Signalment:
Gross Description:
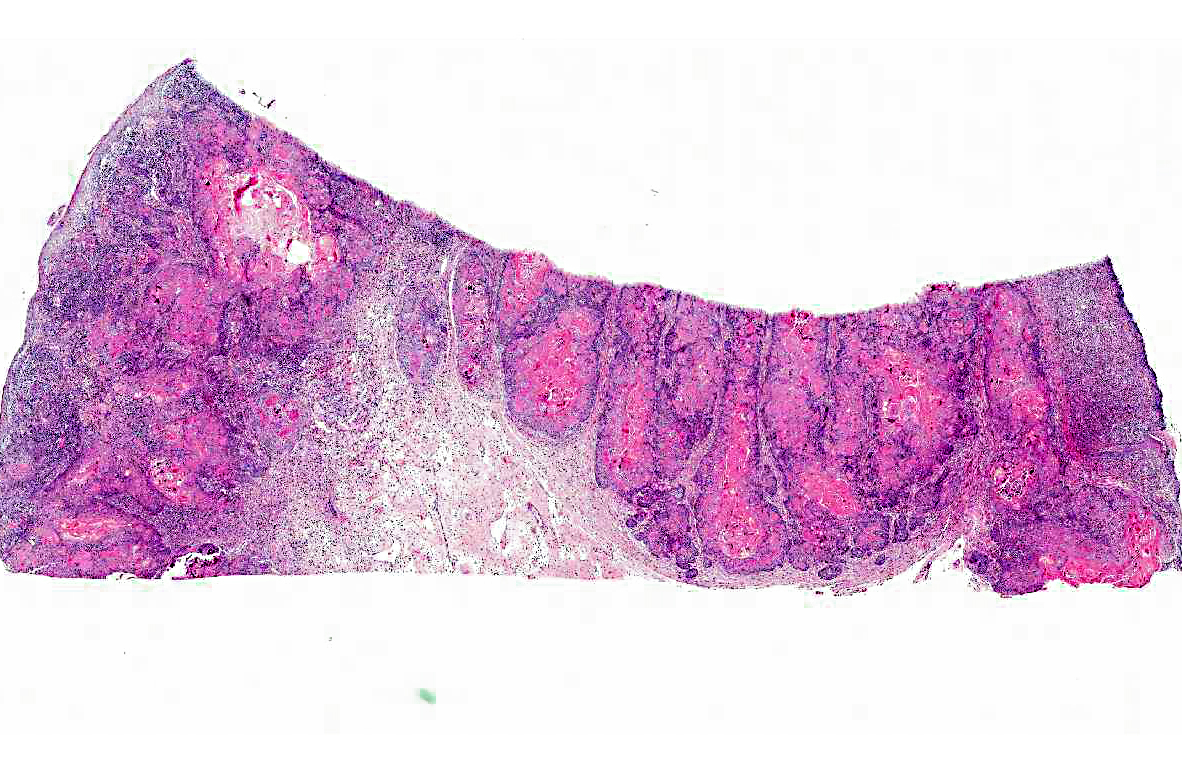
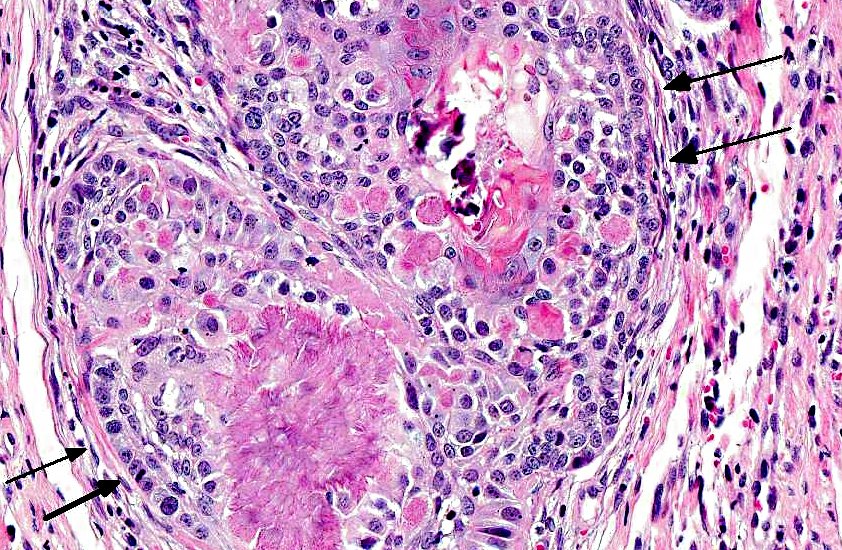
Histopathologic Description:
Morphologic Diagnosis:
Condition:
Contributor Comment:
In humans, a neoplasm similar to amyloid producing odontogenic tumor in animals is named a calcifying epithelial odontogenic tumor (CEOT). Amyloid-producing odontogenic tumor was originally referred to as the counterpart of human CEOT. However, it was later described that APOT in dogs and cats is not a counterpart of CEOT in humans. Human CEOTs consist of sheets of eosinophilic epithelial cells showing considerable nuclear pleomorphism and invasive growth, whereas APOTs in animals mostly show basal cells with hyperchromatic nuclei, are arranged in palisades, and are benign masses that grow by expansion.(5) Due to such differences, amyloid-producing odontogenic tumor (APOT) was proposed as an appropriate alternative term for CEOTs in animals.(4)
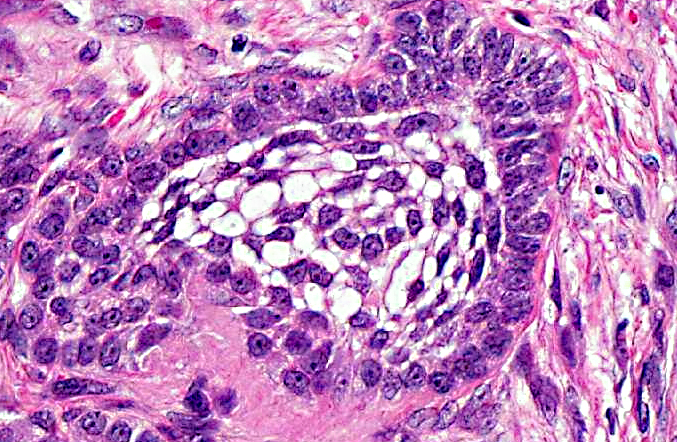
Amyloid-producing odontogenic tumor is characterized by dental epithelium, with deposits of amyloid and sometimes prominent trabeculae of osteoid (dentinoid). The epithelium may be arranged in strands, nests or masses. Occasionally, there may be areas of mineralization of the epithelium or stroma in the form of small nodules or amorphous masses.(2) The distinctive features of calcifying epithelial odontogenic tumors in cats and dogs are the spherical amyloidlike deposits, which may undergo concentrically laminated calcification within the epithelial islands and stroma.(7)
It is believed that APOT passes through various developmental stages in which the neoplastic epithelium degenerates, forming amyloid globules which coalesce and calcify. However, the amyloid component seen in animal odontogenic tumors has not been thoroughly examined. Whether it is a secreted substance or a degenerative product remains controversial.(8) The neoplasm is suggested to originate from oral gingival epithelium or odontogenic epithelium within the connective tissue of the gingiva or within bone.(4) The amyloid in these tumors is suggested to be a secretory product of the neoplastic cells and possibly reflects an attempt to produce enamel by neoplastic ameloblasts.(1) Hirayama and co-workers examined the immunohistochemical profile of the amyloid protein from canine APOT using antibodies to ameloblastin, sheathlin, and amelogenin.(4) The neoplastic epithelial cells of APOT were focally reactive with antibodies to ameloblastin, sheathlin, amelogenin, and canine APOT amyloid. The similarity in amino acid sequence of the amyloid protein of canine APOT to that of enamel proteins, such as ameloblastin, sheathlin, and amelogenin, and the expression of these antigens in both APOT amyloid and in the neoplastic cells suggest that the amyloid of canine APOT is derived from enamel proteins secreted by ameloblasts. Based on these findings, the authors concluded that the precursor protein of amyloid fibrils in canine APOT may be derived from enamel proteins produced by ameloblasts and proposed that canine amyloid-producing odontogenic tumor (APOT) would more properly be named as canine amyloid-producing ameloblastoma (APA).(5) If this would apply to feline amyloid-producing odontogenic tumor remains to be determined.
In animals, almost all epithelial odontogenic tumors warrant a good prognosis. None has ever been reported to metastasize. They remain localized in the mandible or the maxilla, where they cause swelling and distortion. Complete surgical excision is usually curative. There is one exception to this rule: the calcifying epithelial odontogenic tumor (amyloid-producing odontogenic tumor) in cats and dogs, which although histologically benign is usually a locally invasive neoplasm and can cause destruction of bone and displacement of teeth.(7,8) Some cases were described to recur after excision.(4) In most cases, the neoplasms are considered as low grade malignancy and rarely metastasize.(8)
JPC Diagnosis:
Conference Comment:
Additionally, in Hirayama and co-workers study of canine APOTs, the neoplastic cells exhibited positive immunoreactivity for cytokeratins (CK) AE1/AE3, CK9 and CK14. In the feline study, neoplastic cells were positive for CK AE1/AE3, CK14 and CK19. CK14 and CK19 are type I intermediate filaments of odontogenic epithelium; however, they are not specific, as they are also expressed in inner and outer enamel epithelium, cells from the stellate reticulum, stratum intermedium, dental lamina and Serres rests of the developing tooth.(3)
Overall, these results suggest that ameloblasts are the cell of origin of canine and feline APOTs, and ameloblasts produce the amyloid found in APOTs. Furthermore, feline and canine APOTs both apparently produce a similar type of amyloid which is distinct from the ODAM found in human CEOTs.(3)
References:
2. Brown CC, Baker DC, Barker IK. Alimentary tract, oral cavity, amyloid producing odontogenic tumor. In: Grant M Ed. Jubb, Kennedy and Palmers Pathology of Domestic Animals. Vol. II, 5th ed. Philadelphia, PA: Saunders Elsevier; 2007:25.
3. Delaney MA, Singh K, Murphy CL, Solomon A, Nel S, Boy SC. Immunohistochemical and biochemical evidence of ameloblastic origin of feline amyloid-producing odontogenic tumors in cats. Vet Pathol. Published online 25 June 2012. http://vet.sagepub.com/content/early/2012/06/25/0300985812452583 Accessed 23 November 2012.
4. Gardner DG, Dubielzig RR, McGee EV. The so-called calcifying epithelial odontogenic tumour in dogs and cats (amyloid-producing odontogenic tumour). J Comp Pathol. 1994;111:221230.
5. Hirayama K, Miyasho T, Ohmachi T, Watanabe T, Yokota H, Taniyama H. Biochemical and immunohistochemical characterization of the amyloid in canine amyloid-producing odontogenic tumor. Vet Pathol.2010;47(5)915-922.
6. Kuwamura M, Kanehara T, Yamate J, Shimada T, Kotani T. Amyloid-producing odontogenic tumor in a Shih-Tzu dog. J Vet Med Sci. 2000;62(6):655657.
7. Poulet FM, Valentine BA, Summers BA. A survey of epithelial odontogenic tumors and cysts in dogs and cats. Vet Pathol. 1992;29:369380.
8. Tsai YC, CR. Jeng, YX Zhuo, YC. Tsai, CH. Liu, VF Pang. Amyloid-producing odontogenic tumor and its immunohistochemical characterization in a Shih Tzu dog. Vet Pathol. 2007;44:233236.