Conference 20, Case 1
Signalment:
Adult, castrated male, wild donkey (Equus africanus asinus)
History:
Seven wild donkeys died over the course of a week following a history of respiratory signs. Two of the donkeys were submitted for postmortem examination and diagnostic workup. Tissues from one of these animals are included here.
Gross Pathology:
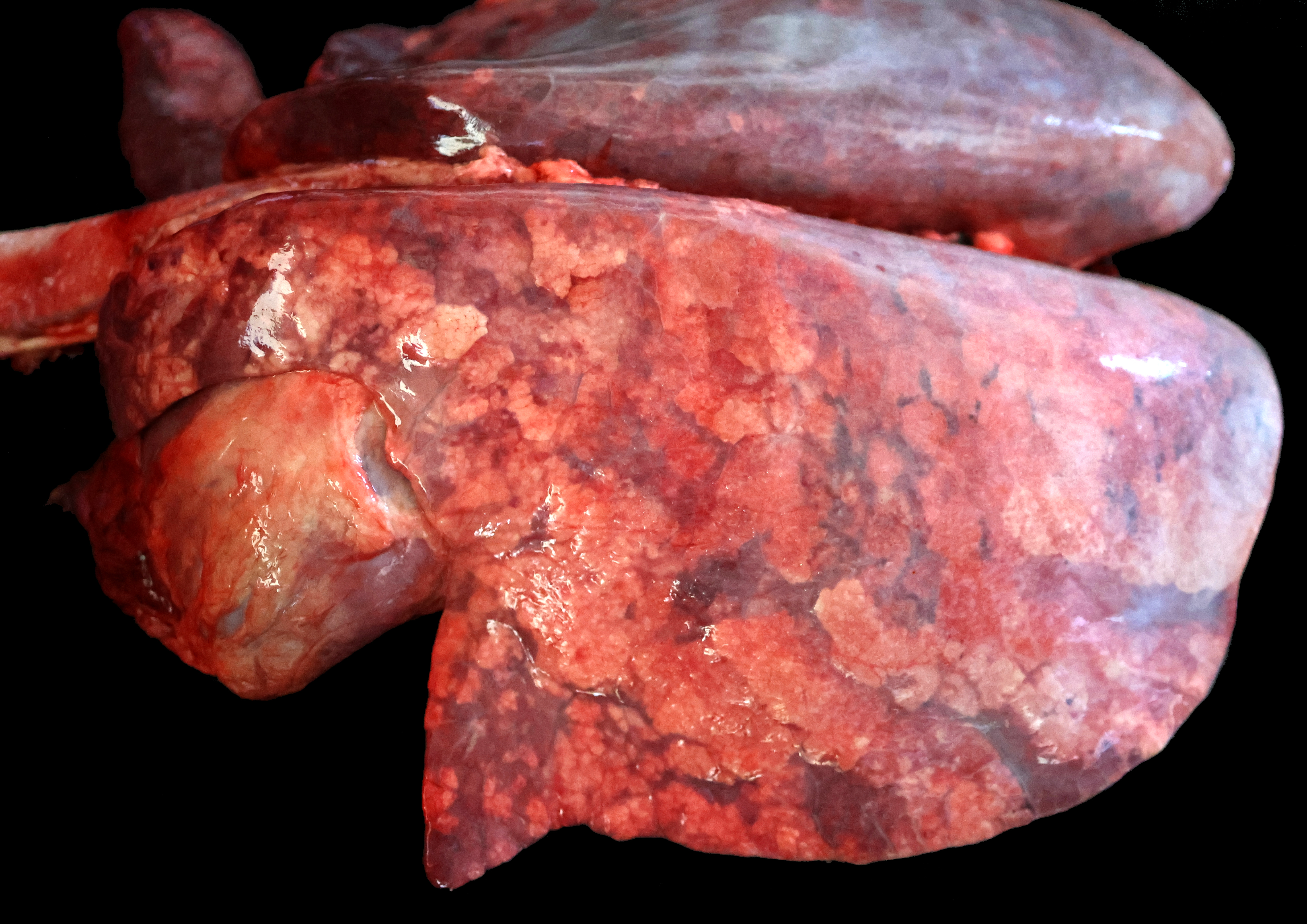
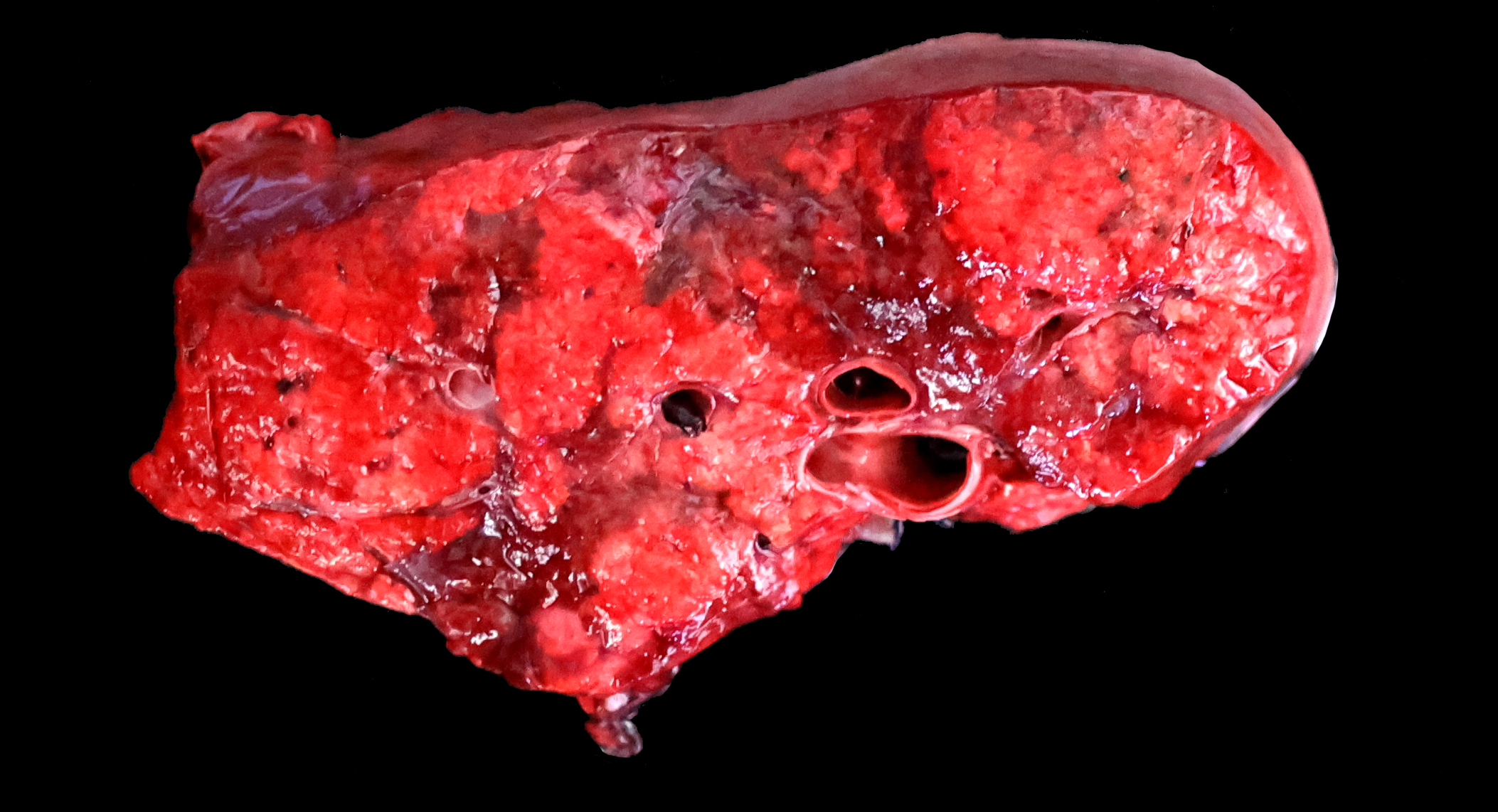
The lungs are diffusely swollen with a lobular pattern. On the cut surface, red areas of pulmonary consolidation are seen in a lobular pattern, separated by unaffected lung lobules. Moderate amounts of thick tan exudate are noted in the airways. The trachea contains moderate amounts of stable foam. The tracheobronchial lymph nodes are enlarged.
Laboratory Results:
Virology: Influenza A virus was detected in nasal and bronchial swabs, as well as in the lung. Whole genome sequencing successfully recovered 16% (2.1 kb) of the entire equine influenza virus genome. The strain was highly similar (>99.7% nucleotide identity) to multiple strains (H3N8) from the United States and Chile.
-Equine herpesvirus 1 and 4 (EHV-1 and EHV-4) were not detected by PCR.
-No viral particles were detected by virus isolation (nasal and bronchial swabs and lung).
Bacteriology: Mixed flora was isolated form the lung.
Electron microscopy: No viral particles were detected in the lung.
Contributor's Comment:
Equine influenza virus A (EIV) is an enveloped, single-segmented, negative-sense RNA virus belonging to the Orthomyxoviridae family and Influenza A genus.8 In equids, two main subtypes have been described: H7N7 (extinct since 1979) and H3N8.1 H3N8 infects and causes disease in horses, donkeys, dogs, pigs, and camels, and there is evidence that it occasionally infects humans.7,14,15 Outbreaks of H3N8 in donkeys have been identified in several regions and countries, including the United States, Brazil, Chile, Europe, China, Senegal, Pakistan, Turkey, West and Central Africa, Egypt, and India.1 Only a few countries such as New Zealand and Iceland are free of equine influenza.3 H3N8 strains have been considered emerging in donkeys in Colorado, California, and China.1,14 Outbreaks of H3N8 show no age predilection; morbidity is often high (81%) and mortality is low, although it can reach up to 20%, particularly in donkeys.8,11,14 Donkeys are more susceptible to EIV infection compared to horses,3,12 possibly due to a higher propensity for bacterial bronchopneumonia or recurrent coinfections with Dictyocaulus arnfieldii.3
EIV is extremely contagious and transmission occurs by direct contact with infected animals or fomites, as well as via aerosols. It can effectively travel through the air over distances of up to 1-2 km.12 The incubation period ranges from 1 to 3 days.12 EIV primarily infects respiratory tract epithelial cells through receptor-mediated endocytosis. The spike glycoprotein, HA, attaches to sialic acid receptors on the host cell surface.10,12 Viral replication leads to epithelial cell death, mostly through apoptosis, and subsequent chemotaxis of immune cells, causing inflammation and reducing mucociliary clearance.10
Uncomplicated cases of EIV infection are typically mild and self-limiting. In equids, clinical signs include nasal serous or mucopurulent discharge, dry cough, pyrexia, loss of appetite, weakness, poor performance, and hyperemia of nasal and conjunctival mucosae.9 Clinical signs are generally milder in horses and mules compared to donkeys.12 Fatal cases may occur with complications from pulmonary bacterial infections such as those caused by Streptococcus equi subsp. zooepidemicus, Streptococcus spp., Klebsiella pneumoniae, and Actinobacillus spp.5,6,11,14 Rare complications include myocarditis, chronic obstructive pulmonary diseases, and encephalitis.9,12
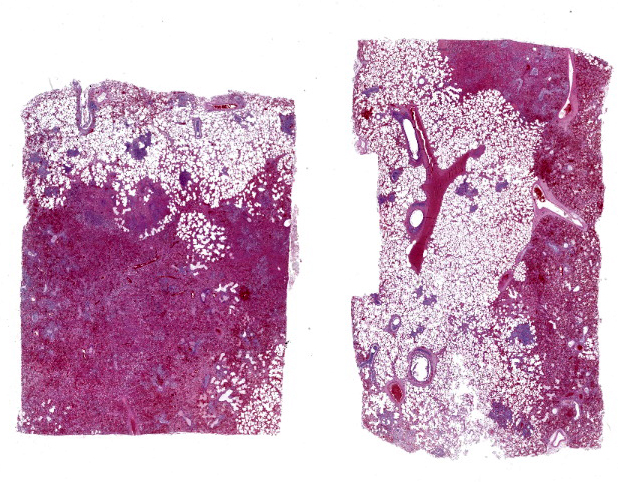
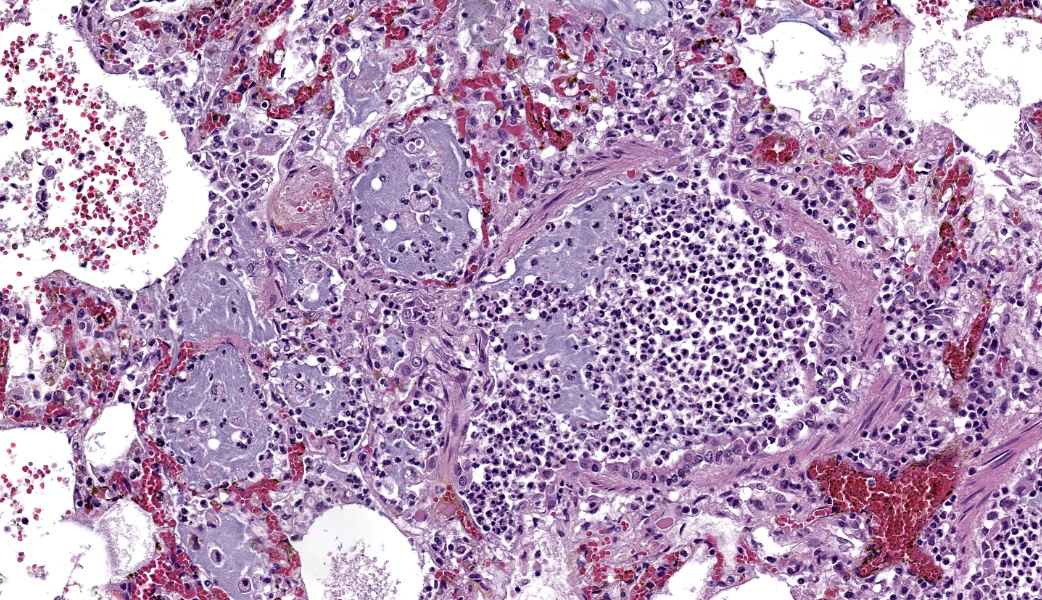
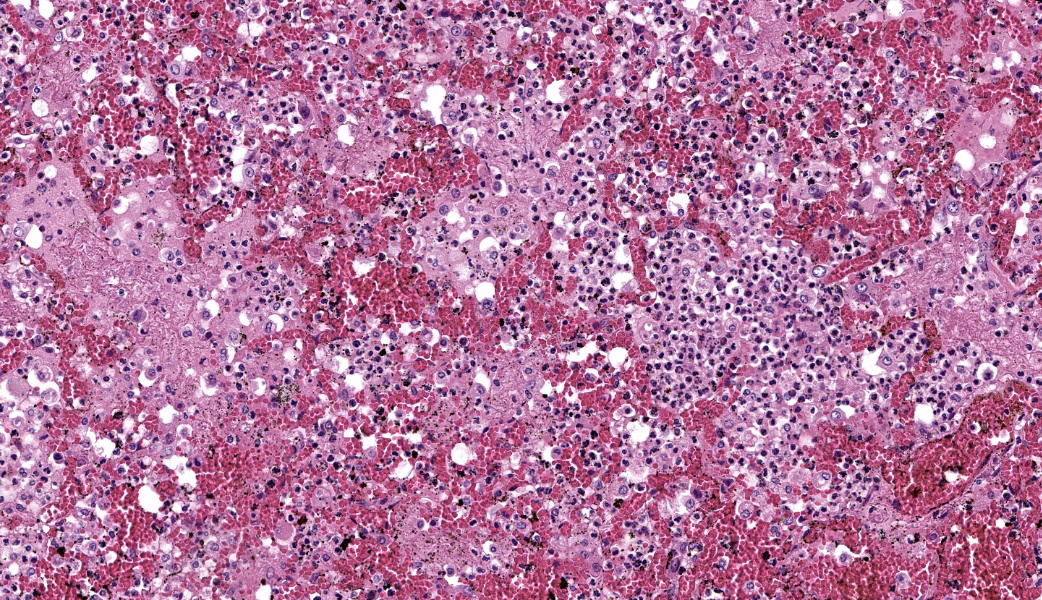
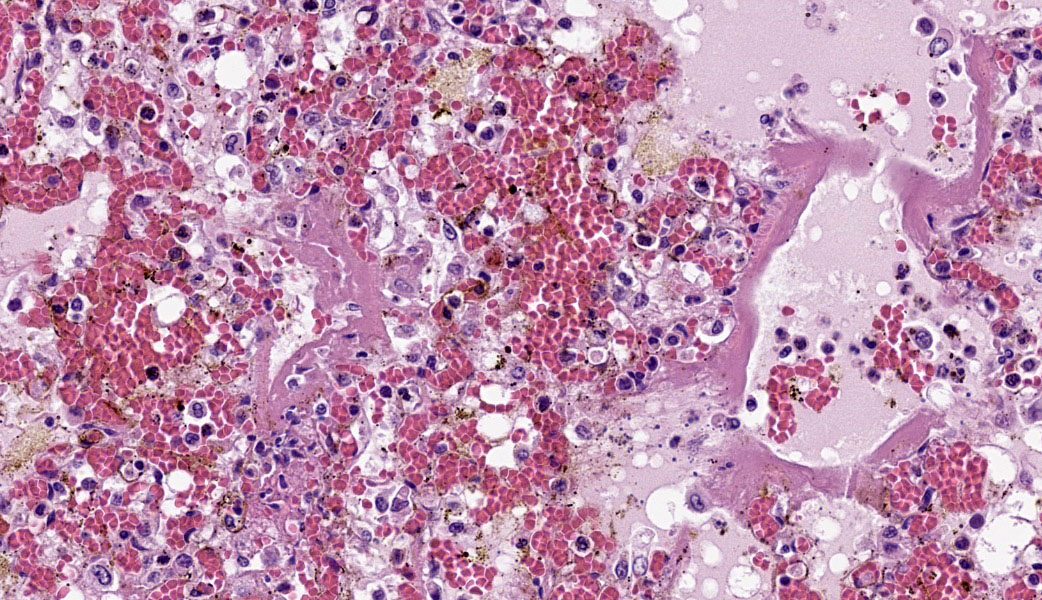
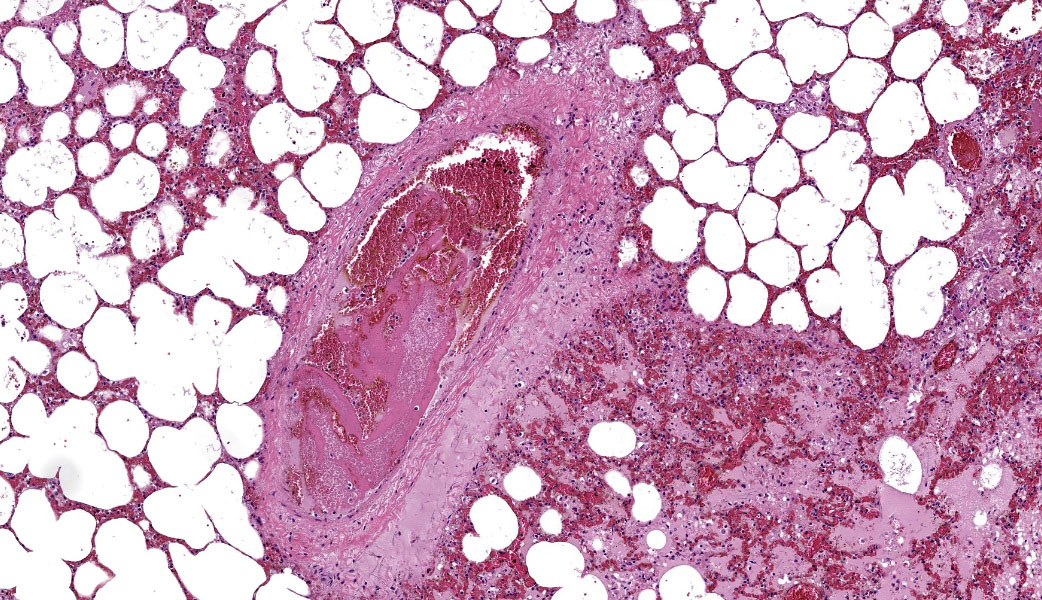
Typical pathological changes in equids with viral pneumonia include pulmonary consolidation in a lobular pattern separated by unaffected or overinflated lung lobules, or less commonly, a diffuse pattern of pneumonia.1 EIV infections complicated by secondary bacterial infections are characterized by fibrinosuppurative exudate and pulmonary consolidation, mostly restricted to the cranioventral pulmonary lobes (bronchopneumonia).1,12,13 Histopathologically, acute stages typically reveal rhinitis and tracheitis with epithelial necrosis and infiltrates of lymphocytes in the lamina propria. Subacute to chronic lesions consist of epithelial hyperplasia and squamous metaplasia.6,11 Affected lungs exhibit bronchointerstitial pneumonia with hyaline membranes in the alveoli, type II pneumocyte hyperplasia, and necrotizing bronchitis/bronchiolitis. Secondary bacterial bronchopneumonia may also develop.4,6,12
Other viral upper respiratory infections such as Equine Herpesvirus 1 (EHV-1) and Equine Herpesvirus 4 (EHV-4) produce similar lesions and should be considered as differential diagnoses in donkeys.2 Confirmation of the etiology in cases of EIV infection should involve fluorescent antibody testing (FAT), immunohistochemistry (IHC), virus isolation, molecular testing, and/or partial genetic and phylogenetic analysis of the virus.10
Contributing Institution:
California Animal Health and Food Safety Lab
105 W Central Ave, San Bernardino, CA 92408, USA.
https://cahfs.vetmed.ucdavis.edu/
JPC Morphologic Diagnosis:
Lung: Pneumonia, bronchointerstitial, necrotizing and fibrinosuppurative, acute, multifocal to coalescing, marked, with thrombosis, edema, and hyaline membranes.
JPC Comment:
The 20th conference of the 2025-2026 Wednesday Slide Conference was moderated by the wonderful and undisputed Queen of Virology, Dr. Patty Pesavento, from the University of California, Davis. This first case was a fantastic example of equine influenza with a secondary bacterial infection, and the contributor provided a fantastic writeup for this entity.
Determining differentials in a case of viral pneumonia in any species requires a working knowledge of viral pathogenesis and target cells to refine the list of potential offenders. In this case, the most striking hallmarks for influenza virus include the end-airway epithelial necrosis and patchy areas of hemorrhage. Influenza is both endotheliotropic and epitheliotropic and, while it affects epithelial cells in the upper airways, it has unique tropism for the type I and type II pneumocytes of the alveoli as well. It causes significant destruction to end-airways as a result. This key difference can help separate an influenza infection from other pneumotropic viruses, such as calicivirus in cats. (Feline calicivirus infection of the lung can look nearly identical to an H5N1 influenza virus infection in a cat, but the state of the end airways can help clue in the pathologist to which virus is most likely and can assist in decision-making for next testing steps. The more virulent strains of feline calicivirus can cause severe interstitial pneumonia, but they do not cause necrosis of end airways the way that influenza does.)
Grossly, an influenza-infected lung has a ?checkerboard? appearance with random, alternating areas of dark red atelectasis and light pink, aerated lung. This pattern manifests because influenza does not uniformly infect every cell it comes into contact with due to varied densities of sialic acid receptors on the host cells. Dark red to purple areas of consolidation represent collapsed alveoli whose airway has been clogged by exudate and which can no longer pass air through to the alveoli.. These solidified, dark red sections contrast sharply with the lighter-colored, air-filled alveoli.
As stated by the contributor, donkeys are more susceptible to equine influenza virus than horses, although both species primarily develop respiratory disease with influenza.1 This is true in pigs and humans, as well, where the lung is the main target of influenza.
In cats, however, the primary presentation of influenza, especially H5N1, is encephalitis. Cats are also susceptible to cerebral infarcts from influenza targeting vascular endothelium. Swine can also be infected with neurotropic strains of influenza A, including H5N1. One of the main mechanisms for infection with H5N1 influenza in cats is consumption of contaminated cow?s milk.9
In cattle, the primary targets for influenza are the mammary glands due to high tropism for and concentration of the virus in bovine mammary gland epithelium with subsequent shedding of the virus into milk.9 It is unclear if this is true in other species, but it has been definitively demonstrated that, in humans, the virus does not target the mammary glands.9 Additionally, modern pasteurization and homogenization methods used in most commercial dairies completely kills any influenza virus that may be present in cow milk, so milk from dairies that use these methods is still very much safe for human consumption.9 In birds, the primary cellular targets for influenza, especially highly pathogenic avian influenza, are the brain and pancreas. Avian influenza was discussed more in depth in this year?s Conference #17, Case 2.
All participants considered the bacterial infection to be secondary and due to aerogenous introduction of bacteria into an already virus-infected lung. This ruled out Streptococcus spp, inculding S. equi or S. zooepidemicus, as contributing to the bacterial infection, as these spread hematogenously.
References:
- Ahearne MM, Pentzke-Lemus LL, Romano AM, Larsen ED, Watson AM, O'Fallon EA, Landolt GA. Disease progression, pathologic, and virologic findings of an equine influenza outbreak in rescue donkeys. J Vet Intern Med. 2022; 36(6):2230-2237.
- Boyle AG, Timoney JF, Newton JR, Hines MT, Waller AS, Buchanan BR. Streptococcus equi Infections in Horses: Guidelines for Treatment, Control, and Prevention of Strangles-Revised Consensus Statement. J Vet Intern Med. 2018;32(2):633-647.
- Camara RJF, Bueno BL, Resende CF, Balasuriya UBR, Sakamoto SM, Reis JKPD. Viral Diseases that Affect Donkeys and Mules. Animals (Basel). 2020;25;10(12):2203.
- Carvallo FR, Stevenson VB. Interstitial pneumonia and diffuse alveolar damage in domestic animals. Vet Pathol. 2022;59(4):586-601.
- Carvallo FR, Uzal FA, Diab SS, Hill AE, Arthur RM. Retrospective study of fatal pneumonia in racehorses. J Vet Diagn Invest. 2017;29(4):450-456.
- Chambers TM. Equine Influenza. Cold Spring Harb Perspect Med. 2022; 4;12(1):a038331.
- Crawford PC, Dubovi EJ, Castleman WL, Stephenson I, Gibbs EP, Chen L, Smith C, Hill RC, Ferro P, Pompey J, Bright RA, Medina MJ, Johnson CM, Olsen CW, Cox NJ, Klimov AI, Katz JM, Donis RO. Transmission of equine influenza virus to dogs. Science. 2005; 21;310(5747):482-5.
- Dionisio L, Medeiros F, Pequito M, Faustino-Rocha AI. Equine influenza: a comprehensive review from etiology to treatment. Anim Health Res Rev. 2021; 22(1):56-71.
- Landolt GA. Equine influenza virus. Vet Clin North Am Equine Pract. 2014; 30(3):507-22.
- Rahman A, Uzal FA, Hassebroek AM, Carvallo FR. Retrospective study of pneumonia in non-racing horses in California. J Vet Diagn Invest. 2022; 34(4):587-593.
- Singh RK, Dhama K, Karthik K, Khandia R, Munjal A, Khurana SK, Chakraborty S, Malik YS, Virmani N, Singh R, Tripathi BN, Munir M, van der Kolk JH. A Comprehensive Review on Equine Influenza Virus: Etiology, Epidemiology, Pathobiology, Advances in Developing Diagnostics, Vaccines, and Control Strategies. Front Microbiol. 2018; 6(9):1941.
- Timoney PJ. Equine influenza. Comp Immunol Microbiol Infect Dis. 1996; 19(3):205-11.
- Yang H, Xiao Y, Meng F, Sun F, Chen M, Cheng Z, Chen Y, Liu S, Chen H. Emergence of H3N8 equine influenza virus in donkeys in China in 2017. Vet Microbiol. 2018; 214:1-6.
- Xie T, Anderson BD, Daramragchaa U, Chuluunbaatar M, Gray GC. A Review of Evidence that Equine Influenza Viruses Are Zoonotic. Pathogens. 2016; 12;5(3):50.