Signalment:
Flock of 20,000 11-week-old commercial meat turkeys,
Meleagris
gallopavo.Flock experienced a spike in mortality.
Flock is housed in barn and bedded with a layer of shavings on top of dirt
floors. This is the first time this disease has been identified on this farm but
the producer does have a second farm where turkeys are also raised and this
disease has been a recurring problem on that farm.
Gross Description:
The submitting vet-erinarian described
swollen livers with yellow streaking and very enlarged dark spleens in the
turkeys that were necropsied. Birds also had unclotted blood in the abdominal
cavity.
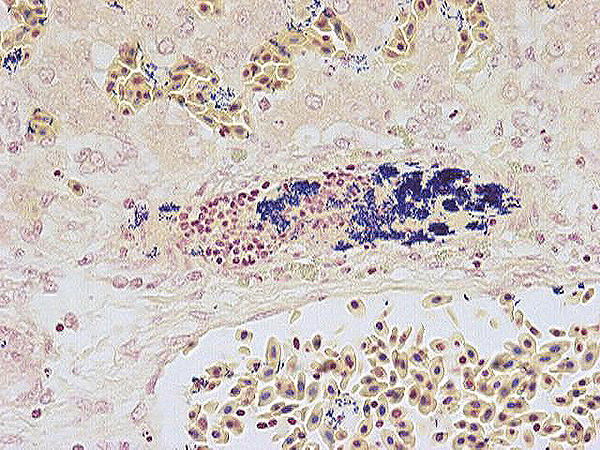
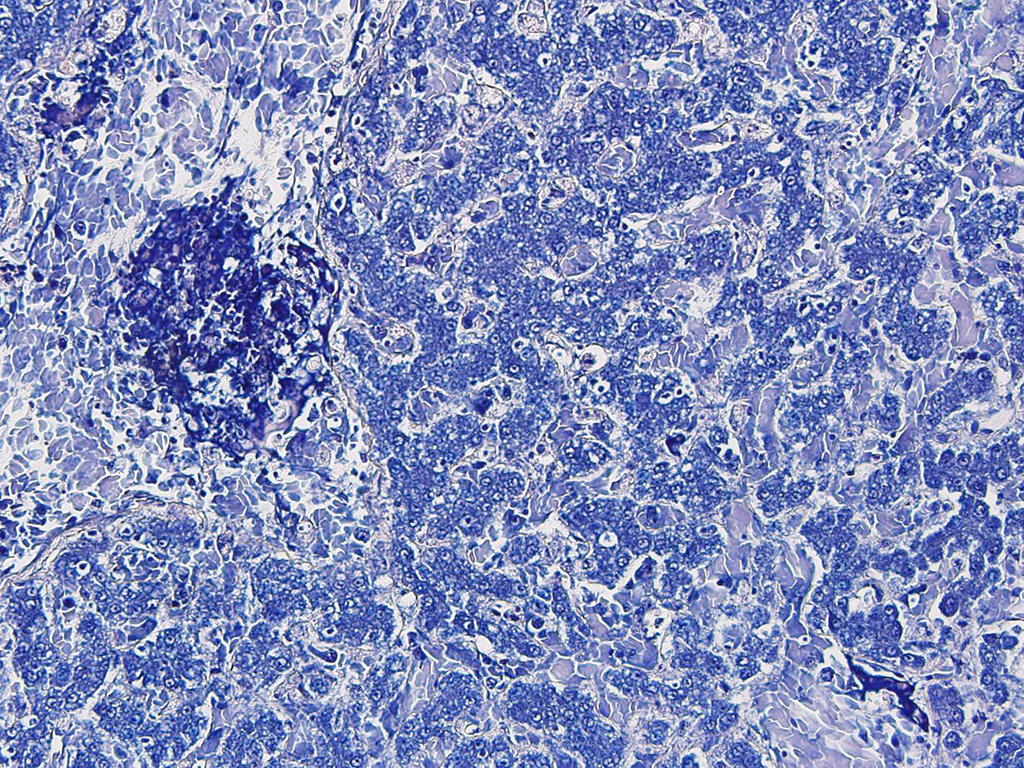
Histopathologic Description:
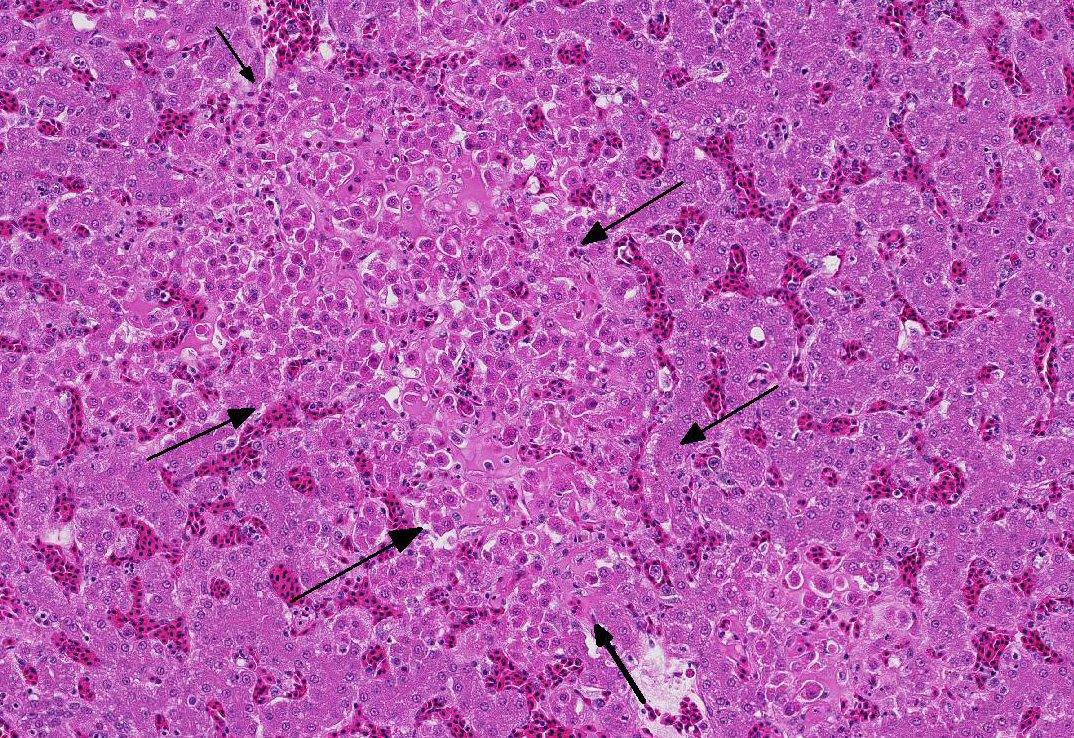
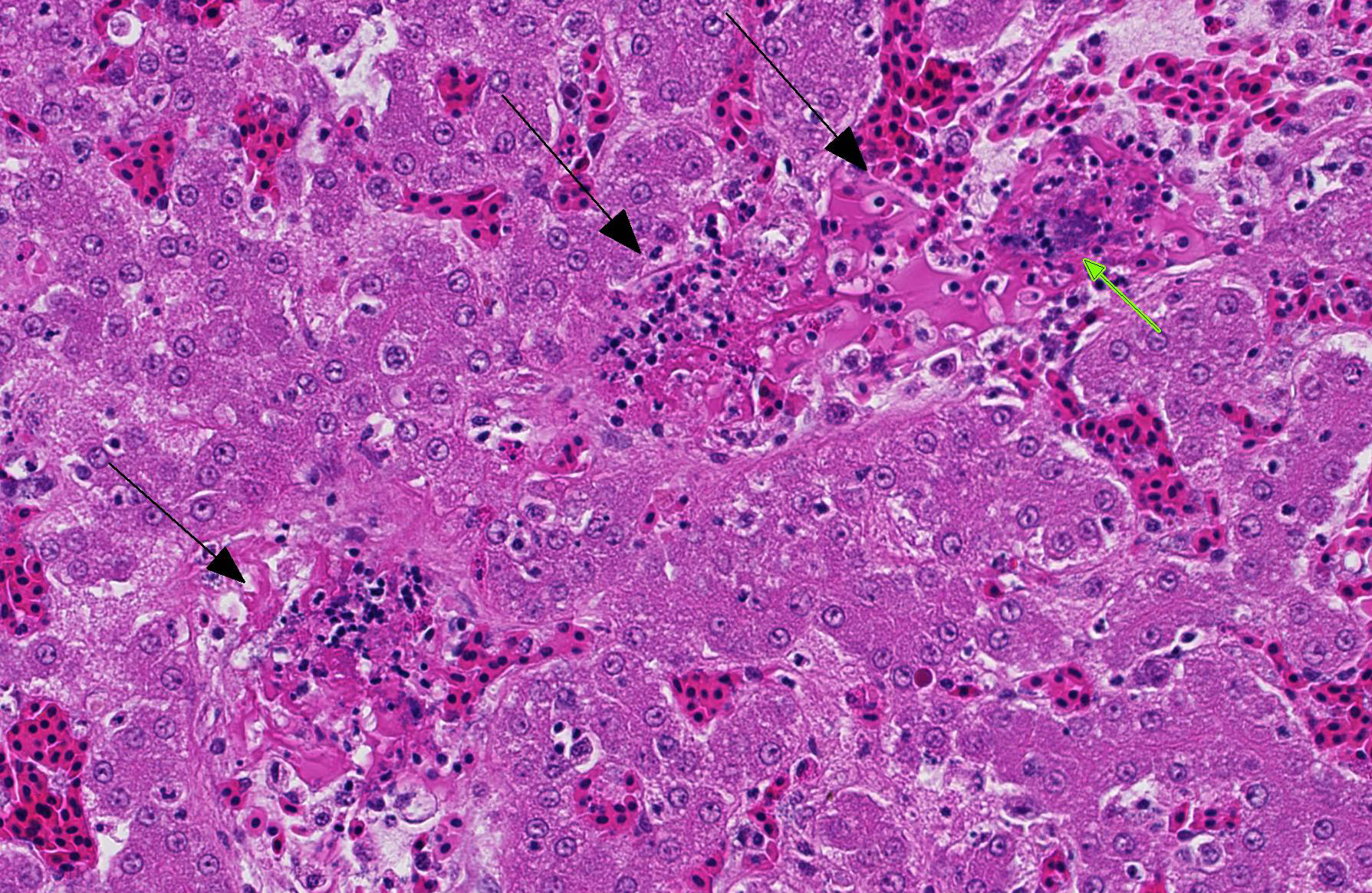
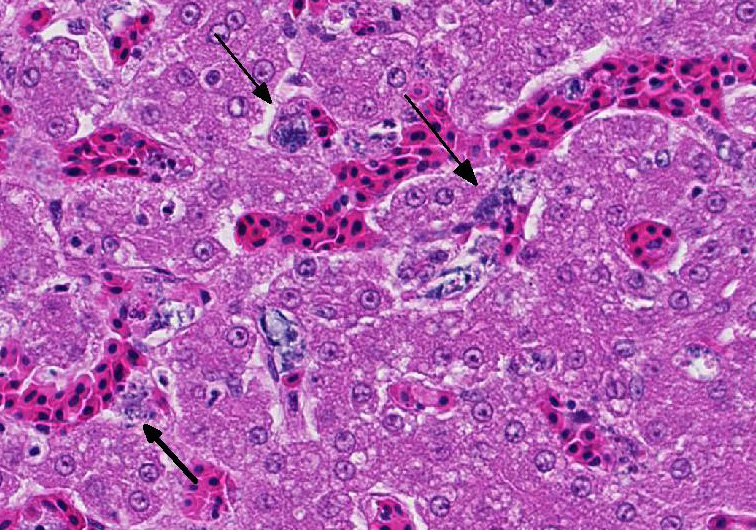
The liver is moderately congested. Multiple variably-sized foci of
hepatic necrosis, characterized by individualized, hy-pereosinophilic
hepatocytes with granular or shrunken, hyalinized cytoplasm and either lacking
nuclei or containing pyknotic or karyorhectic nuclei, are closely associated
with terminal hepatic veins. Hepatocytes surrounding these necrotic foci are
occasionally swollen with pale vacuolated cytoplasm. Multifocally, veins and
sinusoids and less frequently small arteries contain fibrin thrombi bearing mats of slender rod-shaped and sometimes
gently curved bacteria. Similar appearing bacteria are also free in sinusoids
and within activated Kupffer cells, which also contain pha-gocytized debris
including red blood cells. Occasionally the walls of veins and small arteries
containing fibrin thrombi have segmental necrosis, with some affected arteries
having intramural heterophils and rarely small amounts of nuclear debris.
Morphologic Diagnosis:
Liver: Mild, acute, multifocal, hepatic necrosis with necrotizing
vasculitis and intravascular fibrin thrombi containing colonies of pleomorphic
rod-shaped bacteria.
Liver: Moderate hepatic congestion
Lab Results:
4+
Erysipelothrix rhusiopathiae
was recovered from a swab of the internal organs.
Condition:
Erysipelothrix rhusiopathae
Contributor Comment:
Erysipelas
is an acute septicemic disease occurring most commonly in older male turkeys. The
differential diagnosis includes other gram-negative bacterial septicemias
caused by agents such as
E. coli,
Salmonella spp. or
Pasteurella
multocida.2 Histologically
, the pathology of an
Erysipelothrix
rh-usiopathiae infection is different from most gram-positive agents, first
because of the sheer numbers of bacteria present and secondly because of their
variable appearance, with slender rod-shaped to slightly curved bacteria
aggregating in mats within vessel and capillary lumens and entangled in
fibrinous thrombi. Because these bacteria are slow growing, a rapid
presumptive diagnosis can also be made by identification of clumps of
gram-positive slender straight or slightly curved rod-shaped bacteria from
organ or bone marrow smears.
2 This case was
submitted to the lab in early October which is typical for cases of erysipelas,
as outbreaks are reported to occur most often in the late fall or winter. It is
thought that the bacteria can persist in the soil and since many grow-out barns
for turkeys have dirt floors, the risk of repeat occurrences exists.
2
In this case, this farm has never experienced an outbreak of erysipelas but the
other farm has and it is suspected that there was mechanical transfer of the
bacterium from one farm to another. Penicillin is the
antibiotic of choice for treating erysipelas. Vaccination using a killed
bacterin is an option if the risk of infection is high.
2 In humans, the
infection caused by
Erysipelothrix rhusiopathiae is known as
erysipeloid, a skin infection typically localized to fingers and hands and
usually preceded by an abrasion or cut. The lesion is actually a cellulitis and
is very painful. Systemic effects, such as septicemia and endocarditis can
occur but are uncommon.
6 Most cases of human infection are the
result of occupational exposure and those occupations at higher risk include
fish handlers, veterinarians, farmers, slaughter plant workers and butchers.
Some colloquial names for this condition include fish handler's disease, seal
finger and whale finger.
JPC Diagnosis:
Liver: Hepatitis, nec-rotizing, acute, multifocal, random, with septic fibrin thrombi
and vasculitis.
Conference Comment:
In addition to outbreaks in domestic turkeys,
E. rhusiopathiae outbreaks
have also been reported in laying hens in Europe,
3 and sporadically
in a variety of other captive and free-ranging birds. The organism is fas-tidious
and able to survive in the environment for extended periods. It may be
transmitted by cuts and abrasions or through ingestion. It is generally
considered to follow an acute course characterized by septicemia, but a chronic
form also occurs in turkeys,
1 which appear to be most susceptible to
infection. In addition to thromboembolism, bacterial endocarditis and joint
infections
1 may be seen in affected turkeys, among other signs of
septicemia. Thrombosis and hemorrhage are commonly reported in avian species
infected with
E. rhusiopathiae, reflecting the vasculocentric nature of
the disease. Grossly, carcasses of affected birds are in good flesh and
exhibit organomegaly of the liver, spleen and kidneys, as well as eccymotic
hemorrhages in the subcutis and muscles.
1 Routes of infection
include fomites, contaminated soil, insect vectors, asymptomatic carrier animals
and contaminated feed.
3,4 Although
it has been reported, infection in psittacine birds is considered rare. In a
case report of infection in a mixed species aviary, lesions included
thrombosis, bacterial th-romboembolism, necrotizing hepatitis, necrohemorrhagic
myocarditis, and hem-orrhage.
4 E. rhusiopathiae infection
has also been reported in emus, which are large flightless birds that are
grouped with other ratites such as ostriches and rheas. Lesions similar to
those reported in other species are also seen in emus, including hepatocellular
necrosis with absence of an abrupt inflammatory response. Bacteria may be
observed in multiple organs, including the kidneys and small intestine as well
as the liver; the presence of fibrin thrombi, while prominent in many cases,
may be variable.
5
References:
1. Bobrek K, Gawel A, Mazurkiewicz M. Infections with Erysipelothrix
rhusiopathiae in poultry flocks. World's Poultry Science Journal.
2013; 69(4):803-812.
2. Bricker JM, Saif YM. Erysipelas. In: Saif YM, ed. Diseases of Poultry. 12th
ed. Ames, IA: Blackwell Publishing; 2008:909-922.
3. Eriksson H, Bagge E, Båverud V, Fellstrom C, et al. Erysipelothrox
rhusiopathiae contamination in the poultry house environment during
erysipelas outbreaks in organic laying hen flocks. Avian Pathol. 2014;
43(3):231-237.
4. Galindo-Cardiel I, Opriessnig T, Molina L, Juan-Salles C. Outbreak of
mortality in psittacine birds in a mixed-species aviary associated with Erysipelothrix
rhusiopathiae infection. Vet Pathol. 2012; 49(3):498-502.
5. Morgan MJ, Britt JO, Cockrill JM, Eiten ML. Erysipelothrix rhusiopathiae
infection in an emu (Dromaius novaehollandiae). J Vet Diagn Invest.
1994; 6:378-379.
6. Reboli, A, Farrar WE. Erysipelothrix rhusiopathiae: An occupational
pathogen. Clin Microbiol Rev. 1989; 2:354-359.