Signalment:
This animal was part of a research project conducted under an IACUC approved protocol in compliance with the Animal Welfare Act, PHS Policy, and other federal statutes and regulations relating to animals and experiments involving animals. The facility where this research was conducted is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care, International and adheres to principles stated in the Guide for the Care and Use of Laboratory Animals, National Research Council, 2011.
Gross Description:
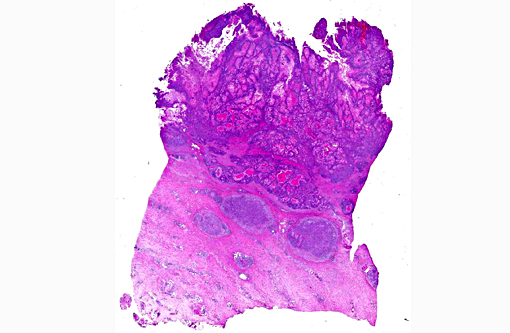
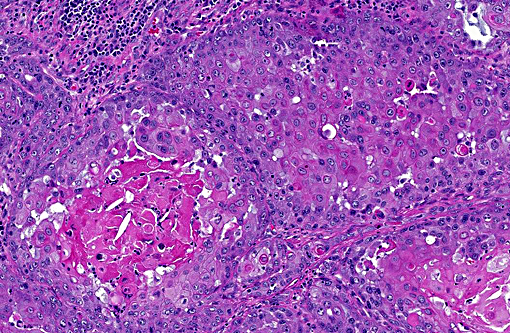
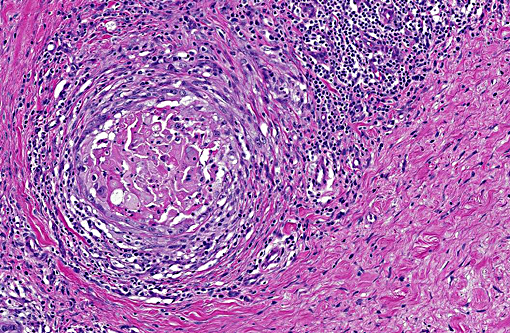
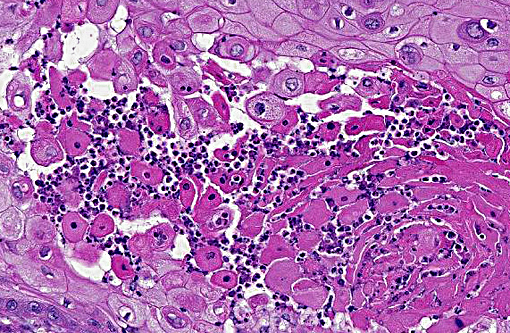
Histopathologic Description:
Morphologic Diagnosis:
Condition:
Contributor Comment:
There are numerous recent reports that reveal increasing evidence to support a potential causal relationship between equine papillomavirus type 2 (EcPV2) and equine penile squamous cell carcinoma (SCC).(1,4,5,8) Studies have suggested that equine penile papillomas, in situ carcinomas, and invasive carcinomas belong to a continuum of papilloma-induced disease, which is supported by detection of EcPV2 DNA in horses with characteristic SCC lesions, and in various lesions of the penis in a proportional number of control cases.(6) When comparing the levels of EcPV2 viral load in equine SCC lesions, penile non-SCC, or precursor disease lesions, and tissues without observable lesions from SCC-prone sites on clinically normal horses, EcPV2 DNA was present significantly more often, and in higher copy numbers, in the equine penile SCC lesions than the others. The presence of EcPV2 DNA has also been demonstrated in anal lesions, a lymph node, and contact metastases.(1) Additionally, in one report, viral mRNA was detected in all examined EcPV2 DNA positive lesions, while only 2.6% of specimens from healthy horses had detectable mRNA.(7) This finding provides evidence of intralesional viral transcriptional activity which further supports an active role of the virus in equine SCC disease.(9)
Note: Opinions, interpretations, conclusions, and recommendations are those of the authors and are not necessarily endorsed by the U.S. Army.
JPC Diagnosis:
Conference Comment:
In addition to horses, squamous cell carcinomas of the eyelid epithelium occur in other species, most commonly cattle, cats, and dogs. Squamous cell carcinomas develop through a stepwise process, often consisting of precancerous stages and the formation of a papilloma. Tumor development begins with initiation, which is an irreversible genetic change typically caused by solar radiation. The next step is promotion, which includes the growth of genetically altered or initiated cells within a favorable environment. Promotion is followed by progression, which results in increasing malignancy of the developing tumor and involves both genetic and epigenetic changes to tumor cells.(7) Ultraviolet radiation induced squamous cell carcinoma of the eyelid occurs most commonly in Hereford cattle which have nonpigmented eyelids.(7) The ultraviolet radiation induces DNA damage and subsequent mutation; of the three wavelengths of ultraviolet light, UVB is thought to be most associated with development of cutaneous neoplasms. The energy in UV light absorbed is by DNA and results in covalent crosslinking of pyrimidine bases and the formation of pyrimidine dimers, preventing proper base pairing. It is postulated that nucleotide excision repair mechanisms are overwhelmed with continued UV exposure, which may result in the propagation of cells with genomic mutations.(6)
As mentioned by the contributor, recent evidence supports an association between equine penile squamous cell carcinoma and equine papillomavirus 2 (EcPV2). The expression of two proteins, E6 and E7, has been demonstrated in the human papillomavirus associated with cervical cancer, which interact with proteins associated with cell cycle regulation.(10) Zhu et al. demonstrated that a subset of equine penile squamous cell carcinomas contained the E6/E7 nucleic acid of EcPV2, and a majority of neoplastic cells contained virus, providing additional evidence for the role of EcPV2 in penile and preputial SCCs in horses. Additionally, in that study the E6/E7 oncogenes of EcPV2 were present in metastatic SCCs. There was also evidence of solar damage in cases of penile and preputial SCC not associated with EcPV2.(10)
References:
1. Bogaert L, Willemsen A, Vanderstraeten E, et al. EcPV2 DNA in equine genital squamous cell carcinomas and normal genital mucosa. Vet Microbiol. 2012;158:33-41.
2. Brinsko SP, Blanchard TL, Varner DD. Male Reproductive Disorders. In: Smith BP, ed., Large Animal Internal Medicine, 4th ed. St. Louis, MO:Mosby Elsevier; 2009;1475-1476.
3. Foster RA, Ladds PW. Neoplasms of the penis and prepuce. In: Maxie MG, ed., Jubb, Kennedy and Palmers Pathology of Domestic Animals, Vol 3. 5th ed. Philadelphia, PA:WB Saunders Co; 2007:617-619.
4. Knight C, Dunowska M, Munday JS, Peters-Kennedy J, Rosa BV. Comparison of the Levels of Equus caballus papillomavirus type 2 (EcPV-2) DNA in equine squamous cell carcinomas and non-cancerous tissues using quantitative PCR. Vet Microbiol. 2013;166:257-262.
5. Knight CG, Munday JS, Peters J, Dunowska M. Equine penile squamous cell carcinomas are associated with the presence of equine papillomavirus type 2 DNA Sequences. Vet Pathol. 2011;48:1190-4.
6. Kumar V, Abbas AK, Aster JC. Pathologic basis of disease. 9th ed. Philadelphia, PA: Elsevier Saunders, 2015: 324-325.
7. Kusewitt DF. Neoplasia and Tumor Biology. In: McGavin MD, Zachary JF, eds. Pathologic Basis of Veterinary Disease. 5th ed. St. Louis, MO: Mosby Elsevier; 2012:298-299, 315.
8. Lange CE, Tobler K, Lehner A, et al. EcPV2 DNA in equine papillomas and in situ and invasive squamous cell carcinomas supports papillomavirus etiology. Vet Pathol. 2012;50:686-692.
9. Sykora S, Samek L, Schonthaler K, et al. EcPV-2 is transcriptionally active in equine SCC but only rarely detectable in swabs and semen from healthy horses. Vet Microbiol. 2012;158:194-198.
10. Zhu KW, Affolter VK, Gaynor AM, Dela Cruz FN, Pesavento PA. Equine genital squamous cell carcinoma: In situ hybridization identifies a distinct subset containing Equus caballus papillomavirus 2. Vet Pathol. Online first May 12, 2015:1-6.