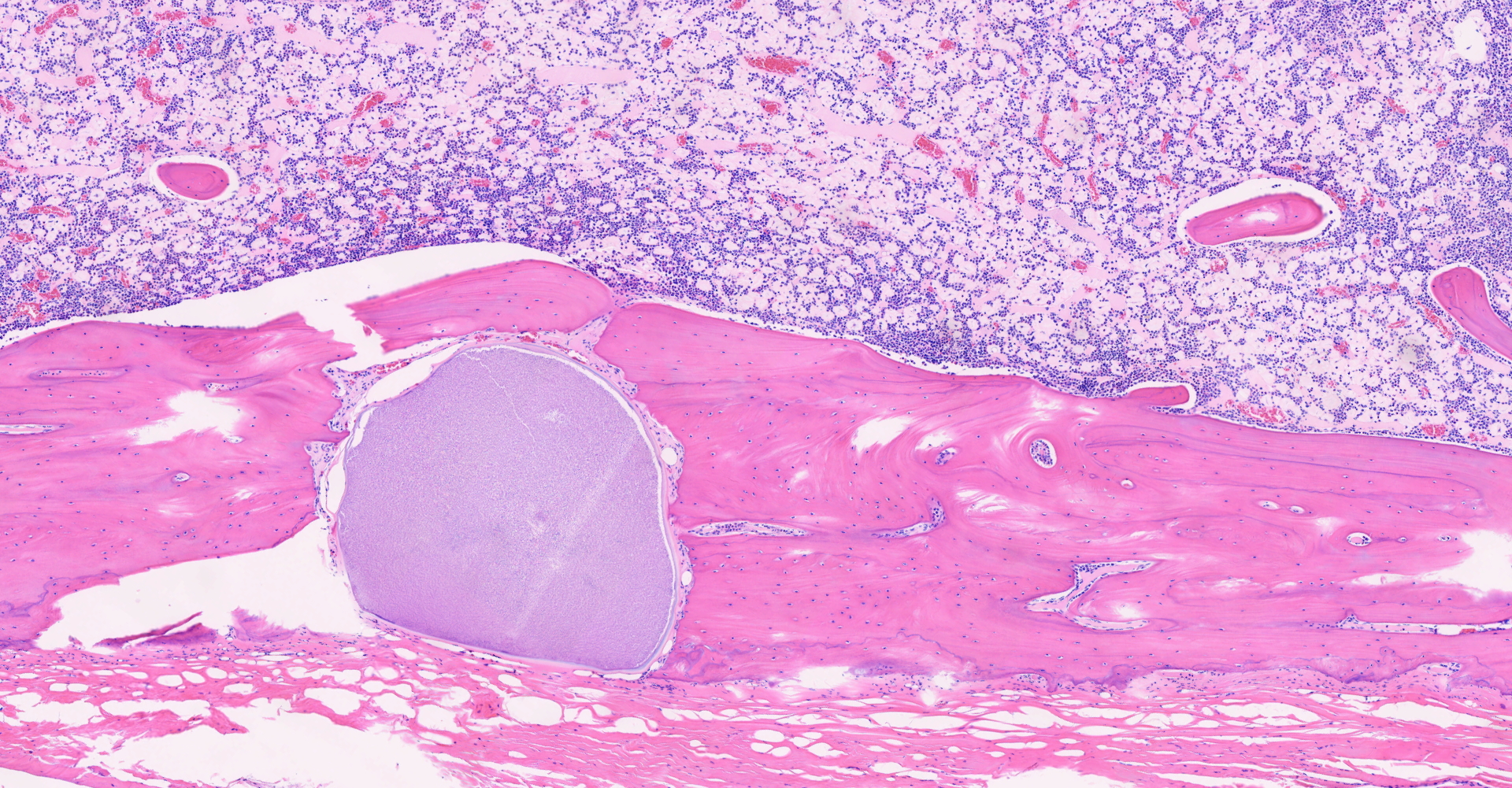
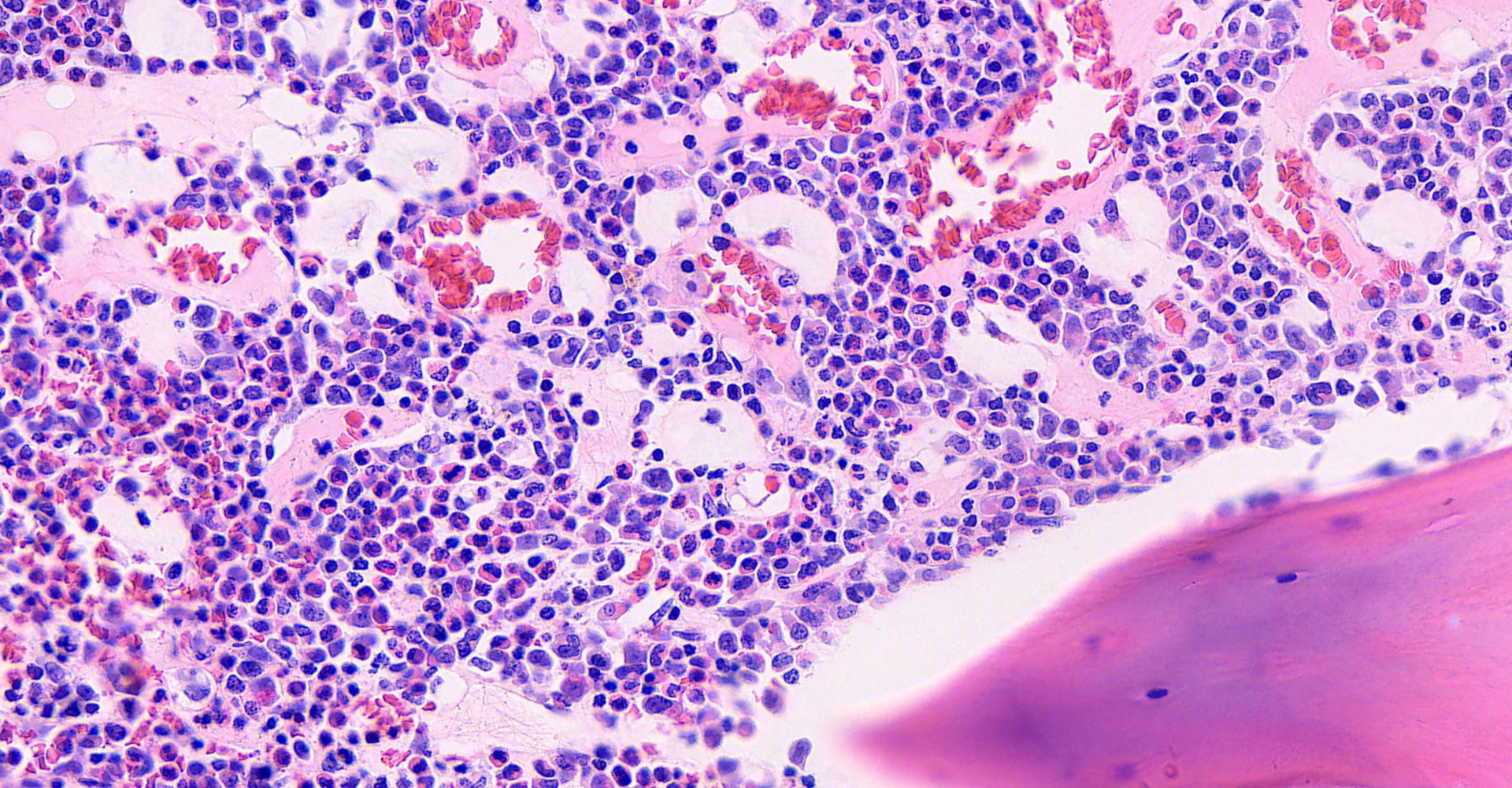
Long bone: Randomly distributed throughout the cortical bone, and within the medullary cavity, are numerous, round, protozoal cysts, ranging in size from 120 to 300 um in diameter. The cyst wall is 15 µm wide and amphophilic and the cyst contains thousands of spindloid, eosinophilic bradyzoites with a single, basophilic nucleus. A moderate number of cysts are necrotic, characterized by a round lightly eosinophilic structure with a fragmented, discontinuous cyst wall and lack of bradyzoites. Necrotic cysts are encircled by a moderate, dense band of macrophages and degenerate leukocytes that occasionally infiltrate the cyst remnant. The medullary cavity is hypercellular with granulocytic hyperplasia. Bone marrow adipocytes are decreased in number and size and frequently replaced by lightly basophilic, wispy material (serous atrophy of fat).
Contributor's morphologic diagnosis:
Long bone, bone marrow: disseminated coccidiosis (consistent with Besnoitia darlingi cysts) with marked medullary granulocytic hyperplasia and serous atrophy of fat.
Stomach (not submitted): moderate, focally extensive gastritis with intraluminal nematodes.
Contributor's comment:
The combination of patient signalment, gross findings, and the morphology of the protozoan cysts were deemed consistent with Besnoitia spp, likely Besnoitia darlingi. Besnoitia are Apicomplexan protozoa, within the Sarcocystidae family, and infect a number of wild and domestic mammals, as well as reptiles.2,7 Felids have been identified experimentally and naturally as the definitive host for a number of Besnoitia spp, including Besnoitia darlingi.7,8 Opossums serve as an intermediate host.5 Diagnosis is based on anatomic lesions, histopathology, immunofluorescence, and electron microscopy.7
The pathogenicity of Besnoitia is variable and while the underlying causes have not been fully elucidated, young age, immunosuppression, and stress have been suggested as predisposing factors for clinical disease.2 Infection frequently causes no adverse effects, although animals may have numerous cysts in the skin, striated muscle, and visceral organs.2 In cattle, Besnoitia besnoitti is an economically important disease; in severe infections, it can result in reduced fertility in females and reduced reproductive efficacy in breeding bulls, secondary to orchitis.3
In opossums, Besnoitia darlingi has historically been considered non-pathogenic, with numerous cysts in the skin, striated muscle and/or visceral organs with lack of an inflammatory response. However, there are reports of severe disease, debilitation, or death.2 In cattle, immunosuppression has been implicated as a factor in the development of severe cases and skin lesions.5 Previous reports of disseminated Besnoitia in opossums have been associated with immunosuppression in the form of recent parturition, poor body condition, and gastrointestinal and respiratory parasitism.5 In this case, the high parasite load may have predisposed the patient to severe emaciation and stress, allowing dissemination of Besnoitia darlingi. However, it is difficult to determine if the Besnoitia dissemination is cause or effect of emaciation. Another contributing factor was the presence of cysts within the anterior chamber of both eyes, which may have compromised eyesight, making foraging difficult.
Contributing Institution:
Cummings School of Veterinary Medicine at Tufts University
Pathology department
200 Westboro Rd
North Grafton, MA 01536
https://vetmed.tufts.edu/pathology-service/
JPC diagnosis:
1. Long bone, medullary cavity, Haversian canals and periosteum: Apicomplexan cysts, disseminated, with mild granulomatous osteomyelitis.
2. Long bone: Serous atrophy of fat, diffuse, marked.
3. Long bone: Myeloid hyperplasia, diffuse, marked.
JPC comment:
The contributor summarizes this entity in opossums concisely. Within the Besnoitia genus, species of importance include this B. darlingi and B. tarandi in opossums, B. besnoitia in cattle, B. bennetti in donkeys, B. caprae in goats, B. wallacei in rats, B. neotomofelis in woodrats, B. oryctofelisi in rabbits, and B. jellisoni in mice. As stated by the contributor, for several species for which the life cycle has been described, the cat is the definitive host, shedding viable oocysts in feces that are morphologically similar to those of Toxoplasma gondii.1
Despite understanding the life cycle of 4 of the 10 species of Besnoitia, the exact mode of transmission remains undetermined. It is currently thought that mechanical transmission by blood-sucking insects is a primary route of transmission, but other routes may exist. In the early stages of disease, tachyzoites in intermediate hosts proliferate in host endothelial cells, monocytes, and neutrophils. As the infection progresses, tissue cysts form, as seen in this case. Each cyst represents a parasitized host cell, with the growing bradyzoites inducing hyperplastic and hypertrophic changes.6
The Virginia opossum also has other host adapted parasites that cause little disease in this definitive host. When considering potential disease in horses kept near natural surroundings, it is important to remember that Didelphis virginiana is the definitive host for Sarcocystis falcatula, and possibly also the definitive host for S. neurona. These noble animals are also a reservoir host for Leishmania brasiliensis, an important agent that causes human and animal mucocutaneous leishmaniasis.1,4
References:
1. Bowman DD. Georgis' Parasitology for Veterinarians, 10th Ed. St. Louis, MO:Elsevier. 2014;109.
2. Ellis, Angela. Debilitation and Mortality Associated with Besnoitiosis. Journal of Zoo and Wildlife Medicine. 2012. 43(2): 367-374.
3. Frey, Caroline F., et al. Besnoitia besnoiti lytic cycle in vitro and differences in invasion and intracellular proliferation among isolates. Parasit Vectors. 2016. 9: 115.
4. Higgins D, Rose K, Spratt D. Monotremes and Marsupials. In: Terio KA, McAloose D, St. Leger J, eds. Pathology of Wildlife and Zoo Animals. San Diego, CA:Elsevier. 2018:473-474.
5. L. Diesing. Besnoitia besnoiti: studies on the definitive host and experimental infection in cattle
6. Mauldin EA, Peters-Kennedy J. Integumentary System. In: Maxie MG, ed, Jubb, Kennedy, and Palmer's Pathology of Domestic Animals, 6th Ed. St. Louis, MO:Elsevier. 2016;661-663.
7. Shaw, Shannon, et. al. Besnoitia darlingi infection in a Virginia Opossum (Didelphis virginiana). Journal of Zoo and Wildlife Medicine. 2009. 40 (1): 220-223.
8. Verma, Shiv. K, et al. Bobcats (Lynx rufus) are natural definitive host of Besnoitia darlingi. Veterinary Parasitology. 2017. 248 (2017): 84-89.