Signalment:
45 day old, female, crossbred, Caesarian-derived, colostrum-deprived (CDCD) domestic swine (
Sus scrofa domestica)This pig was experimentally inoculated with 10
3 TCID
50 porcine circovirus type 2 (PCV 2) at 21 days of age. The pig was anorexic and icteric days 20-24 post inoculation. The pig was also febrile (rectal temperature > 40.0° C) for seven days prior to euthanasia and necropsy on day 24 post inoculation.
Gross Description:
There was marked, generalized lymphadenopathy. Icterus was observed in the skin, sclera, subcutaneous tissue, pericardium, urine, and periosteal tissues. The liver was markedly enlarged and had a mottled yellow, tan, and red color pattern. There were multifocal white foci throughout the parenchyma of the kidney.
Morphologic Diagnosis:
Liver: hepatitis, diffuse, subacute, lymphohistiocytic, necrotizing, severe, with occasional intracytoplasmic botryoid inclusion bodies.
Lab Results:
PCR on fresh tissues for PCV 2 was positive from multiple tissues; PCR on fresh tissues for porcine parvovirus was negative.
Virus isolation for PCV 2 was positive from multiple tissues; virus isolation for PRRS virus was negative.
In situ hybridization for PCV was positive from multiple tissues including the liver.
Condition:
Porcine Multisystemic Wasting Disease
Contributor Comment:
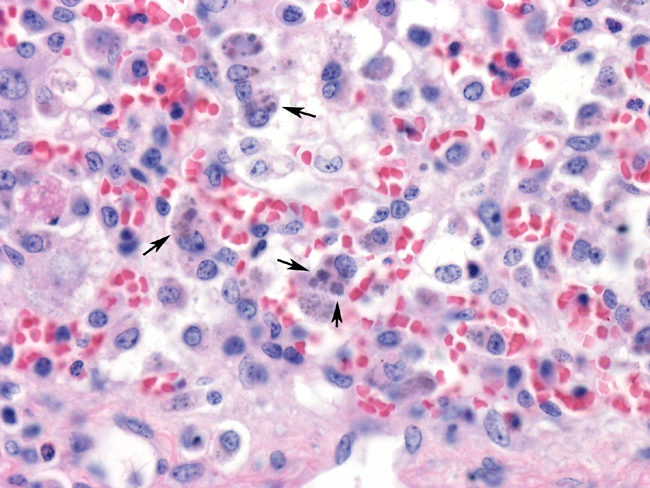
These slides contain sections of liver in which there is diffuse alteration of the normal hepatic architecture. There is marked separation of hepatic cords due to distension of the sinusoids by clear space, erythrocytes, and low to moderate numbers of inflammatory cells. Hepatocytomegally is a pronounced feature, and binucleated hepatocytes are commonly observed. Single-cell necrosis characterized by pyknosis, karyorrhectic debris, and Councilman bodies is a common feature. Foci of lymphocytes and macrophages can be seen haphazardly arranged throughout the sections. Neutrophils can occasionally be observed within the sinusoids. Intrahepatocellular bile pigment can frequently be observed. Some sections contain hepatocytes with intracytoplasmic, ampiphilic to basophilic inclusion bodies. Multiple, variably sized inclusion bodies arranged in clusters (botryoid) can often be seen within a single cell. Less frequently basophilic intranuclear inclusion bodies are present.Â
Porcine circoviruses are members of the family Circoviridae which contain the smallest viruses known to infect animals. Circoviridae contain the genera circovirus (porcine circoviruses, pigeon circovirus, and psittacine beak and feather disease virus) and gyrovirus (chicken anemia virus). The human transfusion-transmitted virus (TTV) has been proposed to be grouped within the family Circoviridae.
Porcine circoviruses are icosahedral, nonenveloped, and contain a single-stranded, circular DNA genome of approximately 1,760 bases, and measure 17-20 nm in diameter. Porcine circoviruses have been sub-grouped into two types based on genomic differences. Porcine circovirus type 1 was first recognized as a contaminant of the PK-15 cell culture line and has not been proven to cause clinical disease in swine. Porcine circovirus type 2 has been associated with outbreaks of postweaning multisystemic wasting syndrome (PMWS) and porcine dermatitis and nephropathy syndrome (PDNS). PCV1 and PCV2 are antigenically similar but can be segregated by serologic tests.
PMWS was first recognized in high-health status swine herds in western Canada in 1991 and has since been reported world-wide. PMWS is a low morbidity syndrome characterized by weight loss, failure to grow, diarrhea, dyspnea, and jaundice. Common gross lesions include generalized lymphadenopathy, hepatomegaly, gastric ulceration, nephritis, and interstitial pneumonia. Microscopically, there is disseminated depletion of lymphoid follicles, lymphohistiocytic inflammation in multiple tissues, interstitial nephritis, hepatitis, and bronchointerstitial pneumonia. The pathognomonic intracytoplasmic botryoid clusters of ampiphilic to basophilic, variably sized inclusion bodies can be found within numerous cell types, particularly macrophages, depending on the stage of infection.
The lesions of PMWS have been reproduced with PCV2 alone and in combination with other viral agents, including porcine parvovirus and PRRS virus. This sample comes from experimental reproduction of PMWS in CDCD pigs with PCV2 alone.Â
JPC Diagnosis:
Liver: Hepatitis, necrotizing and lymphohistiocytic, diffuse, severe,
with karyomegaly and few basophilic botryoid intracytoplasmic inclusions, crossbred,
porcine.
Gallbladder: Cholecystitis, neutrophilic, diffuse, mild, with pericholecystic edema.
Conference Comment:
PMWS develops most often in pigs 5-12 weeks old and has a
morbidity rate of approximately 5-10%. Although PCV2 alone can induce PMWS, PCV2
will result in more severe disease during a co-infection with either porcine parvovirus
(PPV) or porcine reproductive and respiratory syndrome virus (PRRSV). Activation of
the immune response increases replication of PCV2. The role of PCV2 in other
diseases of swine is controversial because PCV2 can be isolated from healthy pigs.
The isolation of PCV2 alone does not result in a diagnosis of PMWS; the diagnosis also
requires the consistent gross and clinical signs.
The primary gross lesion of PMWS is generalized lymphadenopathy. Other gross
findings may include hepatomegaly, gastric ulceration, nephritis and interstitial
pneumonia. The histologic lesions of PMWS include lymphohistiocytic inflammation in
multiple organs with basophilic intracytoplasmic botryoid inclusions, lymphoid depletion,
and granulomatous interstitial pneumonia. Porcine dermatopathy and nephropathy
syndrome (PDNS) is primarily associated with PCV2, but has also been associated with
PRRSV,
Pasteurella multocida, and
Streptococcus sp. Gross lesions of PDNS include
red papules over the hindquarters, perineum and ears, and enlarged edematous
kidneys with petechiae. Histological lesions include vasculitis, hemorrhage, necrosis,
and acute exudative glomerulonephritis.
Conference attendees discussed the differentiating PRRS from PMWS. Lymphocytes
are the predominant inflammatory cell in cases of PRRS, whereas macrophages
dominate in PMWS. The intracytoplasmic basophilic inclusion bodies are specific to a
diagnosis of PCV2 infection.
PCV2 is a nonenveloped, icosohedral, DNA virus that forms paracrystalline arrays.
Conference participants discussed other viruses that form paracrystalline arrays on EM.
A useful mnemonic device used by AFIP residents at the AFIP is -�-�PICA for
Polyomavirus, Picornavirus, Iridovirus, Circovirus, and Adenovirus.
Not all sections contained gallbladder.
References:
Allan GM, Ellis JA. 2000. Porcine circoviruses: a review. J Vet Diagn Invest 12:3-14.
Allan GM, McNeilly F, Kennedy S, Daft, B, Clarke, EG, Ellis, JA, Haines, DM, Meehan, BM, Adair, BM. 1998. Isolation of porcine circovirus-like viruses from pigs with a wasting disease in the USA and Europe. J Vet Diagn Invest 10:3-10.
Allan GM, Phenix KV, Todd D, and McNulty MS. 1994. Some biological and physico-chemical properties of porcine circovirus. J Vet Med B 41-17-26.
Bolin SR, Stoffregen WC, Nayar GPS, and Hamel AL. 2001. Postweaning multisystemic wasting syndrome induced after experimental inoculation of cesarean-derived, colostrum-deprived piglets with type 2 porcine circovirus. J Vet Diagn Invest 13:185-194.
Ellis J, Krakowka S, Lairmore M, Haines D, Bratanich A, Clark E, Allan A, Konoby C, Hassard L, Meehan B, Martin K, Harding J, Kennedy S, McNeilly F. 1999. Reproduction of lesions of postweaning multisystemic wasting syndrome in gnotobiotic piglets. J Vet Diagn Invest. 11:3-14.
Harding JCS, Clark EG, Strokappe JH, Wilson PI, Ellis JA. 1998. Postweaning multisystemic wasting syndrome: epidemiology and clinical presentation. Swine Health and Production 6:249-254.