Signalment:
24 month old, male, Fischer F344 rat (Rattus norvegicus).
An aged male F344 rat enrolled in a behavior study underwent surgery for the placement of head prosthesis. The animal recovered uneventfully, but 4 weeks fol-lowing the surgery, a small swelling on the right side of the face was noticed. The swelling increased in size over several days. An aspirate of the mass showed eosinophilic acellular linear material (keratin), non-degenerative neutrophils, histiocytes, ly-mphocytes, and amorphous debris. The rat was anesthetized and the swelling was lanced and flushed, producing copious amounts of purulent and sebaceous discharge. The rat was treated with oral antibiotic therapy (enrofloxacin ad-ministered in the drinking water); however, facial swelling was still present and animals condition deteriorated. Humane euthanasia via intraperitoneal barbiturate overdose was elected by the investigative group.
Gross Description:
There was an ap-proximately 1 cm diameter, firm swelling at the base of the right ear. There was focal ulceration on the caudoventral aspect of the swelling with adhered bedding material. Both kidneys were diffusely, mildly pale, shrunken and irregular in texture, with pinpoint depressions across the surface. The spleen was approximately twice the normal size with an irregular texture. The liver was diffusely moderately enlarged and irregular. Both testicles were approximately 1.5 times larger than normal and yellow to tan.
Histopathologic Description:
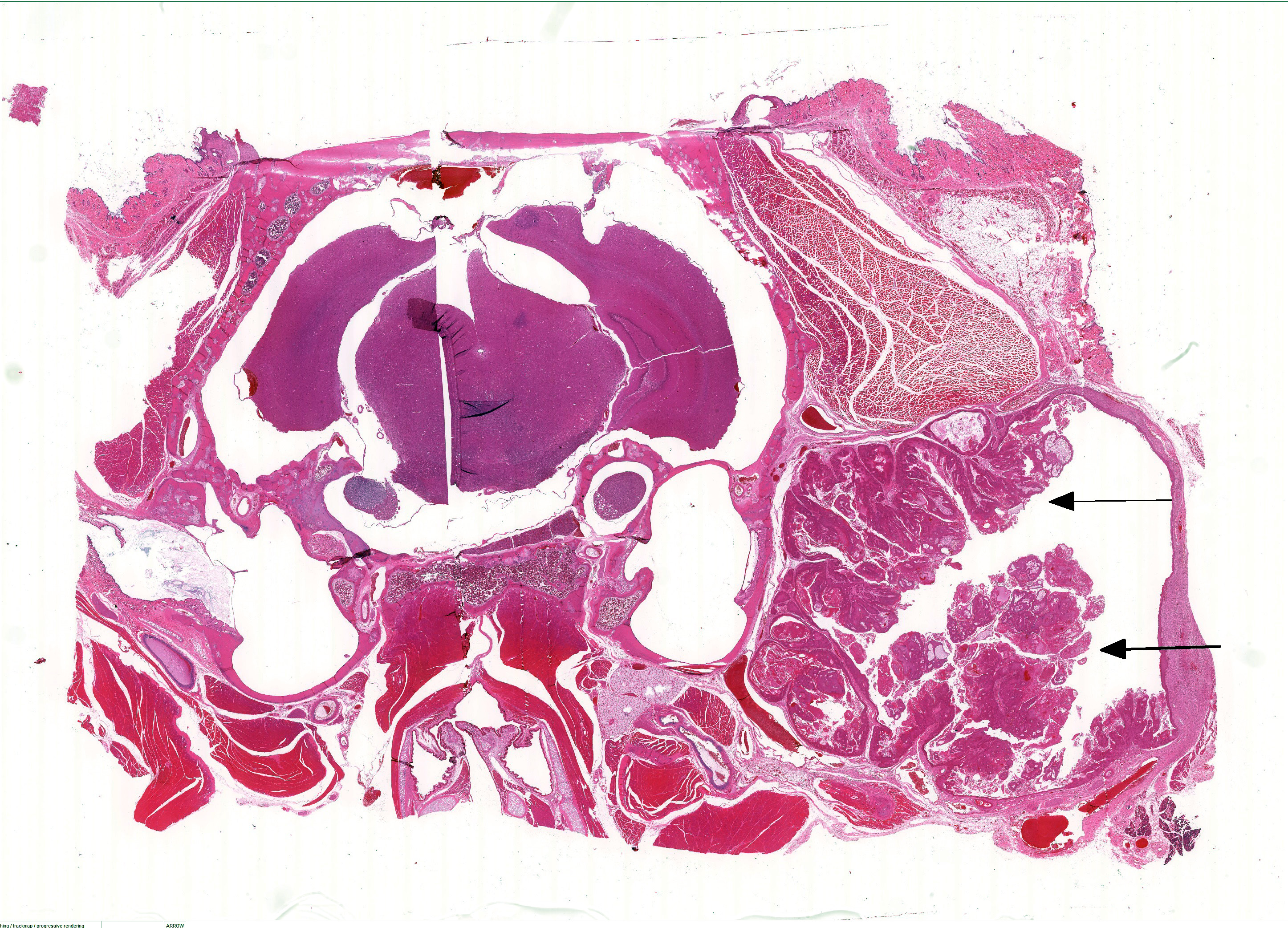
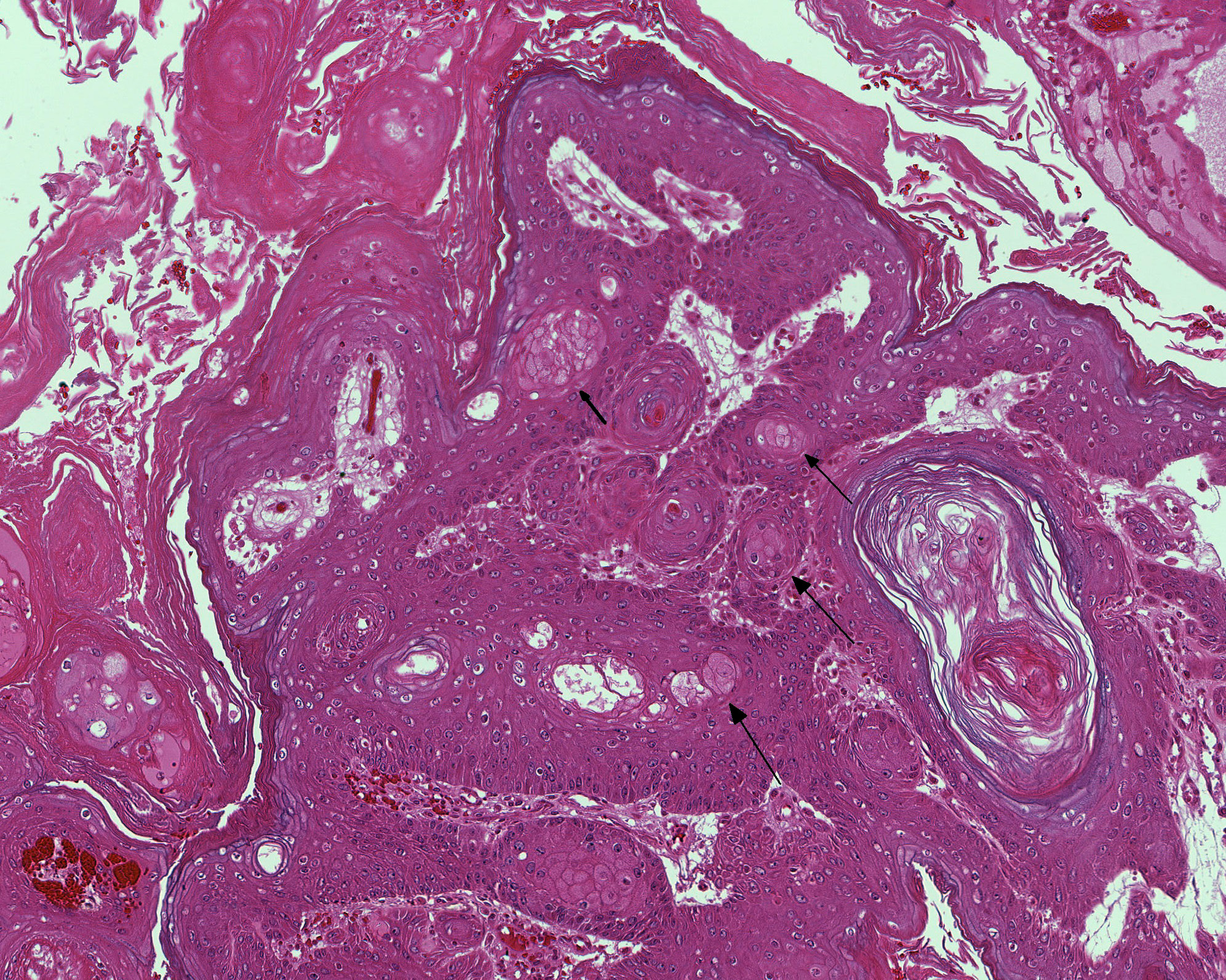
Extending through the subcutis from the lateral aspect of the bulla is a large, well demarcated, non-invasive, mul-tilobulated and nonencapsulated neoplasm consisting of papillary projections of stratified sq-uamous epithelium supported by expanded fibrovascular cores. The basement membrane is intact. There is diffuse, marked hyperkeratosis with abundant strands of eosinophilic acellular material (keratin) present within the center of the neoplasm. There are multifocal circular areas of lamellated keratin (keratin pearls). Neoplastic cells are well differentiated squamous epithelial cells with variably well delineated cytoplasmic borders and moderate eosinophilic cytoplasm. An ovoid central nucleus with a single nucleolus is present. There is mild to moderate anisocytosis and anisokaryosis. Mitotic figures are not observed. There is mild, multifocal in-tercellular edema with accentuation of intercellular desmosomes. Multifocal cystic cavities are also present. Sebaceous cells are moderately to markedly enlarged and increased in number, with abundant pale and vacuolated cytoplasm. Extending along the lateral aspect of the neoplasm is a thick layer of mature fibrous connective tissue with mild multifocal extravascular red blood cells (hemorrhage).
Morphologic Diagnosis:
Zymbals gland: Squamous cell papilloma with sebaceous hyperplasia and cystic degeneration.
Lab Results:
Condition:
Contributor Comment:
In this case, no history of toxin exposure was present.Despite the large size, a benign tumor was diagnosed given the lack of mitoses and cellular atypia as well as the lack of invasion into the surrounding muscle and subcutis. The tumor was characterized as a papilloma given the exophytic mul-tilobulated growth with prominent fib-rovascular core.Marked sebaceous hy-perplasia was present, which is reported to be common in cases of squamous cell papilloma of the Zymbals gland.5
JPC Diagnosis:
Zymbals gland:- Sq-uamous papilloma with multifocal seb-aceous differentiation.
Conference Comment:
Squamous papillomas of the Zymbals gland originate from the duct epithelium, are composed of stratified squamous epithelium and must be differentiated from squamous cell carcinoma based on absence of invasive growth and cellular atypia.Inflammatory, atrophic and hyperplastic changes are also described in the Zymbals gland, although their frequency of occurrence varies.The retention of normal tissue architecture aids in differentiating hyperplasia from adenoma. Adenomas will also generally have in-creased numbers of basaloid cells compared to hyperplasia, are lobulated, and well defined but not encapsulated.Many of the neoplastic cells- demonstrate cytoplasmic vacuolation, akin to mature sebaceous gland cells.5- In this case there was considerable discussion during the conference regarding the presence of vacuolated sebaceous cells in this neoplasm; some participants argued the cells which appear vacuolated may actually represent degenerating squamous epithelium. Regardless, glandular pro-liferation is often associated with squamous papilloma of the Zymbals gland and thus, if present in this case, would not be surprising and would not change the diagnosis.Zymbals gland carcinoma is also described and includes such features as pleomorphism, increased mitotic index, squamous dif-ferentiation, ulceration and absence of ducts.5
In addition to the mammary gland, the Zymbals gland is commonly assessed in xenobiotic studies in rodents since it is a target organ of many carcinogens.Aside from neoplastic changes, degenerative, necrotic, regenerative and vascular changes are also described in the gland and many of the changes are similar to what is observed in the mammary gland.5 Additional tissues which may be sampled in xenobiotic studies include the preputial and clitoral glands, which are also common sites for adenomas in rodents.Degeneration, necrosis and regeneration are often seen in the same gland with repeated toxin exposure.Dilation or cystic degeneration of ducts may also be seen in the Zymbals gland as a spontaneous aging change in rodents.5 Common tumor types in both the F344 and Sprague Dawley rat strains (from the National Toxicology Program historical control database) include mammary gland fibroadenoma, pituitary pars distalis adenoma, thyroid gland C cell adenoma, adrenal medulla benign pheochromocytoma, and uterine stromal polyp.1,2 Mammary gland carcinoma1,2 and histiocytic sarcoma are also common tumors seen in Sprague Dawley rats, and large granular lymphocytic leukemia, interstitial cell tumors in males,1,2 and mesothelioma originating from the testicular tunic are common in F344 rats.
References:
1. Dinse GE, Peddada SD, Harris SF, Elmore SA.Comparison of NTP historical control tumor incidence rates in female Harlan Sprague Dawley and Fischer 344/N rats.Toxicol Pathol. 2010; 38:765-775.
2. Haseman JK, Hailey JR, Morris RW.Spontaneous neoplasm incidences in Fischer 344 rats and B6C3F1 mice in two-year carcinogenicity studies: A National Toxicology program update. Toxicol Pathol. 1998; 26(3):428-441.
3.Hafez E, Takahashi T, Ogawa H, Sato M, Nakai T, Takasu C, et al.: High susceptibility to zymbal gland and intestinal carcinogenesis in diabetic Otsuka long-evans Tokushima Fatty rats. J Toxicol Pathol 2011:24(4):187-193.
4.Haseman JK, Lockhart AM: Correlations between chemically related site-specific carcinogenic effects in long-term studies in rats and mice. Environ Health Perspect 1993:101(1):50-54.
5.Rudmann D, Cardiff R, Chouinard L, Goodman D, Kuttler K, Marxfeld H, et al.: Proliferative and nonproliferative lesions of the rat and mouse mammary, Zymbal's, preputial, and clitoral glands. Toxicol Pathol 2012:40(6 Suppl):7S-39S.
6.Seely JC: Auditory Sebaceous Glands (Zymbal's Glands). In: Mohr U, Dungworth DL, Capen CC, eds. Pathobiology of the Aging Rat. Washington, D.C.: ILSI Press; 1994: 423-433.
7.Sundberg JP, Nanney LB, Fleckman P, and King LE: Skin and Adnexa. In: Treuting PM and Dintzis SM, eds. Comparative Anatomy and Histology: A mouse and human atlas. San Diego, CA: Academic Press; 2012: 433-453.