Signalment:
Gross Description:
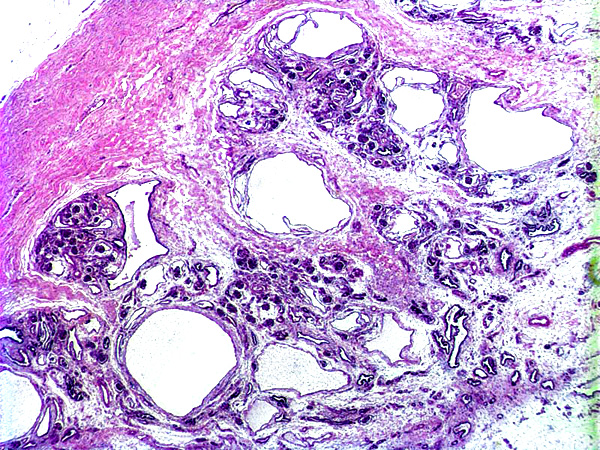
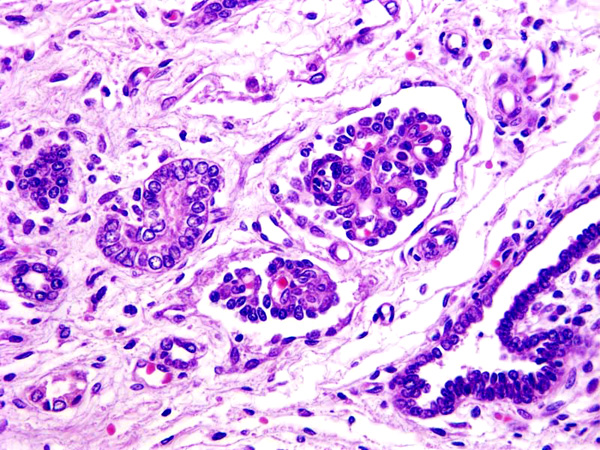
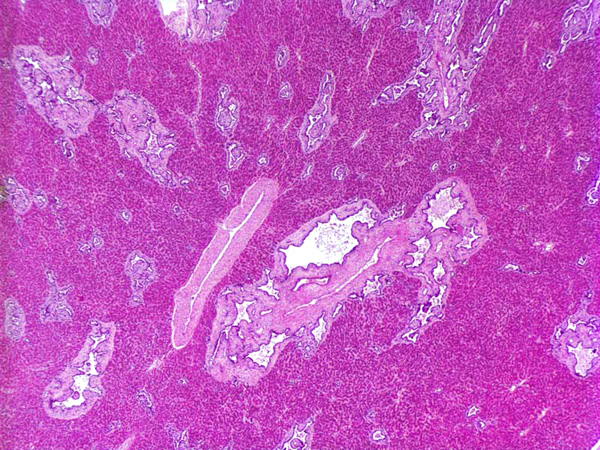
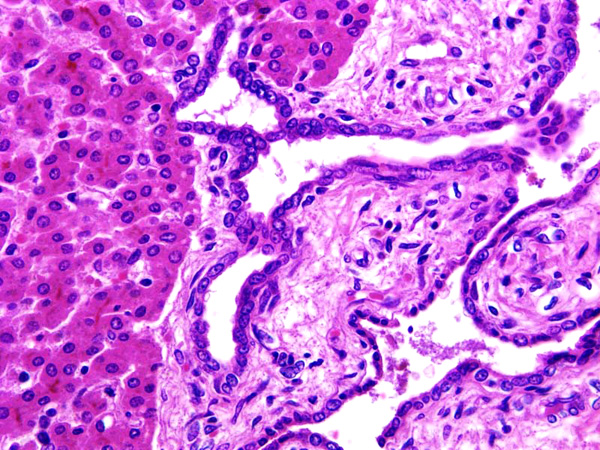
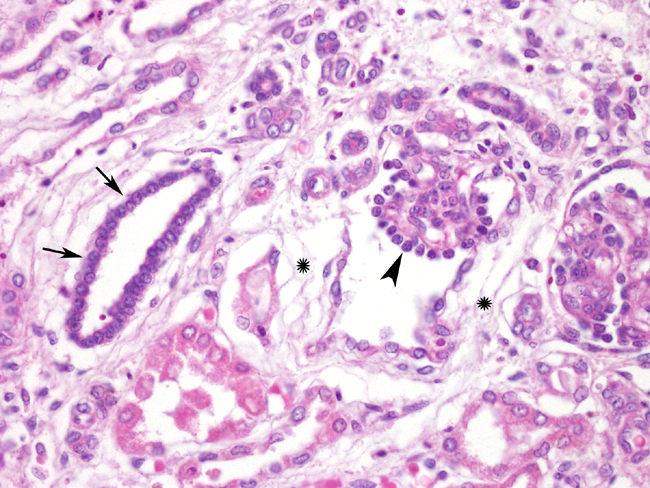
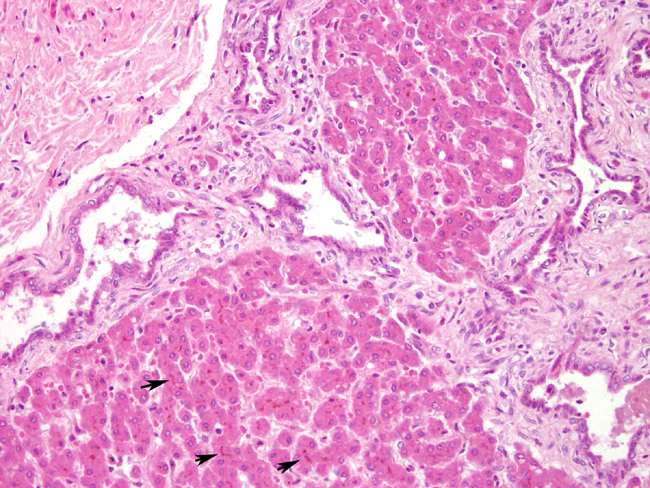
Histopathologic Description:
Morphologic Diagnosis:
1. Kidney: Cystic renal dysplasia
2. Liver: Bile duct ectasia and hyperplasia associated with portal fibrosis
Condition:
Contributor Comment:
- 1. Cystic renal dysplasia
- 2. Polycystic kidney disease
- a. Autosomal-dominant (adult) polycystic disease
- b. Autosomal-recessive (childhood) polycystic disease
- 3. Medullary cystic disease
- a. Medullary sponge kidney
- b. Nephronophthisis
- 4. Acquired (dialysis-associated) cystic disease
- 5. Localized (simple) renal cysts
- 6. Renal cysts in hereditary malformation syndromes (e.g., tuberous sclerosis)
- 7. Glomerulocystic disease
- 8. Extraparenchymal renal cysts (pyelocalyceal cysts, hilar lymphangitic cysts) Renal dysplasia is defined as disorganized development of renal parenchyma due to abnormal differentiation. This disease refers to a developmental disorder of renal parenchyma due to imperfect inductive interaction between the mesonephric duct and the matanephric blastema.2
In human beings renal dysplasia can be unilateral or bilateral and is almost always cystic. The abnormalities in the collecting system are common. In gross appearance, the kidney is usually enlarged, extremely irregular, and multicystic. The cysts vary in size from microscopic structures to some that are several centimeters in diameter.11
In dog the histologic features used to diagnose renal dysplasia include the prescence of 1) fetal or immature glomeruli, 2) fetal or immature tubules, 3) persistent metanephric ducts surrounded by primitive mesenchyma, 4) bone or cartilage in the parenchyma, and 5) anomalous presence of interstitial fibrous connective tissue.5,12 Renal dyspalsia has been reported in many breeds, such as the Golden Retriever, Labrador Retiever, Shih Tzu, Bull Mastiff, Boxer, and Finish Harrier 3,5,6,8,9,13-16 and human patients.11
In Rhesus macaque (Macaca mulatta) adult polycystic kidney disease and infantile polycystic kidney disease has been reported, and in Cynomolgus monkey (Macaca fascicularis) a case of spontaneous congenital polycystic kidney has been also described.3,7,17
In this case, the histological examination of the liver showed hyperplasia with cystic and saccular dilatations of the intraepithelial bile ducts lined by low cuboidal epithelial cells associated with overgrowth of portal connective tissue. In human being abnormalities of the biliary tree are an heterogeneous group of congenital lesions (von Meyemburg complexes, polycystic liver disease, congenital hepatic fibrosis, and Caroli disease) in which the primary abnormality is altered architecture of the intrahepatic biliary tree.4
The precursor of the intrahepatic biliary tree is a double-layered sleeve of cells know as the ductal plate (DP). The DP first arises from hepatocyte precursors surrounding hilar portal vein vessels, and more peripheral regions of the DP then develop sequentially. During the remainder of gestation, a process of DP remodeling occurs in which small areas of the double layer separate to form tubules, which join to form the intrahepatic biliary tree, while the remaining regions of the DP involute. Hepatic fibropolycystic diseases are thought to originate from failures in this process and are known collectively as DP malformations. However depending on the size of the bile ducts affected and the time during organogenesis, different syndromes, with marked overlap, can be differentiated. Caroli disease is a subcategory of these diseases, characterized by multiple cystic and segmental saccular dilations of the larger intrahepatic bile ducts.16 Caroli syndrome involves malformations of smaller bile ducts and congenital hepatic fibrosis, marked by portal tract enlargement with irregular and broad bands of collagenous tissue, in which variable numbers of abnormally shape bile ducts are embedded.4,5 Caroli syndrome is often associated with autosomal recessive polycystic kidney disease (ARPKD), and both the hepatic and renal processes reflect developmental process in the context of different organs. 4,5
The present report described a congenital cystic renal dysplasia associated with bile ducts ectasia and prominent portal fibrosis in a new born Pigtailed macaque (Macaca nemestrina), a combination not previously reported in macaques.Â
The finding in our case of renal and bile duct dysplasia mimics previously reported cases in human and rat of Caroli syndrome.Â
JPC Diagnosis:
1. Kidney: Renal dysplasia, characterized by fetal glomeruli, primitive mesenchyme, immature tubules, tubular ectasia and cysts, and interstitial and capsular fibrosis, with minimal lymphoplasmacytic interstitial nephritis and mineralization.
2. Liver: Biliary duct hyperplasia, diffuse, marked, with biliary duct ectasia, portal fibrosis, and cholestasis.
The contributor gives a good review of the different forms of cystic kidney diseases with emphasis on renal dysplasia. There is debate in the literature on what features are necessary for the diagnosis of renal dysplasia. Although cartilaginous or osseous metaplasia is described in the human literature, it is rarely present in dysplastic kidneys of animals.11
Conference Comment:
Renal dysplasia has been associated with fetal infections of feline panleukopenia virus, canine herpesvirus, and bovine viral diarrhea virus, as well as hypovitaminosis A during gestation in swine.11 There is not an apparent connection between the renal dysplasia and the biliary duct hyperplasia with congenital hepatic fibrosis (Caroli Syndrome) in the present case.Â
It is possible that the small cysts seen in this case are due to autosomal recessive polycystic kidney disease (ARPKD). It is important to make the distinction that although ARPKD is associated with Caroli syndrome, it is not a defining feature of the disease process.Â
1. Abraham LA, Beck C, Slocombe RF: Renal dysplasia and urinary tract infection in a Bull Mastiff puppy. Aust Vet J 81:336-339, 2003
References:
2. Alpers CE: The kidney. In: Robbins and Cotran Pathologic Basis of Disease, eds. Kumar V, Abbas, AK, Fausto N, 7th ed., pp. 955-1021. Elsevier Saunders, Philadelphia, PA, 1999
3. Baskin GB, Roberts JA, McAfee RD: Infantile polycystic renal disease in a rhesus monkey (Macaca mulatta). Lab Anim Sci 31:181-183, 1981
4. Crowford JM: The liver and biliary tract. In: Robbins and Cotran Pathologic Basis of Disease, eds. Kumar V, Abbas, AK, Fausto N, 7th ed., pp. 877-937. Elsevier Saunders, Philadelphia, PA, 1999
5. Desmet VJ: Pathogenesis of ductal plate malformation. J Gastroenterol Hepatol 19:S356-S360, 2004
6. DiBartola SP, Chew DJ, Boyce JT: Juvenile renal disease in related Standard Poodles. J Am Vet Med Assoc 183:693-696, 1983
7. Hoppe A, Karlstam E: Renal dysplasia in boxers and Finnish harriers. J Small Anim Pract 41:422-426, 2000
8. Kessler MJ, Roberts JA, London WT: Adult polycystic kidney disease in a rhesus monkey (Macaca mulatta). J Med Primatol 13:147-152, 1984
9. Kerlin RL, Van Winkle TJ: Renal dysplasia in golden retrievers. Vet Pathol 32:327-329, 1995
10. Lobetti RG, Pearson J, Jimenez M: Renal dysplasia in a Rhodesian ridgeback dog. J Small Anim Pract 37:552-555, 1996
11. Maxie MG, Newman SJ: Urinary system. In: Jubb, Kennedy, and Palmers Pathology of Domestic Animals, ed. Maxie MG, 4th ed., vol. 2, pp. 439-443. Elsevier Limited, St. Louis, MO, 2007
12. Newman SJ, Confer AW, Panciera RJ: Urinary system. In: Pathologic Basis of Veterinary Disease, eds. McGavin MD, Zachary JF, 4th ed., pp. 625-627. Elsevier, St. Louis, MO, 2007
13. Picut CA, Lewis RM: Microscopic features of canine renal dysplasia. Vet Pathol 24:156-163, 1987
14. Miyamoto T, Wakizaka S, Matsuyama S, Baba E, Ohashi F, Kuwamura M, Yamate J, Kotani T: A control of a golden retriever with renal dysplasia. J Vet Med Sci 59:939-942, 1997
15. Morita T, Michimae Y, Sawada M, Uemura T, Araki Y, Haruna A, Shimada A: Renal dysplasia with unilateral renal agenesis in a dog. J Comp Pathol 133:64-67, 2005
16. Ohara K, Kobayashi Y, Tsuchiya N, Furuoka H, Matsui T: Renal dysplasia in a Shih Tzu dog in Japan. J Vet Med Sci 63:1127-1130, 2001
17. Schulze C, Meyer HP, Blok AL, Schipper K, van den Ingh TS. Renal dysplasia in three young adult Dutch kooiker dogs. Vet Q 20:146-148, 1998
18. Sakakibara I, Honjo S: Spontaneously occurring congenital polycystic kidney in a cynomolgus monkey (Macaca fascicularis). J Med Primatol 19:501-506, 1990 - 5. Localized (simple) renal cysts