CASE 2: 1235813-009 (4135955-00)
Signalment: 5.5-month-old, intact male, Beagle (Canis familiaris), canine.
History: This purpose-bred research dog was found laterally recumbent, pale, tachycardic, and dyspneic with blood coming from the mouth during a routine room check. A small amount of vomitus was noted under the cage. Humane euthanasia was elected after veterinary consultation. The dog had arrived at the facility two weeks prior to presentation and had not yet been placed on a study.
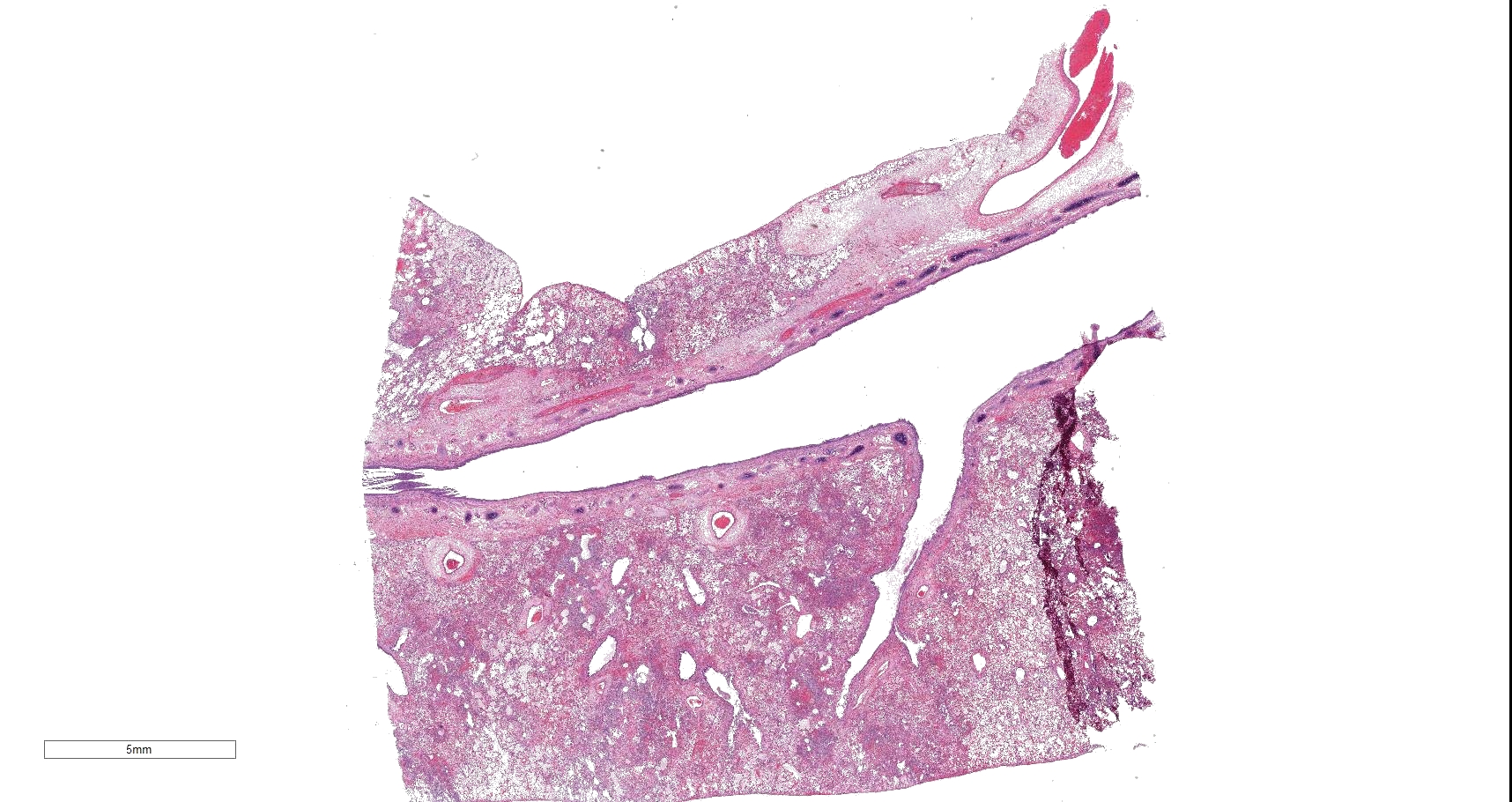
Gross Pathology: All lung lobes were diffusely mottled red to black and palpated as wet and heavy. The remainder of the postmortem examination was unremarkable.
Laboratory results:
Lung bacterial culture: Escherichia coli 3+
E. coli virulence factor PCR: Positive for cytotoxic necrotizing factor 1 (CNF-1)
Gram stain: Gram negative bacilli present
Microscopic description:
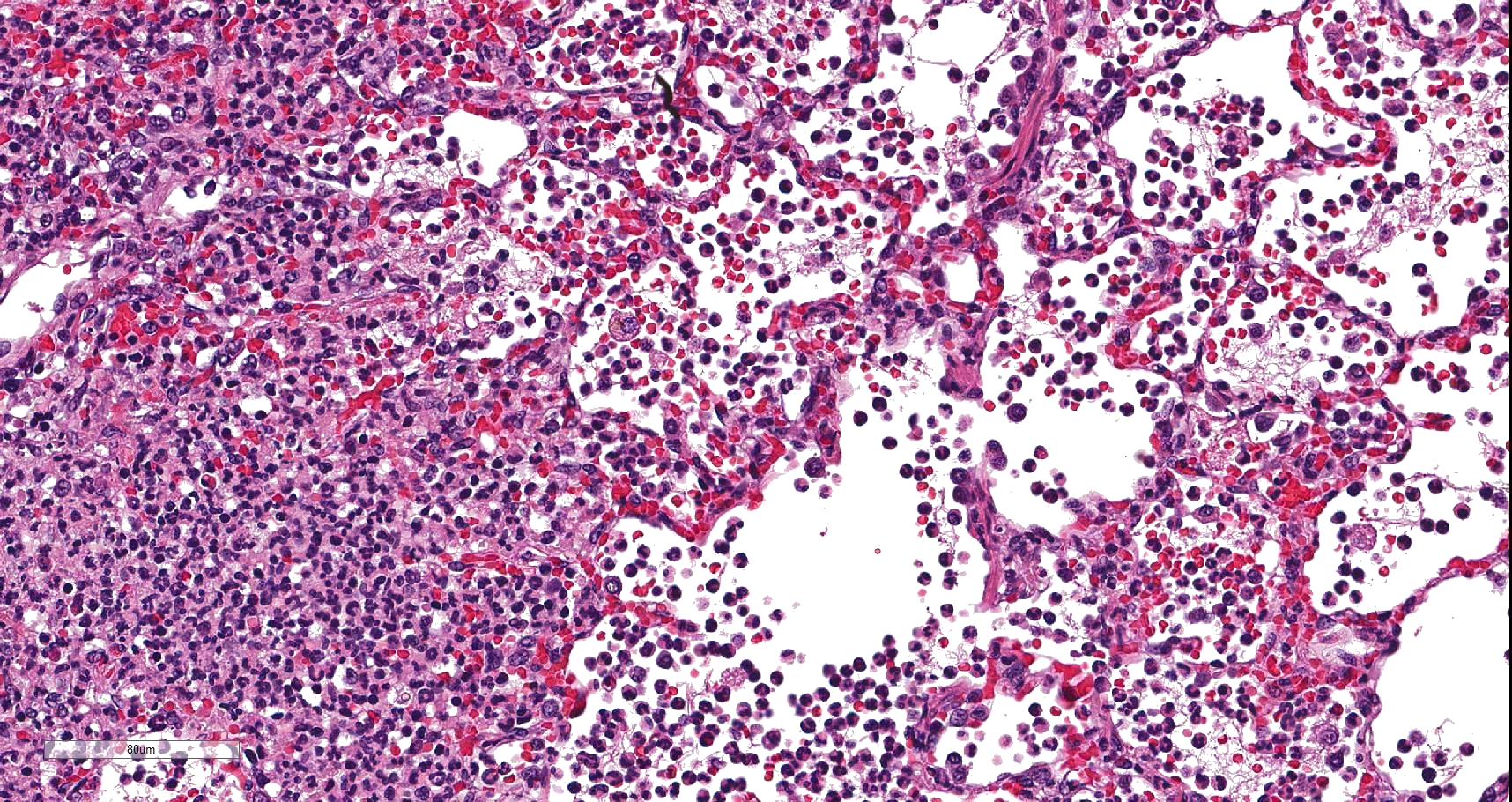
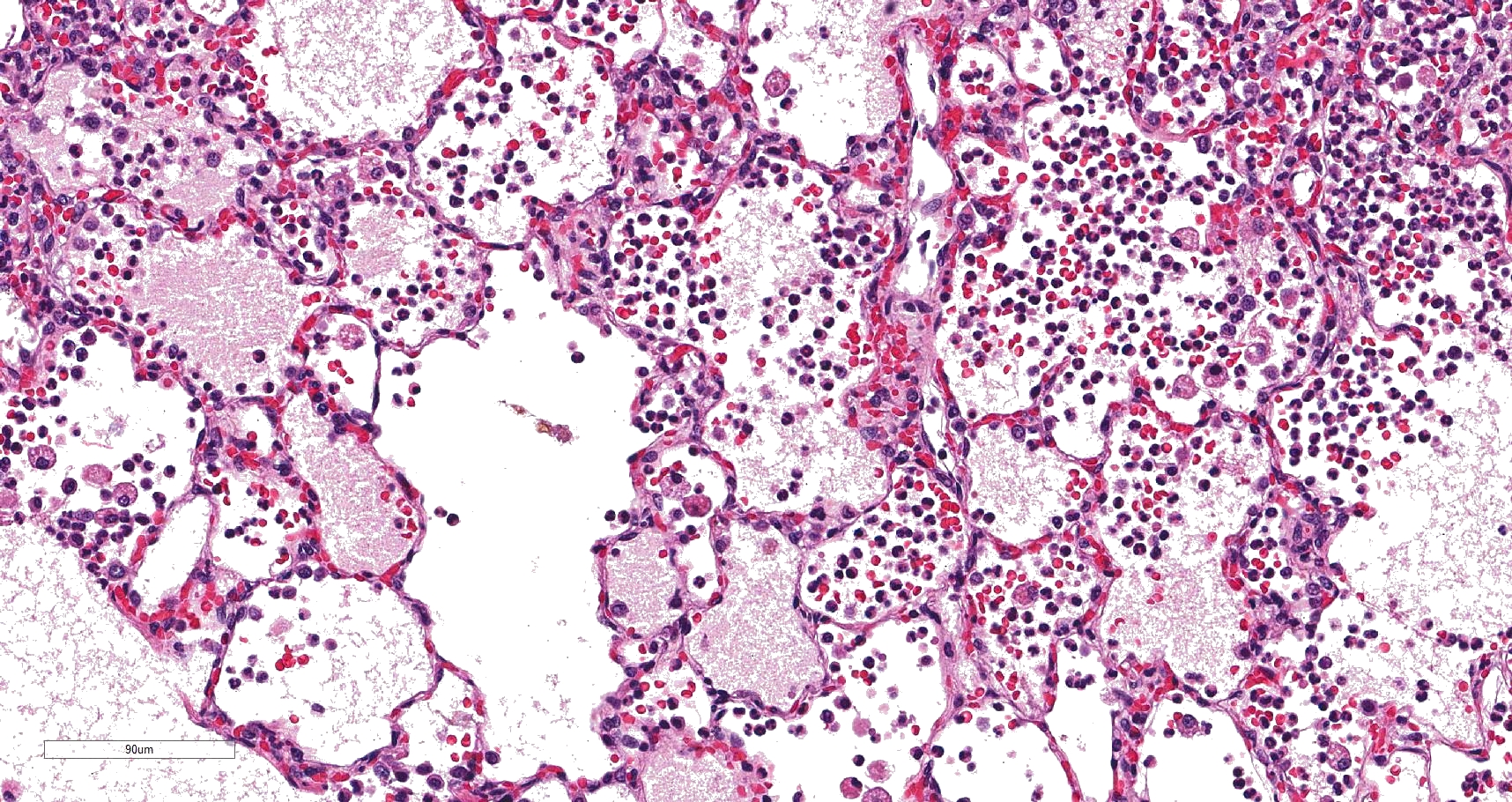
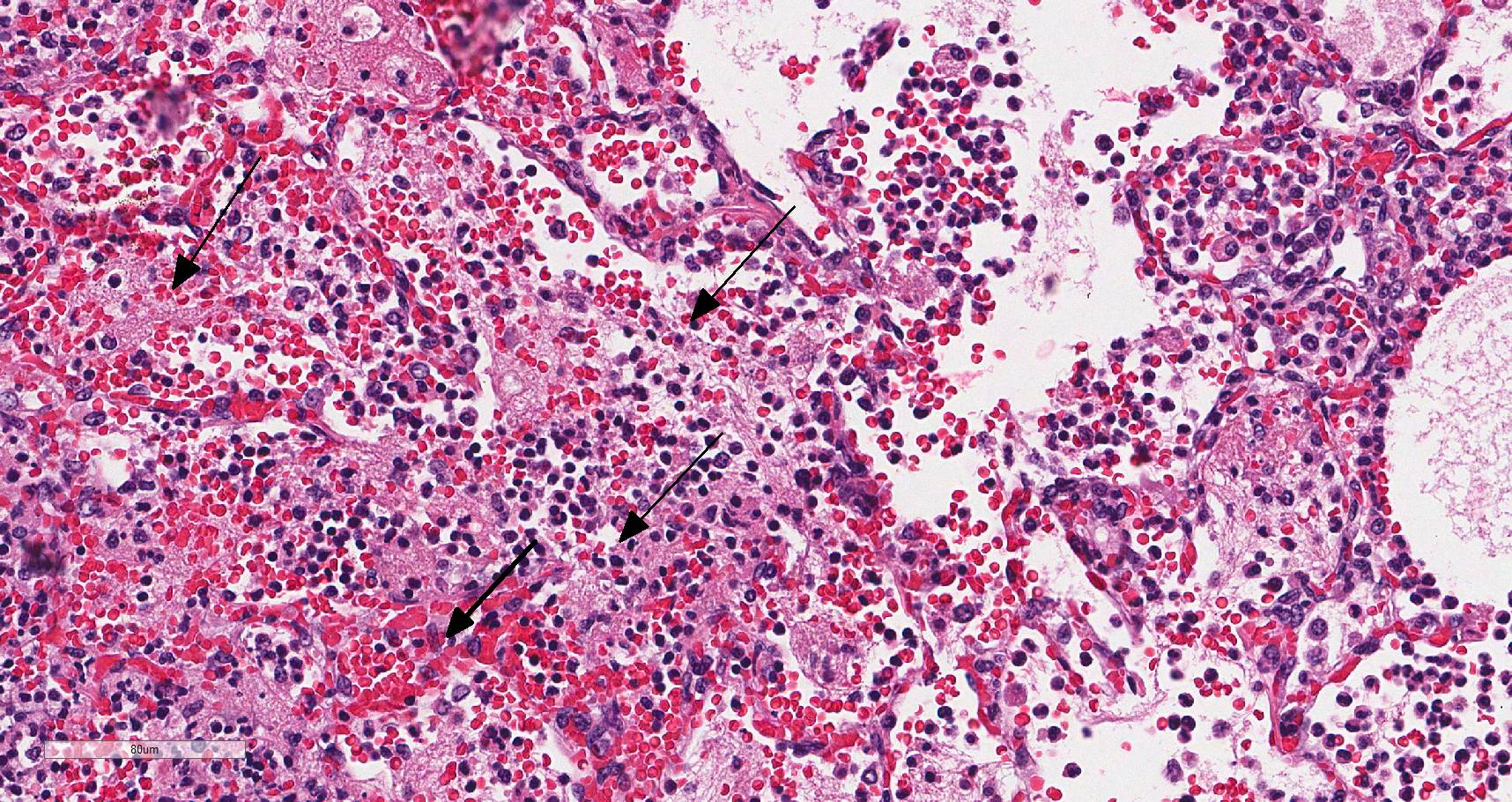
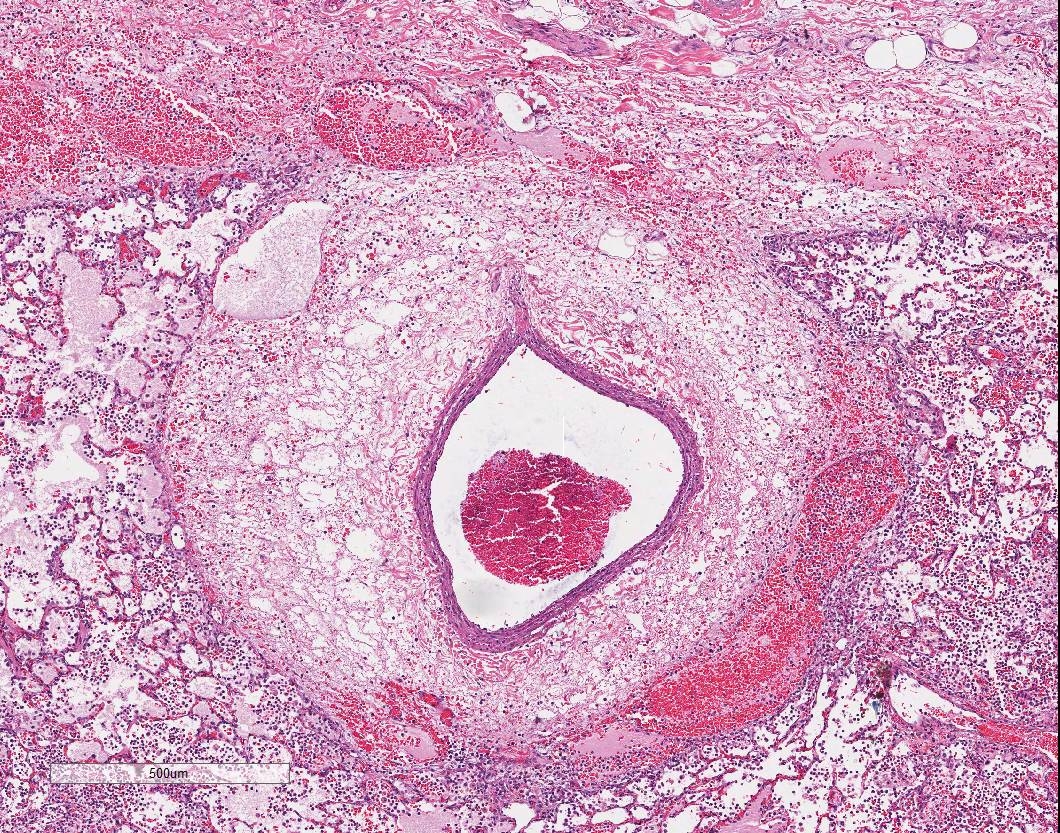
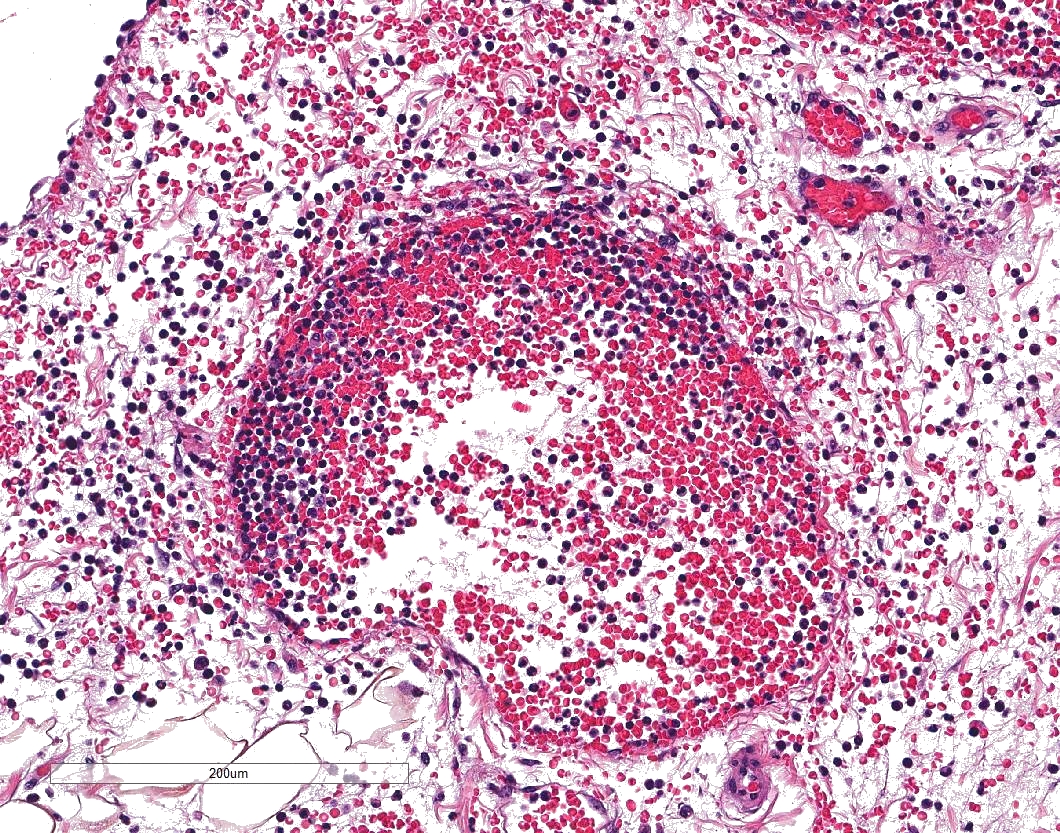
In sections of lung, approximately 75% of the pulmonary parenchyma was effaced by multifocal to coalescing areas of hemorrhage and necrosis. Within these regions, alveoli contained dense accumulations of erythrocytes and neutrophils admixed with aggregates of fibrin, eosinophilic proteinaceous edema fluid, plump vacuolated macrophages, and scattered colonies of bacterial bacilli. Inflammatory infiltrates regionally obliterated alveolar septal architecture. The pleura, interlobular septa, and collagenous connective tissue surrounding bronchovascular bundles were expanded by pale eosinophilic edema fluid and scattered erythrocytes and neutrophils. Vessels frequently contained increased numbers of circulating and marginating leukocytes. Occasional vessels had smudgy, indistinct walls that were segmentally obliterated by neutrophils. Airway lumens multifocally
eosinophilic amorphous material, and necrotic debris.
Contributor's morphologic diagnosis:
Lung: Severe multifocal necrohemorrhagic pneumonia with intralesional bacterial bacilli
Contributor's comment: Extraintestinal patho-genic Escherichia coli (ExPEC) is an infrequently reported cause of acute fatal necrohemorrhagic pneumonia in canines.2 While the majority of Escherichia coli strains are commensals or primary enteric pathogens, ExPEC strains are capable of causing disease in multiple organs systems outside of the gastrointestinal tract. Specifically, ExPEC-associated genitourinary infections, meningitis, pneumonia, and septicemia have been reported in multiple mammalian species including humans.12 ExPEC strains are characterized by the production of certain virulence factors, namely cytotoxic necrotizing factor (CNF) 1 and 2.3 The genes encoding these virulence factors are clustered within a pathogenicity island that has been linked to serogroups O4 and O6.5 The pathogenesis of infection is unknown, but may be related to transmission of bacteria from subclinical carriers via feces or milk.14
Other potential infectious causes of acute hemorrhagic pneumonia in dogs that can have similar clinical presentations and lesions to ExPEC include canine influenza, leptospirosis, and Streptococcus equi spp zoopeidemicus.4,6,11 In canine influenza, the lesions are primarily centered upon the airways with lesser involvement of the alveolar airspaces. 4 Leptospiral pulmonary hemorrhagic syndrome is characterized by severe widespread acute pulmonary hemorrhage with inconspicuous vascular changes.6 Streptococcus equi spp zooepidemicus causes fibrinosuppurative and hemorrhagic pneumonia with prominent colonies of gram-positive cocci.11
Specifically, in purpose-bred beagle research dogs, pneumonia is uncommon and often associated with pulmonary misdosing or potential test article effects rather than infectious etiologies.9 In this case, there were multiple sporadic cases of severe hemorrhagic pneumonia at this facility in recent years, which prompted concern for an underlying infectious cause. Affected dogs were either found dead or presented with fulminant clinical symptoms and were euthanized in extremis. In most cases, dogs were affected immediately post-transport to the facility. CNF 1+ E. coli was isolated from lung tissue for the majority of the cases at this facility. There are rare previous reports of ExPEC-associated disease in other research facilities.5 Given these findings, ExPEC should be considered as an emerging, important cause of acute fatal necrohemorrhagic pneumonia in purpose-bred research dogs and may often be associated with a recent history of transport.
Contributing Institution:
Pathology Department
Charles River Laboratories - Mattawan
www.crl.com
JPC diagnosis:
Lung: Pneumonia, fibrinosuppurative and necrotizing, diffuse, severe, with necrotizing vasculitis and Gram-negative bacilli.
JPC comment:
The contributor describes ExPEC well, and touches upon the variety of pathogenic effects it may have on different organ systems. Recent efforts to perform sequence analysis and whole genome sequencing has identified one sequence type as most prevalent across all regions of the world. This strain of E. coli is known as sequence type 131 (ST131) and possesses a greater number of virulence genes than other identified sequence types. One virulence factor strongly associated with ST131 is the espC gene, which encodes SPATE (serine protease autotransporters of Enterobacteriaceae), while antimicrobial resistance does not appear to be a fitness factor for this bacterium.7 Proteases, and SPATE in particular, allow ExPEC to hydrolyze the peptide bonds present in proteins and increases its virulence. There are more than 20 SPATEs currently identified, with most secreted from members of Enterobacteriaceae.13
Since ExPEC is an important cause of infections in humans (often urinary tract), research has attempted to determine sources of infection. A study of dog parks in Washington, DC in 2015 found that approximately 88% of dog stool samples, and 88% of swabs from shoes of dog park visitors were positive for E. coli. While the majority of these isolates were not drug resistant, a higher percentage of dog park isolates were resistant to more antibiotics than isolates from control shoes.1
Isolates of multiple drug resistant (MDR) ExPEC (ST73, ST127) have been isolated from the feces of endangered southern killer whales (Orcinus orca), though it has not been determined whether these isolates cause disease or contribute to their population decline.8 Comparisons of ExPEC isolates from humans and avians show they have significant genetic overlap and there is evidence for pathogenicity of these avian isolates in mammalian models in vivo. Avian strains may represent an origin of some infections in humans.10
There was debate about the origin of ExPEC in this animal, and whether it most supported an interstitial pneumonia. Many features are consistent with an interstitial pneumonia and hematogenous infection, but further elucidation of pathogenesis would help determine primary changes versus secondary.
References:
- Ahmed L, Price LB, Graham JP. An exploratory study of dog park visits as a risk factor for exposure to drug-resistant extra-intestinal pathogenic E. coli (ExPEC). BMC Research Notes. 2015;8(137).
- Breitschwerdt EB, DebRoy C, Mexas AM, Brown TT, Remick AK. 2005. Isolation of necrotoxigenic Escherichia coli from a dog with hemorrhagic pneumonia. JAVMA 226(12): 2016-2019.
- Carvallo FR, DebRoy C, Baeza E, Hinckley L, Gilbert K, Choi SJ, Risatti G, Smyth JA. 2010. Necrotizing pneumonia and pleuritis associated with extraintestinal pathogenic Escherichia coli in a tiger (Panthera tigris) cub. J Vet Diagn Invest 22: 136-140.
- Castleman WL, Powe JR, Crawford PC, Gibbs EP, Dubovi EJ, Doris RO, Hanshaw D. 2010. Canine H3N8 influenza virus infection in dogs and mice. Vet Pathol 47(3): 507-17.
- Handt LK, Stoffregen DA, Prescott JS, Pouch WJ, Ngai D, Anderson CA, Gatto NT, DebRoy C, Fairbrother JM, Motzel SL, Klein HJ. 2003. Clinical and microbiological characterization of hemorrhagic pneumonia due to extraintestinal pathogenic Escherichia coli in four young dogs. Comparative Medicine 53(6): 663-670.
- Kohn B, Steinicke K, Arndt G, Gruber AD, Guerra B, Jansen A, Kaser-Hotz B, Klopfleisch R, Lotz F, Luge E, Nockler K. 2010. Pulmonary abnormalities in dogs with Leptospirosis. J Vet Intern Med 24: 1277-1282.
- Manges AR, Geum HM, Guo A, Edens TJ, Fibke CD, Pitout JDD. Global extraintestinal pathogenic Escherichia coli (ExPEC) lineages. Clinical Microbiology Reviews. 2019;32(3):e00135-18.
- Melendez D, Roberts MC, Greniger AL, et al. Whole-genome analysis of extraintestinal pathogenic Escherichia coli (ExPEC) MDR ST73 and ST127 isolated from endangered southern resident killer whales (Orcinus orca). J Antimicrob Chemother. 2019;74:2176-2180.
- Mukaratirwa S, Garcia B, Isobe K, Petterino C, Bradley A. 2016. Spontaneous and dosing route-related lung lesions in beagle dogs from oral gavage and inhalation toxicity studies: differentiation from compound-induced lesions. Toxicologic Pathology 44(7): 962-973.
- Nandanwar N, Janssen T, Kuhl M, Ahmed N, Ewers C, Wieler LH. Extraintestinal pathogenic Escherichia coli (ExPEC) of human and avian origin belonging to sequence type complex 95 (STC95) portray indistinguishable virulence features. International Journal of Medical Microbiology. 2014;304:835-842.
- Pesavento PA, Hurley KF, Bannasch MJ, Artiushin S, Timoney JF. 2008. A clonal outbreak of acute fatal hemorrhagic pneumonia in intensively housed (shelter) dogs caused by Streptococcus equi subsp. zooepidemicus. Vet Pathol 45: 51-53.
- Sura R, Van Kruiningen HJ, DebRoyD, Hinckley LS, Greenberg KJ, Gordon Z, French RA. 2007. Extraintestinal pathogenic Escherichia coli-induced acute necrotizing pneumonia in cats. Zoonoses Public Health 54:307-313.
- Tapader R, Basu S, Pal A. Secreted proteases: a new insight in the pathogenesis of extraintestinal pathogenic Escherichia coli. International Journal of Medical Microbiology. 2019;309:159-168.
- Turchetto S, Ustulin M, Citterio CV, Zanardello C, Vascellari M, Vio D, Conedera G, Ferro NM, Milone F, Cocchi M. 2015. Phenotypic features and phylogenetic background of extraintestinal hemolytic Escherichia coli responsible of mortality in puppies. Veterinary Microbiology 179: 126-130.