WSC 2022-2023
Conference 12
Case IV
Signalment:
A 1.6 year-old, female, domestic short hair, Felis catus, cat
History:
Acute death with no prior clinical signs.
Gross Pathology:
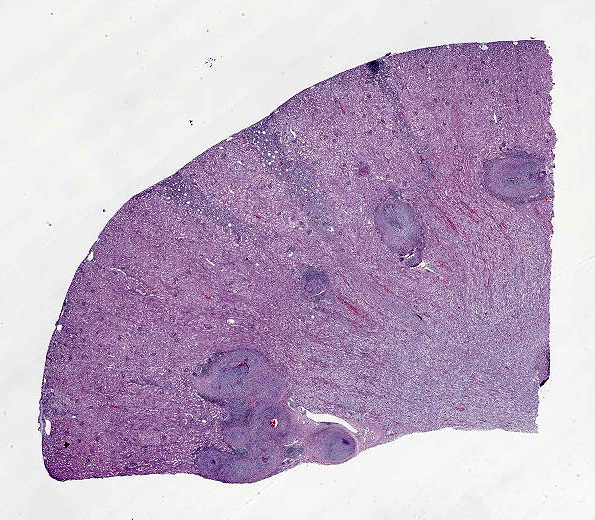
This is the body of a 4.5 kg, reportedly 1.6 year-old female domestic short hair with a body condition score 6/9 and minimal autolysis. The apex of the heart is slightly rounded. Multiple coronary vessels, especially those in the paraconal groove, are segmentally thickened. There are multiple irregular, depressed areas scattered on the renal cortical surfaces (chronic infarctions/tubulointerstitial nephritis). On section, arcuate renal vessels appear up to 3mm thick. There are no other significant findings.
Laboratory Results:
No laboratory results reported.
|
Special stains |
Result |
|
GMS staining |
Negative |
|
PAS staining |
Negative |
|
Acid-fast staining |
Negative |
Microscopic Description:
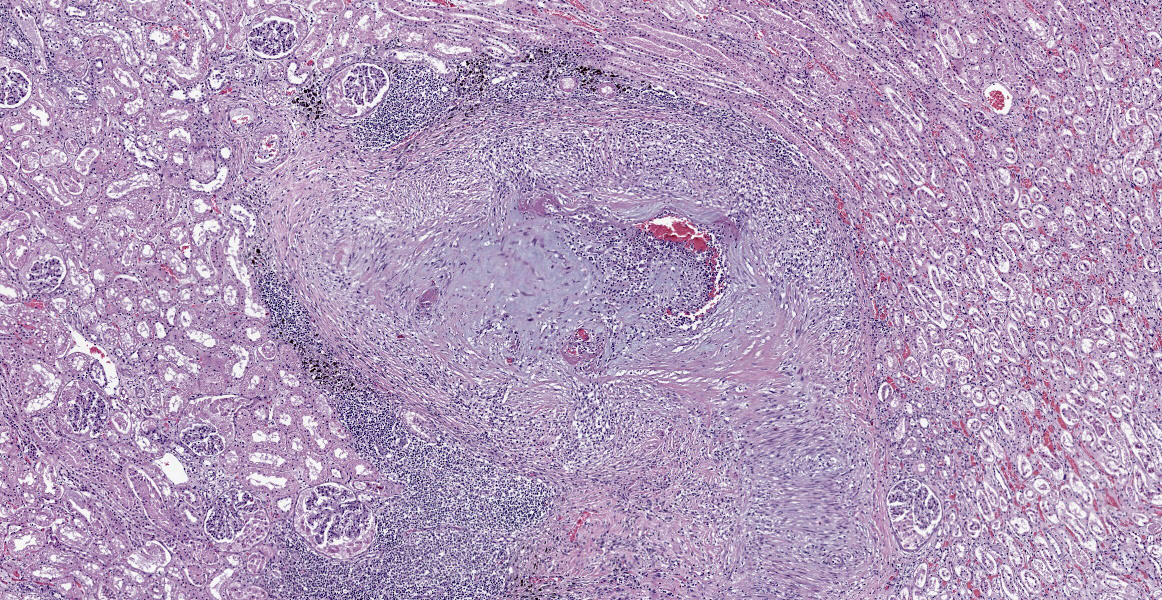
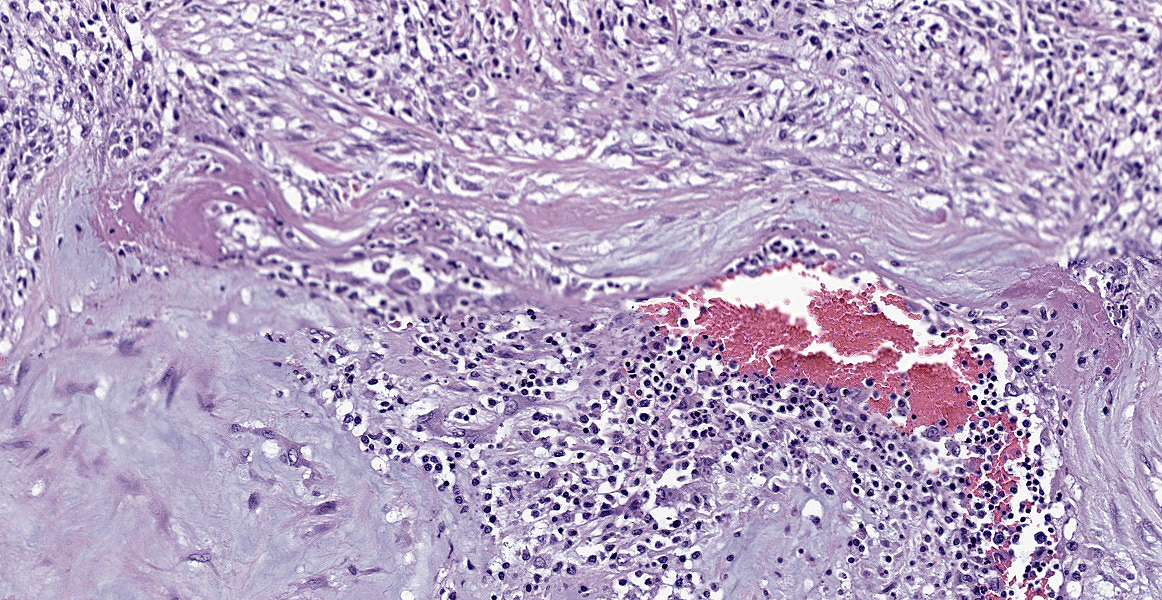
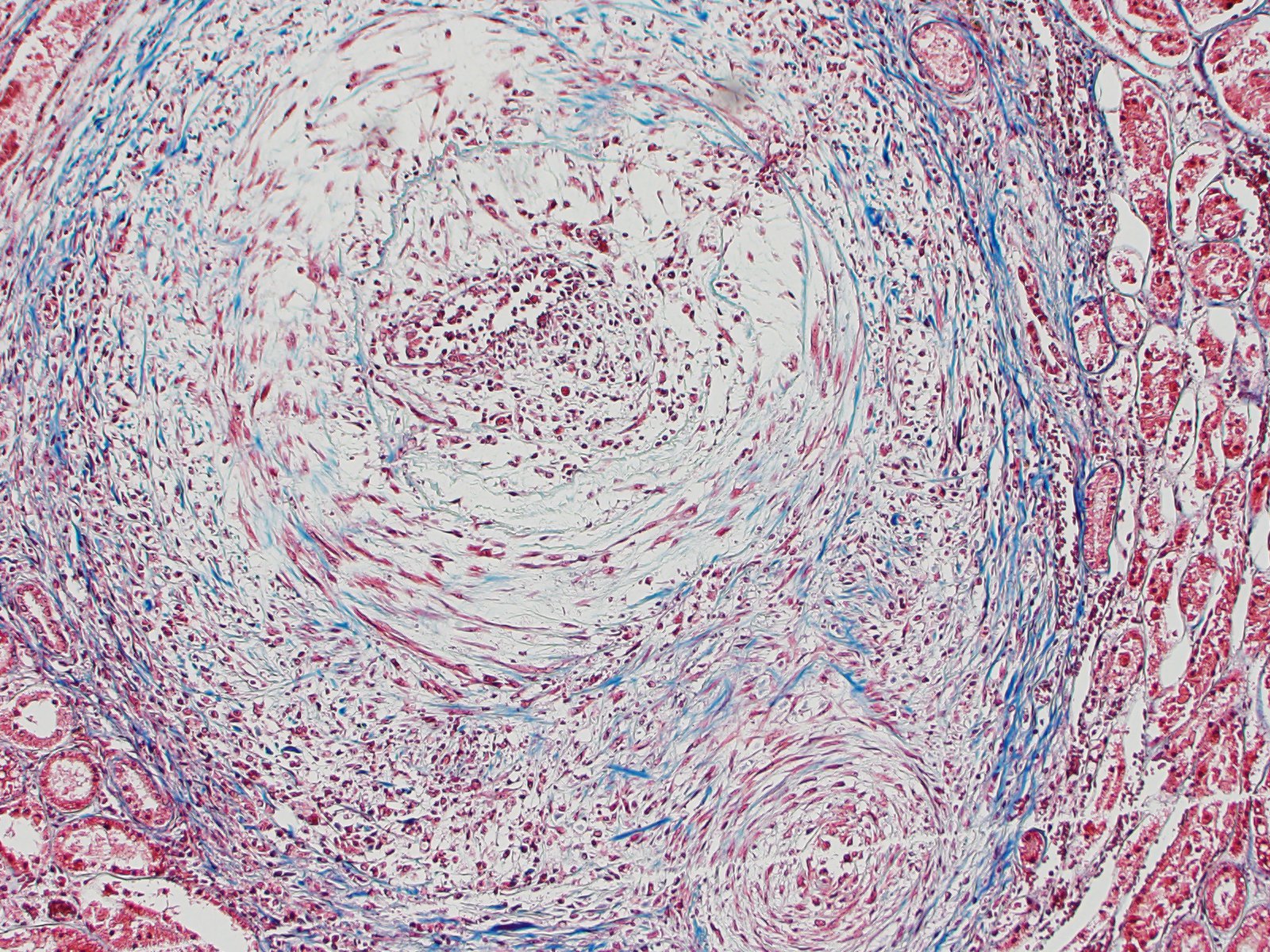
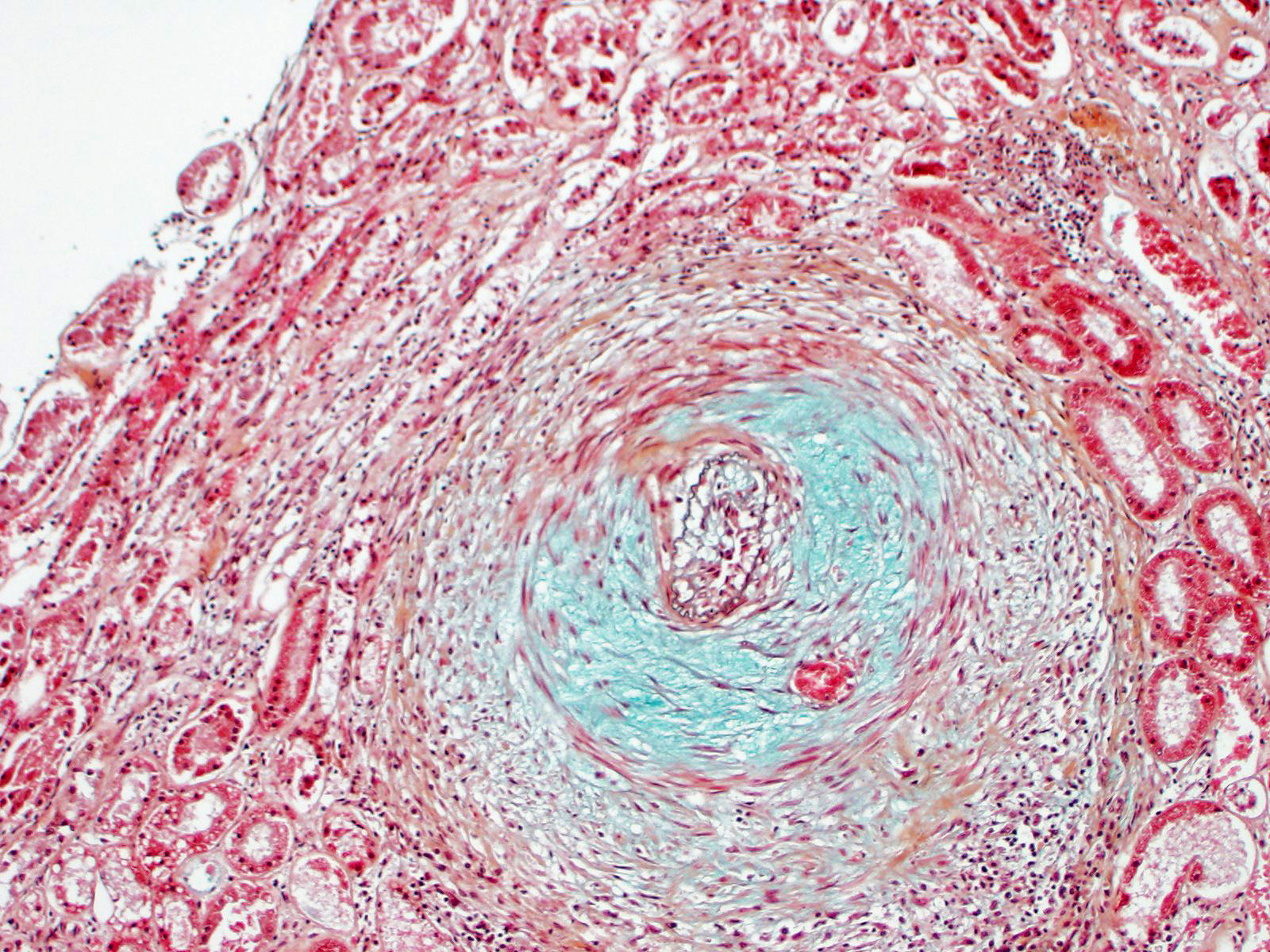
Kidney: Multiple small to large sized arteries are circumferentially obliterated, partially to completely occluded and markedly expanded by a moderately cellular proliferation of spindle shaped cells (presumptive smooth muscle cells and fibroblasts cells) within the tunica intima and media admixed with the pale basophilic extracellular matrix. Occasionally, there are also foci of myointimal necrosis. Multiple dense lymphoid aggregations surround within the tunica adventitia of these affected vessels, and there are moderate lymphocytic, histiocytic and neutrophilic infiltrates transmurally. Multifocally, the endothelial lining of the vessels is plump reactive, infrequently sloughed off from the basement membranes, and lost. The vascular lumens also contain numerous lymphocytes, plasma cells, and infrequent basophilic karyorrhectic debris. Multifocally, the inflammatory reactions that surrounded the vessels extent to the renal interstitial spaces. Multifocally, there is a well-delineated regional infarction involving the renal cortex to the renal medulla.
Contributor’s Morphologic Diagnoses:
Kidney: Severe, multifocal, chronic, necrotizing and pyogranulomatous vasculitis
Contributor’s Comment:
Histopathology has confirmed severe multisystemic vascular disease that is most pronounced in the kidney, heart and a mesenteric artery. Our primary differential is a sterile/immune mediated arteritis, perhaps resembling polyarteritis nodosa (syn. juvenile polyarteritis) as seen in other species (ie. dogs, cynomolgus macaques, human, rat, others).
Polyarteritis nodosa (PAN) is a systemic necrotizing vasculitis mainly involving small- and medium-sized muscular arteries of visceral organs.11, 12 The pathogenesis of PAN is still poorly understood.11,12 In human, PAN has been associated with some virus infections including hepatitis B and C viruses, human immunodeficiency virus, parvovirus B19 and hairy cell leukemia.5,6 An immune-mediated vasculopathy is highly suggestive due to the infiltrates of histiocytes and CD4+ T lymphocytes within the vascular walls.3,5 In veterinary medicine, most cases of PAN have not been associated with infectious agents. However, there are some reports described in many species including blue foxes associated with encephalitozoonosis, in sheep infected with ovine herpesvirus 2 and a streptococcal infection has been suspected in pigs.7,8,9
PAN has been recently reported in a cat and in our case, the histopathological findings and gross findings resemble to recently described in the cat which includes systemic necrotizing vasculitis involved the small, medium and large arteries of the heart, kidneys, mesentery.11 In addition to the recent report, our case demonstrates intense neutrophilic infiltration of the vascular walls.11
Some other diagnostic considerations are discussed below. Perhaps the best recognized vasculopathy in the cat is feline infectious peritonitis. Although we have not definitively excluded this pathogen, we suggest that the histologic picture is not entirely typical of this syndrome (ie. morphologic pathology, distribution of lesions). Another possibility might be severe systemic hypertension, but we note that some organs typically affected by hypertension are not involved (ie. meninges, eye).
Contributing Institution:
Oregon State University Magruder Hall Rm. 134 700 SW 30th Street Corvallis, OR 97331
https://vetmed.oregonstate.edu/diagnostic
JPC Diagnosis:
Kidney: Arteritis, proliferative and necrotizing, chronic, diffuse, severe, with cortical infarcts.
JPC Comment:
While relatively uncommon in cats, systemic necrotizing vasculitis is a common background lesion in laboratory species, specifically mice, rats, beagle dogs, and Gottingen minipigs.1,2,4 In mice, polyarteritis can affect multiple organ systems and tends to occur in strains predisposed to autoimmune diseases, such as MRL and NZB strains. In the kidneys, it can cause segmental infarction, similar to what was seen in this case.1 Other organs that may be infected include the tongue, pancreas, mesentery, and heart.1 Polyarteritis may also affect medium sized vessels around the inner and middle ear, causing vestibular signs.1 Polyarteritis nodosa is a chronic progressive disease of rats and occurs more commonly in rats that are older, male, or Sprague Dawley or spontaneous hypertensive strains.1,4 Many organ systems can be affected, including the mesentery, kidneys, and testis; however, the lung is spared.1,4 In beagles, polyarteritis occurs in small to medium caliber arteries of the leptomeninges, cranial mediastinum, and heart and is characterized by lymphoplasmacytic and histiocytic inflammation with fibrinoid necrosis and possible hemorrhage.2,4 Previously known as beagle pain syndrome for the primary breed affected, this condition is now referred to as steroid responsive meningitis-arteritis and is known to affect other medium and large breeds as well.2,4 Affected animals have fever and severe spinal pain.2 Spontaneous polyarteritis also occurs occasionally in Gottingen minipigs, most commonly in small to medium caliber vessels in a single or multiple organs. Affected sites include the heart, reproductive tract (epididymis, oviduct, vagina), kidney, rectum, stomach, urinary bladder, and spinal cord.4 An important differential to consider in laboratory species used in pre-clinical drug safety studies is drug-induced vasculopathy, which may be induced by vasodilators (minoxidil, endothelin receptor antagonists), bronchodilators (phosphodiesterase inhibitors), and immunomodulatory agents (hydrocortisone, betamethasone).4
References:
- Barthold SW, Griffey SM, Percy DH. Pathology of Laboratory Rodents and Rabbits. 4th John Wiley & Sons, Inc: 2016; 98, 99, 156.
- Cantile C, Youssef S. Nervous System. In: Maxie MG, ed. Jubb, Kennedy, and Palmer’s Pathology of Domestic Animals. Vol 1. 6th Elsevier; 2016: 395.
- Cid MC, Grau JM, Casademont J, Campo E, Coll?Vinent B, Lopez?Soto A, Ingelmo M, Urbano?Marquez A. Immunohistochemical characterization of inflammatory cells and immunologic activation markers in muscle and nerve biopsy specimens from patients with systemic polyarteritis nodosa. Arthritis Rheum. 1994;37(7):1055-1061.
- Dincer Z, Picciuto V, Walker UJ, Mahl A, McKeag S. Sponatneous and Drug-Induced Arteritis/Polyarteritis in the Gottingen Minipig-Review. Tox Pathol. 2018; 46(2):121-130.
- Forbess L, Bannykh S. Polyarteritis nodosa. Rheum Dis Clin North Am. 2015;41(1):33-46.
- Ishiguro N, Kawashima M. Cutaneous polyarteritis nodosa: a report of 16 cases with clinical and histopathological analysis and a review of the published work. J Dermatol. 2010;37(1):85-93.
- Liu CH, Chiang YH, Chu RM, Pang VF, Lee CC. High incidence of polyarteritis nodosa in the brains of culled sows. J Vet Med Sci. 2005;67(1):125-7.
- Nordstoga K, Westbye K. Polyarteritis nodosa associated with nosematosis in blue foxes. Acta Pathol Microbiol Scand A. 1976;84(3):291-6.
- Pesavento PA, Dange RB, Ferreras MC, et al. Systemic necrotizing vasculitis in sheep is associated with ovine herpesvirus 2. Vet Pathol. 2019: 56(1):87-92.
- Robinson WF, Robinson NA. Cardiovascular System. In: Maxie MG, ed. Jubb, Kennedy, and Palmer’s Pathology of Domestic Animals. Vol 3. 6th Elsevier; 2016.
- Salvadori C, Vezzosi T, Marchetti V, Cantile C. Polyarteritis Nodosa in a Cat with Involvement of the Central and Peripheral Nervous Systems. J Comp Path. 2019;167:6-11.
- Wessels M, Strugnell B, Woodger N, Peat M, La Rocca SA, Dastjerdi A. Systemic necrotizing polyarteritis in three weaned lambs from one flock. Jour Vet Diagn Invest. 2017;29(5):733-7.