Signalment:
2-month-old, filly, Quarter Horse, Equine,
Equus caballusFilly had body temperature of 103F and harsh lung sounds. Ultrasound examination showed "comet tails" on pleural surface. Animal was treated using azithromycin and banamine. Animal improved slightly and naxcel was included in the treatment. Despite the treatment animal was found dead in the stall. Prior to this episode, filly was given two injections of hyperimmune plasma to prevent
Rhodococcus equi infection.
Gross Description:
A 2-month-old quarter horse filly was submitted for necropsy. The foal was in good body condition with adequate depot fat stores present. Hydration appeared adequate. The lungs were diffusely reddened, firm, and sank in formalin. There was a single 7 cm nodular area of caseation within the right cranioventral lung lobe. The tracheobronchial lymph nodes were markedly enlarged and contained a thick creamy exudate. The distal third of the trachea was hemorrhagic and contained linear streaks of ulceration/erosion. The omentum and mesentery were hemorrhagic. There was a large bilobed abscess, approximately 15 cm in diameter, within the mesentery by the ceco-colic junction. The center of the abscess was filled with pasty white necrotic material. A similar abscess was present within the mesentery adjacent to the jejunum. This abscess was adhered to the wall of the jejunum, and the overlying mucosa was focally ulcerated. There was a focal irregular area of hyperkeratosis in the nonglandular portion of the stomach next to the margo plicatus. There was an area of subcutaneous hemorrhage in the dorsal lumbar area.
Histopathologic Description:
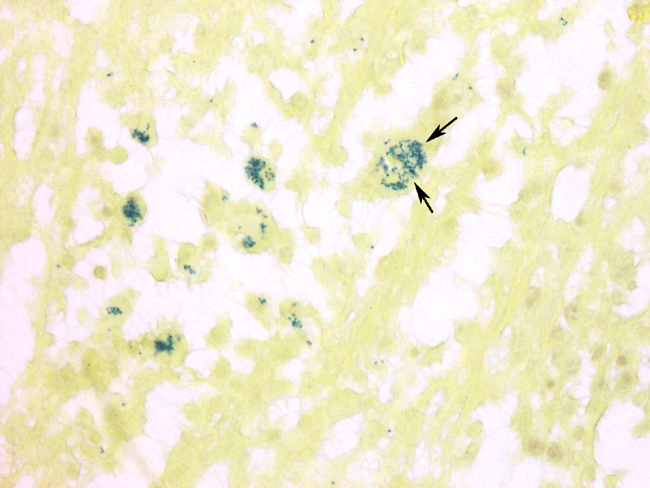
A section of consolidated lung was examined, and is characterized by filling of alveoli with fibrin, macrophages, and occasional neutrophils. Occasional type II cell hyperplasia is present. Scattered alveoli contain multinucleated giant cells. Small numbers of plasma cells are present within some alveoli and within the thickened alveolar septae. The alveolar exudate occasionally is necrotic. Bronchioles and bronchi are generally devoid of inflammatory cells. A section of lung from the right cranioventral lung lobe contains large areas of abscessation. These foci contain sheets of degenerating neutrophils admixed with fewer macrophages and replace normal pulmonary parenchyma. Many of the macrophages contain intracytoplasmic bacteria consistent with
Rhodococcus. There is mild fibroplasia around these areas, and the adjacent alveoli contain multinucleated giant cells, macrophages, lymphocytes, and plasma cells. There is acute hemorrhage within the deep lamina propria of the trachea. There is diffuse congestion, and the overlying mucosa is focally ulcerated. Ulcerated foci are covered with degenerated neutrophils and fibrin, and the base is infiltrated with neutrophils, macrophages, and occasional multinucleated giant cells.
Morphologic Diagnosis:
1. Severe diffuse histiocytic bronchointerstitial pneumonia with focal abscessation with intracellular bacteria
2. Acute ulcerative tracheitis with hemorrhage and pyogranulomatous inflammation
Lab Results:
1. Bacteriology: Lung abscess, lymph node swab yielded
Rhodococcus equi
2. Fluorescent Antibody Tests: Negative for Adenovirus, EHV 1 & EIV
3. Virus isolation from lung: Negative
Condition:
Rhodococcus equi
Contributor Comment:
The pulmonary lesions present in the caudoventral portion of the lungs were typical in distribution and gross and microscopic appearance to lesions caused by
Rhodococcus equi. The mesenteric lymph node involvement is also common in this disease. The diffuse inflammatory changes present in the rest of the lung actually predominated in this case, and have not been typically associated with this disease. These changes have been described in a group of foals with
Rhodococcus infection
3. An underlying viral etiology was suspected in these foals, but attempts to demonstrate a viral component were generally unrewarding. It may be that the bacteria are inducing a hypersensitivity type response in areas of the lung not colonized by bacteria. The reason this response occurs in particular groups of animals is unknown.
Twenty-three foals, between 1 and 7 months old, with signs of acute respiratory distress, were examined at the Veterinary Medical Teaching Hospital (VMTH), University of California, Davis, between 1984 and 1989. Characteristic features included sudden onset of severe respiratory distress and tachypnea, cyanosis unresponsive to nasal oxygen, pyrexia, hypoxemia, hypercapneic respiratory acidosis, poor response to treatment, and histopathologic lesions of bronchiolitis and bronchointerstitial pneumonia. Seven of the 23 foals were normal before the onset of respiratory distress, 3 foals were found dead, and 13 foals were being treated for respiratory tract infections at the time of presentation. Laboratory data obtained for 13 horses showed increased plasma fibrinogen concentration (630.7 +/- 193 mg/dL), leukocytosis (18,607 +/- 7,784/microL), and neutrophilia (13,737 +/- 8,211/microL). Thoracic radiographs showed a diffuse increase in interstitial and bronchointerstitial pulmonary opacity and, in 5 foals, an alveolar pulmonary pattern of increased density was also seen. In 3 foals heavy interstitial infiltration proceeded to a coalescing nodular radiographic appearance. Microbiological culture of tracheobronchial aspirates (TBA) from 9 foals yielded bacterial growth, but no one bacterial species was consistently isolated. Microbiological culture of postmortem specimens of the lung from 6 foals yielded growth of bacteria that included Escherichia coli, Enterobacter spp., Proteus mirabilis, Klebsiella pneumoniae, Rhodococcus equi, or beta-hemolytic Streptococcus spp. Tracheobronchial aspirates from 4 foals and lung samples collected from a further 4 foals at necropsy yielded no bacterial growth. Cultures were not taken from two foals premortem or postmortem. Virologic examination of TBA, lung tissue, or pooled organ tissue from 12 foals was negative. Viral culture of TBA from 1 foal showed cytopathic effects and positive immunofluorescence for equine herpes virus type II (EHV-II). In addition to the 3 foals that were found dead, 11 foals died or were euthanatized. Pathologic lesions were limited to the lungs in 50% of the foals; the remainder also had bowel lesions suggestive of hypoxic injury. The predominant histopathologic pulmonary lesions included bronchiolitis, bronchiolar and alveolar epithelial hyperplasia, and necrosis. Many bronchioles were filled with mucoid and fibrinocellular exudate. The peribronchiolar interstitium and adjacent alveolar spaces were also infiltrated with inflammatory cells and contained proteinaceous edema fluid. Type II cell hyperplasia and hyaline membrane formation were observed in the majority of foals and in 2 foals alveolar multinucleate giant cells were also present
3.
Later, another foal from the same farm was submitted for necropsy. The second foal had similar gross and histopathologic lesions indicating an endemic infection
2.
JPC Diagnosis:
1. Lung: Pneumonia, interstitial, necrotizing, histiocytic,
lymphoplasmacytic, and neutrophilic, diffuse, marked, with fibrin and hyaline
membranes, Quarter horse (
Equus caballus).
2. Lung: Pneumonia, pyogranulomatous, focally extensive, severe, with intrahistiocytic
coccobacilli.
Conference Comment:
Rhodococcus equi is a facultative, intracellular, Gram-positive
bacteria that is present in soil and feces and is often enzootic on farms.
4 Two classic
forms of the disease are suppurative to pyogranulomatous bronchopneumonia and
ulcerative enterocolitis. Approximately half of the foals affected with the respiratory form
have concurrent intestinal lesions. Intestinal lesions without the respiratory form is not
common.
4 The lymph nodes, joints, bones, genital tract, and other organs may also be
involved.
4 There are sporadic reports in other species, including cattle, goats, pigs,
dogs, cats, and immunocompromised humans.
Rhodococcus equi appears to be easily killed by neutrophils but not macrophages.
Upon entry through either inhalation or ingestion the bacteria are phagocytosed by
either alveolar or intestinal macrophages. Several proposed virulence factors encoded
by plasmids allow survival within macrophages. Vap A, Vap B, and Vap C, as well as
glycolipids, capsular polysaccharides, and equi factors (cholesterol oxidase and
choline phosphohydrolase) contribute to the virulence of certain
Rhodococcus equi
strains.
1 They prevent lysosomal fusion and/or result in premature lysosomal
degranulation, survival of the bacteria, and death of the macrophage.
4
Diffuse interstitial pneumonia is not a classic lesion of
Rhodococcus equi pneumonia
and is likely due to a separate disease process. The findings of necrotizing interstitial
pneumonia with hyaline membrane formation are suggestive of the acute phase of
diffuse alveolar damage (DAD). DAD results from diffuse injury to type I pneumocytes
with subsequent hyaline membranes formation, type II pneumocyte proliferation and
interstitial fibrosis.
3 These histologic lesions are non-specific, and identification of an
etiologic agent is often difficult. Causes of DAD include, but are not limited to, thermal
injury, toxic gases, septicemia, ingested toxins (paraquat, kerosene, Brassica, and
perilla mint), endotoxemia, acute hypersensitivity reactions, ventilator-induced injury,
and chronic left heart failure.
3
References:
1. Brown CC, Baker DC, Barker IK: Alimentary system. In: Jubb, Kennedy, and
Palmers Pathology of Domestic Animals, ed. Maxie MG, 5th ed., vol. 2, pp. 226.
Elsevier Limited, St. Louis, MO, 2007
2. Chaffin MK, Cohen ND, Martens RJ, Edwards RF, Nevill M. Foal-related risk factors associated with development of
Rhodococcus equi pneumonia on farms with endemic infection. J Am Vet Med Assoc. 2003;223(12):1791-1799.Â
3. Lakritz J, Wilson WD, Berry CR, Schrenzel MD, Carlson GP, Madigan JE.
Bronchointerstitial pneumonia and respiratory distress in young horses: clinical, clinicopathologic, radiographic, and pathological findings in 23 cases (1984-1989). J Vet Intern Med. 1993;7(5):277-288.
4. L³pez A: Respiratory system. In: Pathologic Basis of Veterinary Disease, ed.
McGavin MD, Zachary JF, 4th ed, pp.519-520. Mosby, Inc., St. Louis, MO, 2007