Signalment:
Female, 11-year-old, Taita falcon (
Falco fasciinucha).
This animal was one of seven captive Taita falcons (
Falco fasciinucha) that died within 3 weeks during the breeding season of 2005. The falconer had 7 breeding pairs. Four of his females and three of his males died after being lethargic and anorexic for few hours to one day. Three of the four female animals died about one day after laying an egg.
Gross Description:
The animal was in a good to fair postmortem preservation state. It was anemic and had marked hemorrhage into the reproductive tract. The liver was diffusely beige. The spleen was mildly to moderately enlarged and had multiple beige well demarcated foci, up to 2 mm in diameter.
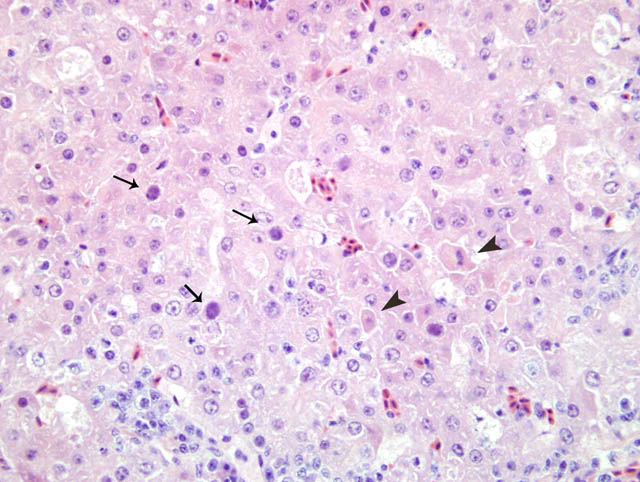
Histopathologic Description:
The nuclei of numerous hepatocytes contained one large basophilic inclusion body
(Fig. 3-1). Occasionally, a halo was present around the intranuclear inclusion and the nuclei membrane was hyperchromatic. A low to moderate number of necrotic individual hepatocytes were scattered within the parenchyma. Multiple portal fields were infiltrated by a moderate number of lymphocytes, plasma cells and macrophages.
Morphologic Diagnosis:
1. Hepatitis, necrotizing, multifocal, moderate with abundant hepatocellular intranuclear inclusion bodies.
2. Hepatitis, portal, lymphoplasmacytic and histiocytic, mild to moderate.
Lab Results:
The animal was negative for adenovirus-specific antibodies in 2002. Nucleic acid of falconid adenovirus was detected by PCR in the liver. The liver was negative for dependovirus (parvovirus) nucleic acid, herpesvirus nucleic acid, polyomavirus nucleic acid and chlamydial nucleic acid. Aviadenoviral nucleic acid was also detected in the liver by
in situ hybridization. Adenoviral particles were present in the intestinal content. Few non-hemolytic
E. coli were isolated from the liver by aerobic culture.
Condition:
Falcon adenovirus
Contributor Comment:
Primary histological differential diagnosis in this case included herpesvirus hepatitis and adenovirus hepatitis. The clinical presentation and finally the results of electron microscopy,
in situ hybridization and PCR supported and confirmed a diagnosis of adenovirus hepatitis. Recently, a falconid adenovirus has been identified.
3 This virus may be fairly widespread in indigenous species, such as peregrine falcons in which it does not seem to cause clinical disease.
2 However, if naive tropical falcon species become infected with the virus, it may cause fatal peracute disease.
2,3,5
JPC Diagnosis:
1. Liver, hepatocytes: Degeneration and necrosis, single cell, random, moderate, with basophilic intranuclear inclusion bodies, Taita falcon (
Falco fasciinucha), avian.
2. Liver: Hepatitis, portal, lymphoplasmacytic, multifocal, moderate.
Conference Comment:
The recently identified falcon adenovirus, most closely related to fowl adenovirus type 1 and 4, has been associated with disease in a variety of falcons, with a particularly severe course of infection within falcons originating from more isolated island/tropical regions or within the smaller falcon species such as kestrels and merlins.
2,3 The disease appears to be widespread within the Peregrine falcons (
Falco peregrinus) population which are considered to be a potential reservoir species for the falcon adenovirus.
1,2,4,5
There are four genera within the family Adenoviridae (mastadenovirus, Aviadenovirus, Atadenovirus, and Siadenovirus).
3 Aviadenovirus is further subdivided into subgenera and groups based on shared neutralizing epitopes.Â
Major groups of the Aviadenoviruses, adapted from Schrentzel et al.
3 and tomaszewski et al.
4
| Name |
Fowl adenovirus 1
(FAV-1) | Chickens | Includes chicken embryo lethal orphan strain (CELO).
Tropic for hepatocytes, pancreatic acinar cells, and gizzard epithelium |
Hemorrhagic enteritis
virus (HEV) | Turkeys | Non-pathogenic in pheasants; Produces severe enteritis in turkeys and guinea fowl |
Egg drop syndrome
(EDS) | Chickens | Little or no disease in waterfowl;
Reproductive abnormalities in chickens |
| Falcon adenovirus | Falcons | Necrotizing hepatitis and splenitis |
Characteristic histopathological findings include a multifocal, necrotizing hepatitis and splenitis, with hepatocytes, biliary epithelium, and mononuclear cells resembling lymphocytes containing intranuclear basophilic inclusion bodies that frequently marginate the chromatin.
1,3,4 Electron microscopic evaluation will demonstrate a 70-90nm nonenveloped icosahedral viral particle occasionally arranged in paracrystalline arrays.
3 In one report of infection in gyrfalcon and peregrine falcon hybrids, intranuclear inclusions were identified within renal tubular epithelial cells and not hepatocytes, suggesting that disease presentation may be species dependent.
5 Other lesions reported in different species include, vasculitis, heterophilic nephritis, myocardial necrosis, and hemorrhagic enteritis.
1-3,5
Conference participants are encouraged to examine a similar case in a Northern aplomado falcon (
Falco femoralis septentrionalis) that was presented in the
1996-1997 Wednesday Slide Conference number 29, Case 4, and subsequently published by Schrenzel et al.
3
References:
1. Dean J, Latimer KS, Oaks JL, Schrenzel M, Redig PT, W+�-+nschmann A: Falcon adenovirus infection in breeding Taita falcons (
Falco fasciinucha). J Vet Diagn Invest 18:282-286, 2006
2. Oaks JL, Schrenzel M, Rideout B, Sandfort C: Isolation and epidemiology of falcon adenovirus. J Clin Microbiol 43:3414-3420, 2005
3. Schrenzel M, Oaks JL, Rotstein D, Maalouf G, Snook E, Sandfort C, Rideout B: Characterization of a new species of adenovirus in falcons. J Clin Microbiol 43:3402-3413, 2005
4. Tomaszewski EK, Phalen D: Falcon adenovirus in a n American kestrel (
Falco sparverius). J Avian Med Surg 21:135-139, 2007
5. Van Wettere AJ, W+�-+nschmann A, Latimer KS, Redig PT. Adenovirus infection in Taita falcons (
Falco fasciinucha) and hybrid falcons (
Falco rusticolus x Falco peregrinus). J Avian Med Surg 19:280-285, 2005