Joint Pathology Center
Veterinary Pathology Services
Wednesday Slide Conference
2018-2019
Conference 21
27 March 2019
CASE I: NCAH 2013-1 (JPC 4033381)
Signalment: 38 day old specific-pathogen-free chicken, Gallus gallus domesticus
History: The chicken was inoculated intratracheally with 0.5 mL of an experimental agent (laryngotracheitis virus). Clinical signs recorded on day 2 and 3 post inoculation include dyspnea, conjunctivitis, and depression. The bird died 4 days post inoculation.
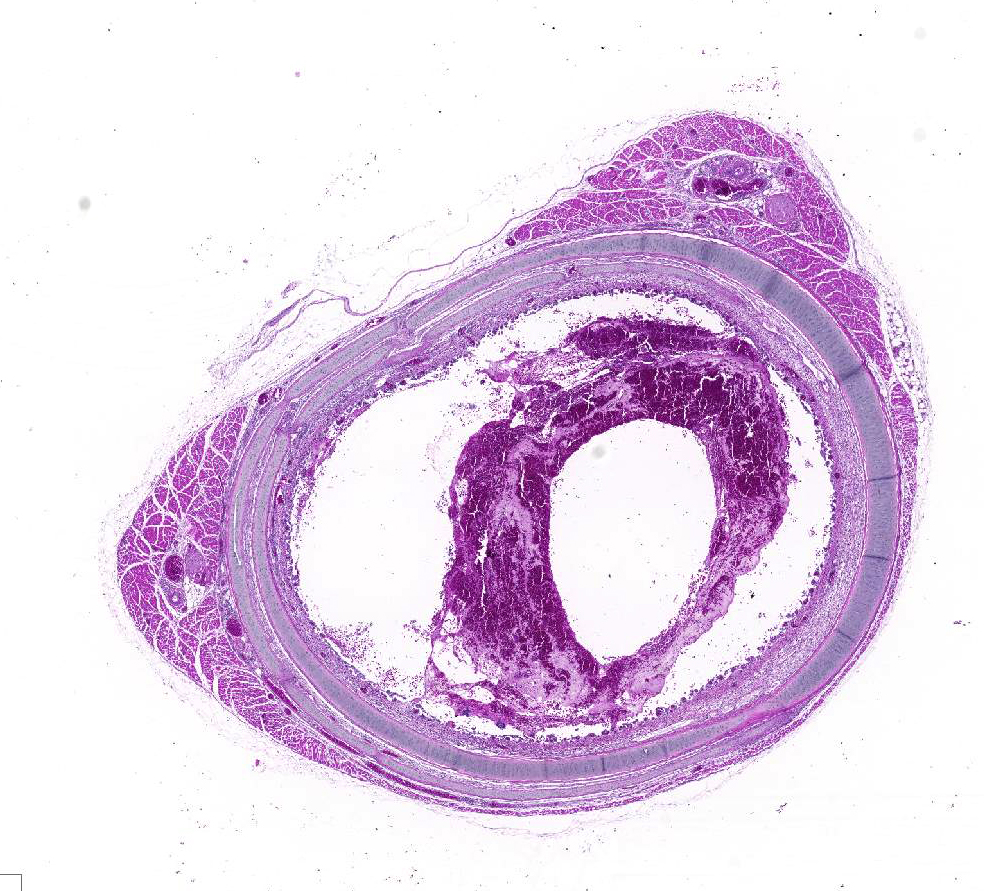
Gross Pathology: On cut in, the tracheal lumen was partially obstructed by an opaque, thick blood-tinged exudate.
Laboratory results: N/A
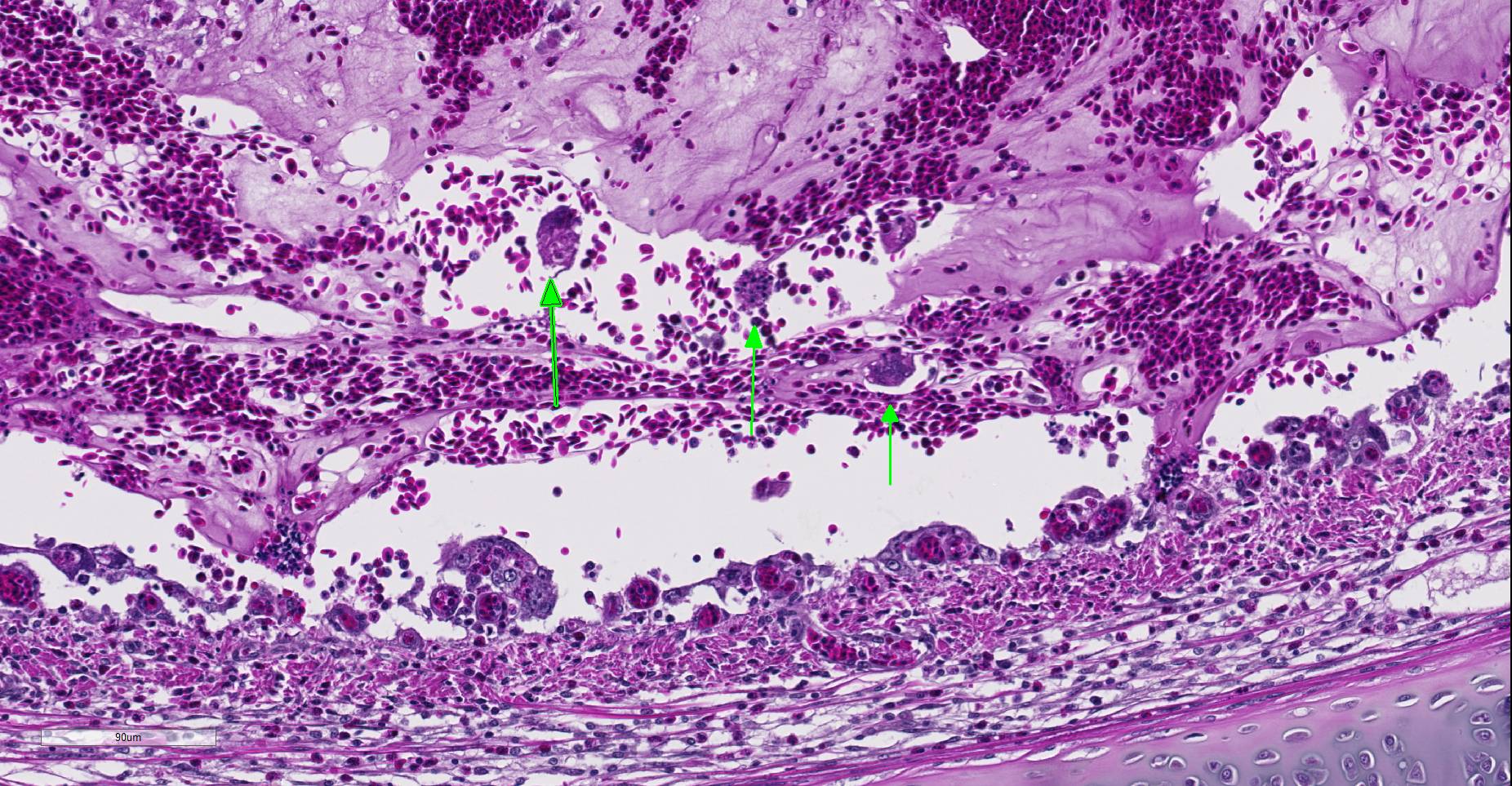
Microscopic Description: Trachea: The lumen is 50% occluded by a coagulum composed of predominantly free erythrocytes admixed with eosinophilic proteinaceous fluid and fibrillar material (serum and fibrin), heterophils, and sloughed degenerate individual or syncytial epithelial cells. Syncytia contain up to 20 nuclei, often with large eosinophilic intranuclear inclusion bodies and marginated chromatin. The coagulum is multifocally adhered to the extensively eroded or ulcerated tracheal mucosa with some degree of artifactual separation. Ulceration extends into the lamina propria and submucosa with complete loss of goblet cells and projection of capillaries into the lumen. The mucosa consists of loosely adhered ciliated epithelia that often form syncytia, attenuated non-ciliated epithelium, or necrotic cellular debris. The lamina propria and submucosa are expanded by clear space (edema), fibrin, heterophils, and fewer lymphocytes and plasma cells. There is diffuse vascular congestion and multifocal hemorrhage. Within the surrounding adventitia and muscular interstitia are small numbers of predominantly lymphocytes.
Contributor’s Morphologic Diagnoses:
Trachea: Tracheitis, hemorrhagic and ulcerative, acute, severe with epithelial syncytia and intranuclear inclusion bodies.
Contributor’s Comment: Avian laryngotracheitis (LT) is a viral respiratory tract infection of chickens, caused by the alphaherpesvirus Gallid herpesvirus I, also known as laryngotracheitis virus (LTV).6 The disease has a worldwide distribution, but has the highest incidence in areas of concentrated poultry production.1 LT causes increased mortality and decreased egg production in chickens, leading to economic losses for the poultry industry.6 The eye and respiratory tract are the usual portals of entry of LTV.6 Sources of infection include chickens with acute laryngotracheitis disease, latently infected chickens shedding virus, and fomites. Clinical signs generally appear 6-12 days following natural exposure. Clinical signs in severe cases include marked dyspnea, conjunctivitis, and depression.4 Severe forms of the disease cause high morbidity and variable mortality. Mild enzootic cases of laryngotracheitis cause unthriftiness, decreased egg production, watery eggs, conjunctivitis, and nasal discharge with low morbidity and mortality.6
Innate and cell-mediated immune responses appear to be more important than humoral responses in protection against disease.2 Most alphaherpesviruses code for glycoprotein G which binds to a range of chemokines, modulating the host immune response, e.g. blocking binding of the chemokine to its host receptor. 2 In addition, glycoprotein G may enhance viral survival by modulating the host response toward humoral immunity rather than cell-mediated immunity.2
The diagnosis of LT can be made by histopathology, virus detection, or serology.1 The disease is controlled through strict biosecurity measures and vaccination. Although attenuated viruses have been used by the poultry industry for years, virally vectored vaccines have recently become available, and deletion-mutant vaccines are under development.2
Differential diagnoses include infections with avian poxvirus, avian influenza virus, Newcastle disease virus, infectious bronchitis virus, fowl adenovirus, and Aspergillus spp.4
Contributing Institution:
National Animal Disease Center, Ames, IA http://ars.usda.gov/main/site_main.htm?modecode=36-25-30-00
http://www.aphis.usda.gov/animal_health/lab_info_services/about_nvsl.shtml
JPC Diagnosis: Trachea: Tracheitis, necrotizing and heterophilic, moderate with multifocal ulceration, epithelial intranuclear viral inclusion bodies and viral syncytia.
JPC Comment: The viral cause of infectious laryngotracheitis in poultry is gallid herpesvirus-1, a member of the Iltovirus (ILTovirus – get it?) genus of the subfamily Alphaerpesviridae. The disease was first reported in the 1920s, and since the advent of vaccination, two distinct clinical forms have been recognized – the classical severe form resulting in necrohemorrhagic lesions of the upper respiratory tract, and a milder form resulting in depression, reduced feed conversion and egg production, conjunctivitis, nasal discharge and sinusitis. This mild form is the most common type in the US, and is called the “vaccinal form” as analysis of the virus from many of these outbreaks demonstrate viral genome regions characteristic of vaccine strains.5
While effective in preventing infection, vaccines can also create latent carrier birds, which may spread virus to non-vaccinated flocks. As modified live vaccines may increase their virulence by bird to bird passage, especially those grown in chick embryos rather than in tissue culture, this may result in outbreaks of vaccinal forms of ILT when used over time in areas in which field-type strains are not endemic.5 Moreover, CEO vaccines, often administered in drinking water, require contact with the nasal tissues during this process, and may not result in adequate coverage of birds within a given flock. This, in turn, allows for longer circulation of the virus within said flock, increased numbers of passages within birds, and ultimately predisposes to outbreaks of vaccinal laryngotracheitis.3
A recent publication by Sary et al7, demonstrated plaque-like necrotic lesions in the oral cavity, buccal mucosa, and esophagus of one laying hen and one pheasant from the same mixed species flock. Mucosal necrosis, viral inclusions, and viral syncytia which demonstrated immunopositivity for ILTV were seen in the lesions. The laying hen had additional typical lesions characteristic of ILT in the respiratory tract. This was the first description of oral lesions since very early descriptions of the disease in the 1920s.
The moderator imparted his extensive experience with this disease. He mentioned that many infections are seen in older birds up to 40 days (heavier birds which result in increased labor costs to remove from houses in outbreaks). A marked drop in water consumption occurs 24 hours prior to the break, and many birds show conjunctivitis rather than respiratory signs – for this reason, conjunctiva should always be examined as well as trachea. In the US, the disease oftenpeaks in the winter months, but this may not be true in other parts of the world. Vaccination does not prevent infection, but may mask or hide signs of an outbreak. The moderator thinks that the presence of viral inclusions and syncytia is diagnostic and unique for this condition; infectious bronchitis is a differential in this case gross and microscopically to an extent, but will never make syncytia.
References:
- Bagust TJ, Jones RC, Guy JS: Avian infectious laryngotracheitis. Rev Sci Tech 2000; 19: 483-492.
- Coppo MJ, Hartley CA, Devlin JM: Immune responses to infectious laryngotracheitis virus. Dev Comp Immunol, 2013
- Menendez KR, Garcia M, Spatz S, Tablante NL. Molecular epidemiology of infectous laryngotracheitis: a review. Avian Pathol 2014; 43(2):108-117.
- Oldoni I, Rodriguez-Avila A, Riblet SM, Zavala G, Garcia M: Pathogenicity and growth characteristics of selected infectious laryngotracheitis virus strains from the United States. Avian Pathol 2009:38: 47-53,
- Ou SC, Giambrone JJ. Infectious laryngotracheitis virus in chickens. World J Virol 2012:1(5):142-149.
- Saif YM, Barnes HJ: Diseases of poultry, 12th ed. Blackwell Pub. Professional, Ames, Iowa, 2008
- Sary K, Chenier S, Gagnon CA, Shavaprasad HL, Sylvester D Boulianne M. Esophagitis and pharyngitis associated with avian infectious laryngotracheitis in backyard chickens: two cases. Avian Dis 2017: 61:255-260.