Signalment:
12-year-old male castrated domestic shorthaired cat (
Felis cats).Presented to the referring veterinarian with a history of anorexia, pyrexia, lethargy, diarrhea and oral ulcers of seven days duration.
Gross Description:
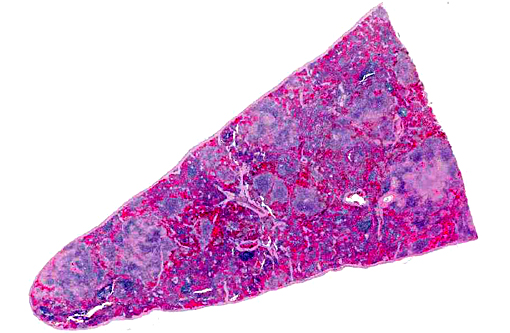
The mesenteric and mandibular lymph nodes were markedly enlarged, and the corticomedullary architecture was effaced by friable, pinpoint yellow-white raised nodules. The pulmonary parenchyma contained several slightly raised, white nodules which were miliary to 2 mm in diameter. 10 and 20 ml of clear, yellow-red fluid was found in the abdominal cavity. The livers were yellowish-brown and the hepatic parenchyma contained several white, slightly raised nodules which were miliary to 2 mm in diameter and evenly distributed through the parenchyma. The spleens contained multiple white, round, raised nodules and were approximately 1-2 mm in diameter.
Histopathologic Description:
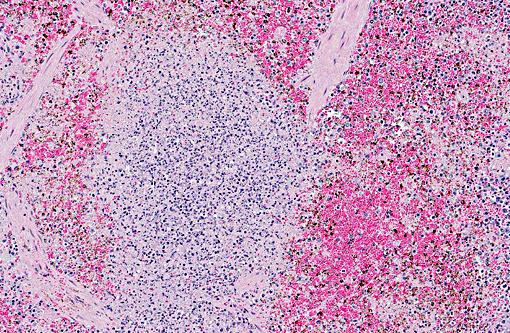
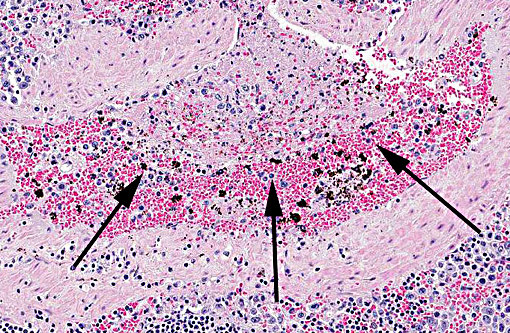
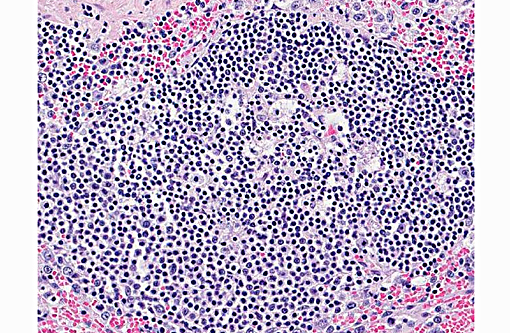
Spleen: The parenchyma is disrupted by multiple nodules composed of degenerate neutrophils, and macrophages and varying amounts of amorphous to slightly fibrillar eosinophilic material (fibrin) and basophilic cellular and nuclear debris (liquefactive necrosis) with loss of lymphoid tissue from the areas surrounding these nodules. Overall, there is a moderate degree of depletion of the periarterial lymphoid sheaths. Erythrocyte and myeloid precursors are scattered throughout the splenic parenchyma with rare mega-karyocytes (extramedullary hematopoesis).
Morphologic Diagnosis:
Moderate to severe multifocal acute necrosuppurative splenitis
Lab Results:
Specimens of spleen, liver, and lung submitted for culture, PCR and DNA sequencing were positive for
Francisella tularensis.
Condition:
Tularemia
Contributor Comment:
Francisella tularensis, the causative agent of tularemia, is an aerobic, gram-negative coccobacillus. The two most common biovars are
F. tularensis biovar
tularensis and
F. tularensis biovar
palaeartica. F. tularensis biovar
tularensis biovar is more common in North America.(1, 10, 6, 2, 8)
F. tularensis biovar
tularensis has been the only biovar identified in fatal systemic infections in cats.(2) Infections have been reported in cats, rabbits, rodents, dogs, cattle, sheep, horses, non-human primates, and humans with cats, rabbits, rodents, and sheep appearing to be the most susceptible to natural infection.(1,6,2) Tularemia is transmitted via bites of infected arthropods (most often ticks or flies), ingestion of infected rabbits and or rodents, inhalation, and mucous membrane exposure.(1, 10, 6, 8) Clinical signs commonly associated with tularemia include fever, anorexia, lymphadenopathy, hepatosplen-omegaly, dehydration, and neutrophilia or neutropenia with toxic change. Common gross findings with tularemia include multifocal splenic, hepatic, lymphoid, and pulmonary necrosis, oral and gastrointestinal ulceration, and enterocolitis.(1, 10, 6, 2, 8) Microscopic findings typically include necrotizing inflammation in multiple organs (spleen, liver, lungs, lymph nodes, and gastrointestinal tract common) with varying numbers of associated neutrophils and macrophages. Colonies of intralesional gram negative coccobacilli may be visualized as well.(1,6,2) Definitive diagnosis is based on successful isolation of
F. tularensis from specimens obtained from affected animals,(1, 10, 6, 2, 8) fluorescent antibody reaction and agglutination with
F. tularensis anti-serum,(1, 10, 6) immunohistochemistry on formalin fixed paraffin embedded tissues,(2,8) or PCR and DNA sequencing.(8)
JPC Diagnosis:
Spleen: Splenitis, necrotizing, multifocal, marked with lymphoid depletion, fibrin thrombi and extramedullary hematopoiesis.
Conference Comment:
Conference participants agreed both red and white pulp are affected by necrosis, but the lesions are centered on the white pulp, with severe lymphoid depletion. Participants described the abundant fibrin thrombi within the section, but noted they are non-occlusive with no evidence of infarction. Differential diagnoses which were discussed included
Yersinia pseudotuberculosis, Yersinia enterocolitica and
Salmonella spp. infection, which share many gross and histologic characteristics with tularemia, as well as
Yersinia pestis. The history provided is typical for both plague and tularemia infection. However,
Yersinia enterocolitica and
pseudotuberculosis are generally associated with large colonies of gram-negative bacilli visible histologically, and
Salmonella spp. infection is less commonly seen in domestic cats. Special stains that can be used to identify
F. tularensis include Gram stains as well as both Giemsa and Warthin-Starry. In this case Gram stains did not help identify organisms in the section.
Tularemia has been reported in nearly all 50 states except Hawaii, and in 2015 is on the rise in North and South Dakota, Colorado, Wyoming, and New Mexico. More common modes of infection in humans include cleaning/skinning infected lagomorphs and arthropod bites, but also include infectious from contaminated water supplies as well as consumption of contaminated undercooked meat. Sporadic outbreaks of tularemia are known to occur in sheep resulting in late term abortion and may be associated with abundant tick populations.(5) In some cases of small mammal infection, classic lesions may not be present grossly or histologically. Additionally, in wildlife species, autolysis and freeze artifact may interfere with evaluation of gross lesions. Subclinical infections are known to occur, but the role of subclinically infected animals in disease transmission is unclear, which serves to highlight the importance of taking appropriate safety precautions when working with the carcasses of deceased wildlife in endemic areas.(4)
The agent is highly infectious with as few as 10-50 organisms capable of causing an infection and it is classified as a category A bioterrorism agent. Experimentally induced lesions from inhalation in African green monkeys included necrotizing pyogranulomatous lesions which targeted the lung and lymphoid tissue.(9) The organisms are most commonly located intracellularly in macrophages, in both the inhalation form in primates(9) as well as in small mammals, but may also be present extracellularly in exudates and necrotic debris as well as in other cell types.(3)
F. tularensis is able to evade the immune system by infecting and replicating in macrophages and disseminating to distal organs in the acute phase of the disease without being detected. It prevents activation of macrophages that would result in an appropriate host innate immune response, which is important in survival in infections with virulent
F. tularensis.(7)
References:
1. Baldwin CJ, Panciera RJ, Morton RJ, Cowell AK, Waurzyniak BJ. Acute tularemia in three domestic cats. JAVMA 1991;199:1602-1605.
2. DeBey BM, Andrews GA, Chard-Bergstrom C, Cox L. Immunohistochemical demonstration of Francisella tularensis in lesions of cats with tularemia. J Vet Diagn Invest. 2002; 14:162-164.
3. Gyuranecz M, Szeredi L, Makrai L et al. Tularemia of European Brown Hare (Lepus europaeus): A pathological, histopathological, and immunohistochemical study. Vet Pathol. 2010;47(5):958-63.
4. Nelson DD, Haldorson GJ, Stanton JB. Francisella tularensis infection without lesions in gray tree squirrels (Sciurus griseus): a diagnostic challenge. J Vet Diag Invest. 2014;26(2):312-315.
5. OToole D, Williams ES, Woods LW et al. Tularemia in range sheep: An overlooked syndrome? J Vet Diagn Invest. 2008;20(4):508-513.
6. Rhyan JC, Gahagan T, Fales WH. Tularemia in a cat. J Vet Diagn Invest. 1990; 2:239-241.
7. Russo BC, Brown MJ, Nau GJ. MyD88-Dependent signaling prolongs survival and reduces bacterial burden during pulmonary infection with virulent Francisella tularensis. Am J Pathol. 2013;183(4):1223-32.
8. Splettstoesser WD, Tomaso H, Al Dahouk S, Neubauer H, Schuff-Werner P. Diagnostic procedures in Tularemia with special focus on molecular and immunological techniques. J. Vet. Med. 2005;52:249-261.
9. Twenhafel NA, Alves DA, Purcell BK. Pathology of inhalational Francisella tularensis spp. tularensis SCHU S4 infection in African green monkeys (Chlorocebus aethiops). Vet Pathol. 2009;46:698-706.
10. Woods JP, Crystal MA, Morton RJ, Panciera RJ. Tularemia in two cats. JAVMA. 1998; 212:81-83.