CASE 4: 8002353 4001 33 ABL (4136107-00)
Signalment:
14-month-old male Beagle dog (Canis familiaris)
History:
The dog was part of a 13-week toxicity study. On Day 68 of the study, the animal appeared to be in some discomfort, voided a small amount of red urine in a discontinuous stream, and then exhibited transient tremors and adopted the "prayer stance" suggestive of abdominal pain. The animal was subsequently removed from study and humanely euthanized.
Gross Pathology:
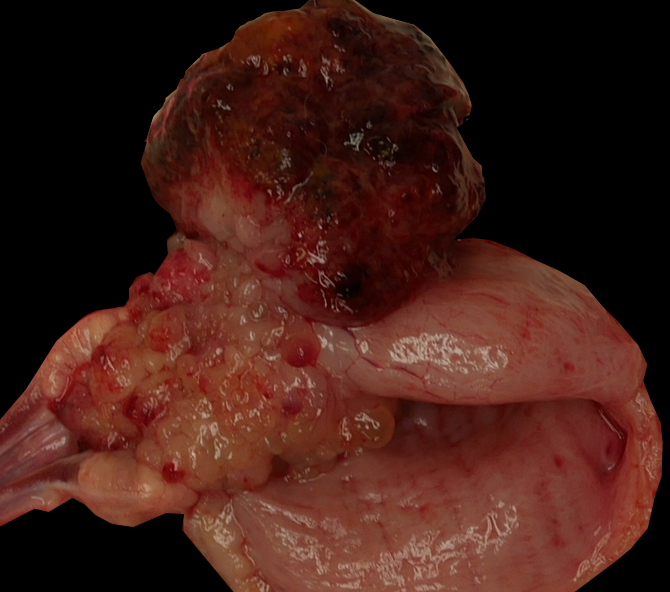
Expanding and partially effacing the urinary bladder mucosa in the area of the trigone and the prostate was a firm 5.5x4x3 cm, variably pale tan to dark red, multilobular mass that protruded and partially obstructed the neck of the bladder and urethral lumen. The bladder lumen was dilated, and the mucosa of the adjacent bladder and urethra were discolored dark red and were mildly gelatinous (edematous). The left kidney was mildly enlarged, and the left renal pelvis and ureter were moderately dilated (hydronephrosis and hydroureter).
Laboratory results:
Prior to study initiation, urinalysis, hematology, and serum chemistry was performed. Notable results on urinalysis consisted of 3+ to 4+ blood and protein, with abundant red blood cells and fewer white blood cells present in urine sediment. On hematology and serum chemistry were minimally decreased red blood cell parameters (red blood cell count, hemoglobin) and evidence of inflammation consisting of mildly elevated total white blood cells attributable to increases in neutrophils and monocytes, and decreased albumin to globulin ratio attributable to both increased serum globulins and decreased serum albumin. These analyses were repeated on Day 68 of the study and the results were similar to the pre-study timepoint.
Microscopic description:
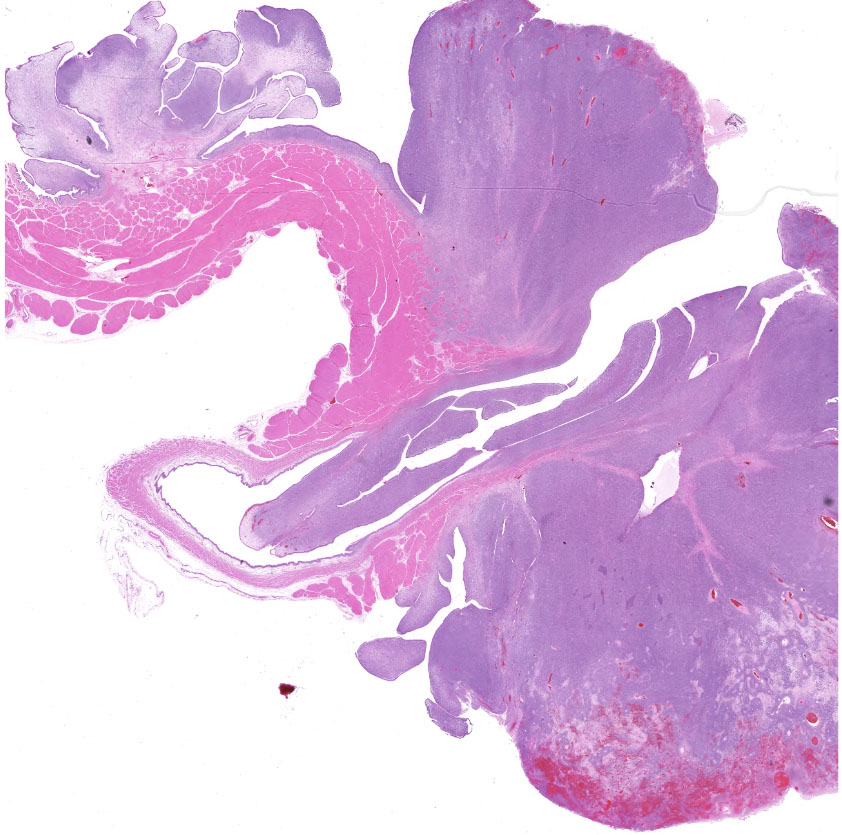
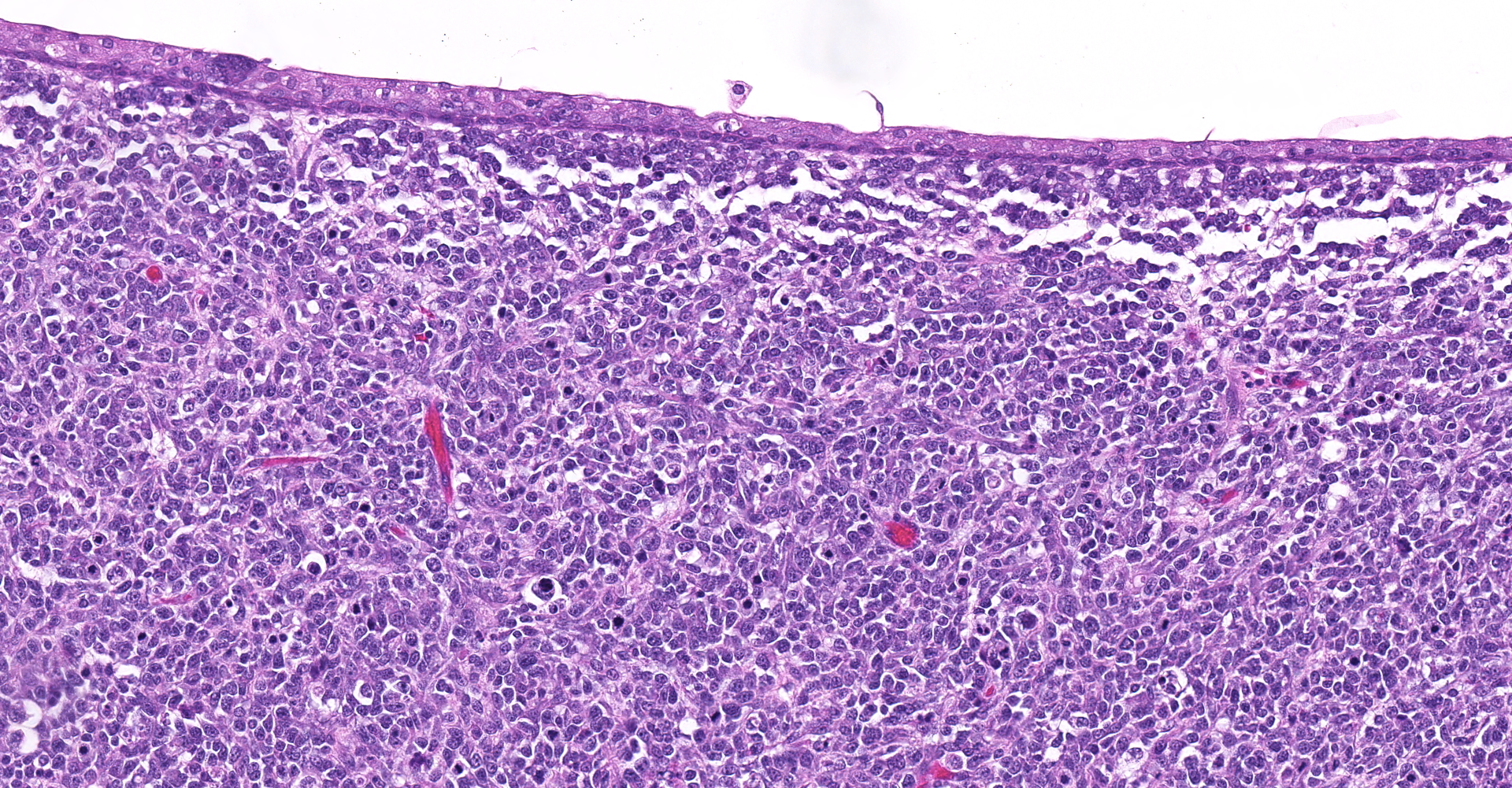
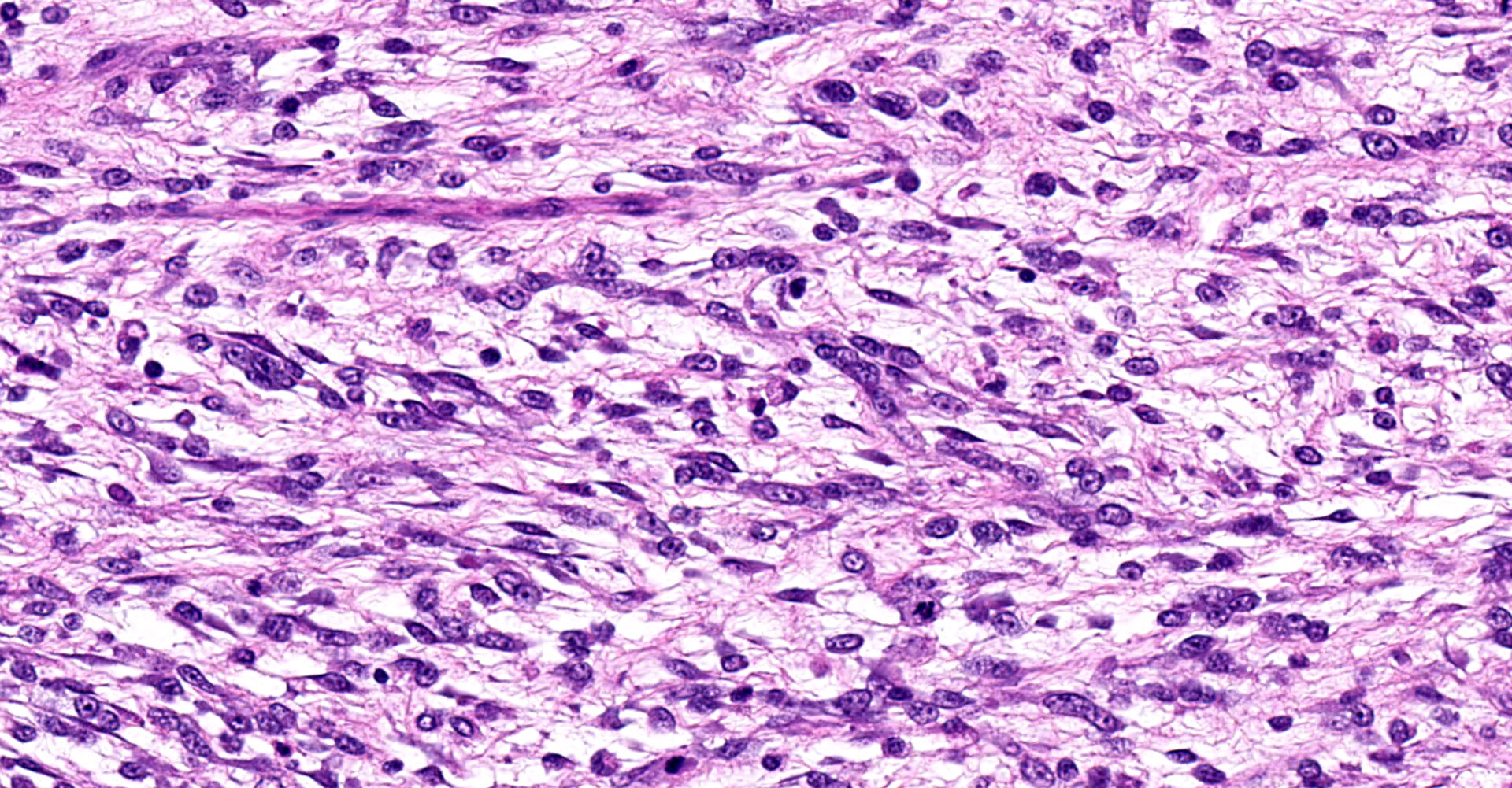
Urinary bladder: Partially effacing the wall of the bladder in the area of the trigone, elevating the overlying urothelium, and extending into the bladder and partially obstructing the urethral lumen, was a poorly demarcated, unencapsulated neoplasm composed of cells exhibiting various morphologies. The majority were small, polygonal, undifferentiated cells with minimal cytoplasm, compact round nuclei with moderate mitotic rate (7/10 HPF), in nests and packets separated by fine fibrovascular stroma. In some areas, particularly in areas underlying the urothelial mucosa, the neoplasm was composed of sheets of sparse pleiomorphic spindle to stellate cells with low mitotic rate (<1/10/HPF) embedded in abundant myxomatous matrix. Elongated, rarely multinucleated cells with large round to oval vesicular nuclei and visible cytoplasmic striations ("strap cells") were occasionally present in these areas. Minimal mixed inflammatory cell infiltrates were scattered throughout the section. The superficial aspect of the tumor that was protruding into the bladder lumen was necrotic and hemorrhagic, and the overlying urothelium was absent (ulcerated). In other areas the urothelium was variably hyperplastic or vacuolated.
Contributor's morphologic diagnosis:
Urinary bladder: Botryoid rhabdomyosarcoma
Contributor's comment:
This is the classic presentation for spontaneous canine botryoid rhabdomyosarcoma: a multilobular "grape-like" mass at the trigone of the bladder in a young dog. These tumors are rare overall (<0.5% of all bladder tumors) but are most commonly reported in dogs less than two years old with some predilection for larger breeds such as Saint Bernards. However, sporadic reports of occurrence in small breeds and in other species (horse) are also present in the literature.15 Other types of rhabdomyosarcoma include embryonal, alveolar, or pleiomorphic. The clinical significance of the different histologic types in the dog is unknown; in humans the alveolar variant is associated with poor prognosis.2
Although to the authors knowledge botryoid rhabdomyosarcoma has not been reported in the beagle, there is one case report of a beagle with cardiac alveolar rhadbdomyoma.
Histologically, the cells comprising botryoid rhabdomyosarcoma resemble variably differentiated myoblasts (small polygonal cells) to skeletal myocytes, and the presence of well-differentiated strap cells containing cross-striations is characteristic. Consistent with a proposed origin of remnant embryonic myoblastic cells, these tumors may be variably positive for immunohistochemistry markers of mesenchymal cells and skeletal muscle based on the degree of differentiation (vimentin, desmin, S100, myoglobin, myogenin, or MyoD immunohistochemistry and PTAH special stain), and are negative for markers of epithelial cells or smooth muscle (cytokeratin, ?SMA).8 This case was strongly desmin positive, multifocally PTAH positive only in more differentiated areas of the tumor, inconclusive for myoglobin, and ?SMA negative.
Metastasis has been reported with botryoid rhabdomyosarcomas but is generally considered rare with this tumor type. However, given the location of the tumor surgical resection is often challenging and local complications such as urinary obstruction are common clinical sequela.1,13 Other common clinical manifestations, some of which were present in this dog, are cystitis and hydronephrosis/hydroureter, as well as peripheral fibromatosis and hypertrophic osteopathy.16
The macroscopic, multinodular appearance of a mass in the trigone of the bladder of a young dog is highly suggestive of botryoid rhabdomyosarcoma. Differential diagnoses for a mass in this location may include transitional cell carcinoma, leiomyosarcoma, other primary or secondary tumors reported in the bladder of dogs (e.g. histiocytic sarcoma, lymphoma, osteosarcoma, hemangiosarcoma, etc.), inflammatory cystitis, or extension of prostatic neoplasia, e.g. squamous cell carcinoma.6,17 Unless the rhabdomyosarcoma is highly undifferentiated, each of these has histologically distinct features.
Contributing Institution:
Takeda Pharmaceuticals International Co.
35 Landsdowne St. Cambridge, MA 02139
JPC diagnosis:
Urinary bladder: Botryoid rhabdomyosarcoma.
JPC comment:
Canine rhabdomyosarcoma is most often found in laryngeal and cardiac tissues, but the botryoid variant arises most commonly in the trigone of the urinary bladder and urethra. However, it is also rarely reported to arise in the uterus and vagina.
In human medicine, an additional differential diagnosis is fibroepithelial polyp. A recent case illustrated a growth in the urinary bladder with prominent botryoid features and an embryonal rhabdomyosarcoma pattern, though lacking nuclear or cellular atypia. Sequencing the affected tissue revealed two pathogenic mutations in the dicer1 ribonuclease III (DICER1) gene. While we rarely sequence tissues for routine diagnosis, it remains possible that this mutation or similar may cause pathology that mimics botryoid rhabdomyosarcoma.5
Rhabdomyosarcoma arises in many species, in a variety of tissues. In horses, botryoid rhabdomyosarcoma has been reported in the uterus and urinary bladder, with embryonal rhabdomyosarcomas arising most often in the tongue and limb muscles.3 Rhabdomyosarcoma has also been reported in cats10, sheep4, goats9, mice (rare natural occurrences; frequent in experimental models)12, pigs4, cattle4, and brook trout.14
As correctly identified by the resident, the moderator emphasized that the correct terminology is now "urothelial cell", which replaces "transitional cell" when referencing the urinary epithelial cell layer.
References:
- Bae IH: Genitourinary rhabdomyosarcoma with systemic metastasis in a young dog. Vet Pathol 44(4):518-20, 2007
- Caserto B: A Comparative Review of Canine and Human Rhabdomyosarcoma with Emphasis on Classification and Pathogenesis. Vet Pathol 55(5):806-26,2013
3. Castleman WL, Toplon DE, Clark CK. Rhabdomyosarcoma in 8 horses. Vet Pathol. 2011; 48(6):1144-50.
- Cooper BJ, Valentine BA. Muscle and Tendon. In: Maxie MG ed. Jubb, Kennedy, and Palmer's Pathology of Domestic Animals, Vol. 1, 6th Ed. St. Louis, MO: Elsevier. 2016:241-245.
- Eckstein M, Agaimy A, Woenckhaus J, et al. DICER1 mutation-positive Giant Botryoid Fibroepithelial polyp of the urinary bladder mimicking embryonal rhabdomyosarcoma. Human Pathology. 2019;84:1-7.
- Griffin MA: Lower Urinary Tract Neoplasia. Vet Sci 5(4):E96, 2018
- Jubb KVF; Kennedy PC; Palmer N, Eds.: Pathology of Domestic Animals, 4th Edition, Academic Press, Vol. 2:536-8, 1993.
- Kobayashi M et al: Expression of myogenic regulating factors, Myogenin and MyoD, in two canine botryoid rhabdomyosarcomas. Vet Pathol 41(3):275-7. 2004
9. Lohr CV. One hundred two tumors in 100 goats (1987?2011). Vet Pathol. 2012; 50(4): 668-675.
- Minkus G, Hillemanns M. Botryoid-type embryonal rhabdomyosarcoma of liver in a young cat. Vet. Pathol. 1997;34:618-621.
- Pletcher JM, and Dalton L: Botryoid rhabdomyosarcoma in the urinary bladder of a dog. Vet Pathol 18(5): 695-7, 1981.
12. Rehg JE, Ward JM. Morphological and immunohistochemical characterization of sarcomatous tumors in wild-type and genetically engineered mice. Vet Pathol. 2012; 49(1):206-17.
- Saulnier-Troff FG et al: A technique for resection of invasive tumors involving the trigone area of the bladder in dogs: preliminary results in two dogs. Vet Surg 37(5):427-37, 2008
14. Sirri R, Bianco C, et al. Rhabdomyosarcoma of the soft tissues in an adult brook trout (Salvelinus fontinalis). J Comp Pathol. 2015; 153:190-195.
- Turnquist et al.: Botryoid rhabdomyosarcoma of the urinary bladder in a filly. J Vet Diagn Invest 5:451453, 1993
- Withers et al.: Paraneoplastic hypertrophic osteopathy in 30 dogs. Vet Comp Oncol 13(3):157?65, 2015
- Woldemeskel, M.: Primary Urinary Bladder Osteosarcoma in a Dog. J Comp Path 157(2?3): 141-4, 2017