Joint Pathology Center
Veterinary Pathology Services
Wednesday Slide Conference
2017-2018
Conference 23
April 11th, 2018
CASE I: 164361-16 (JPC 4100935)
Signalment: 3-year-old, male, castrated, Doberman pinscher (Canis familiaris), canine.
History: The patient presented to Cornell University Hospital for Animals (CUHA) Emergency Service for respiratory distress. Nine months prior, he had been evaluated for bilateral swollen carpi and radiographs revealed an aggressive bone lesion; the carpi became progressively worse leading to forelimb lameness, coughing, sneezing and epistaxis. The patient was administered diphenhydramine, 1.25 mg/kg (0.56 mg/lb), PO, q 12 h, as needed and amoxicillin, 25 mg/kg (11.36 mg/lb), PO, q 12 h, for 30 days. Cytological examination of a needle aspirate from an enlarged popliteal lymph node revealed histiocytic inflammation. The patient was administered prednisone, 0.5 mg/kg (0.22 mg/lb), PO, q 12 h for 7 days, then 0.5 mg/kg (0.22 mg/lb), PO, q 24 h for 14 days and doxycycline, 5 mg/kg (0.22 mg/lb), PO, q 12 h for 30 days. Because the carpal swelling continued to increase, the dose of prednisone was increased to 1 mg/kg (0.45 mg/lb), PO, q 12 h, at which point the respiratory signs worsened. Bronchoscopy showed inflamed, hemorrhagic bronchi, and radiographs showed progressive bilateral periosteal reaction and lytic lesions of the right and left distal radius and ulna, and a severe generalized miliary nodular lung pattern.
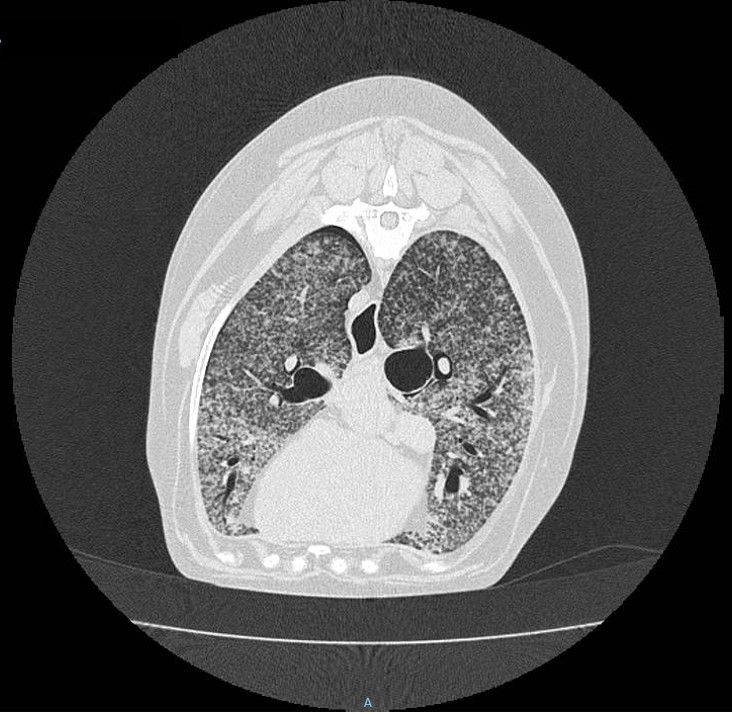
Gross Pathology: Prior to post-mortem examination, educational computed tomography (CT) scans were recorded of the head, neck and thorax and confirmed the bilateral, generalized miliary nodular pattern within the lungs. The CTs also revealed periosteal new bone formation on the calvaria.
At necropsy, the dog was in good body condition (4 out of 9, Purina scale) with mild postmortem autolysis. Bilaterally, the carpal joints were severely enlarged, with the left being worse than the right. Both carpal joints were hard with focal, soft, depressed areas. On the medial aspect of the right carpus, there was a 0.6 cm diameter full-thickness defect containing 0.1 mL of creamy, soft material (draining tract). Bilaterally, the skeletal musculature of the brachium, predominantly the triceps brachii, was less prominent (atrophy). The left popliteal lymph node was firm.
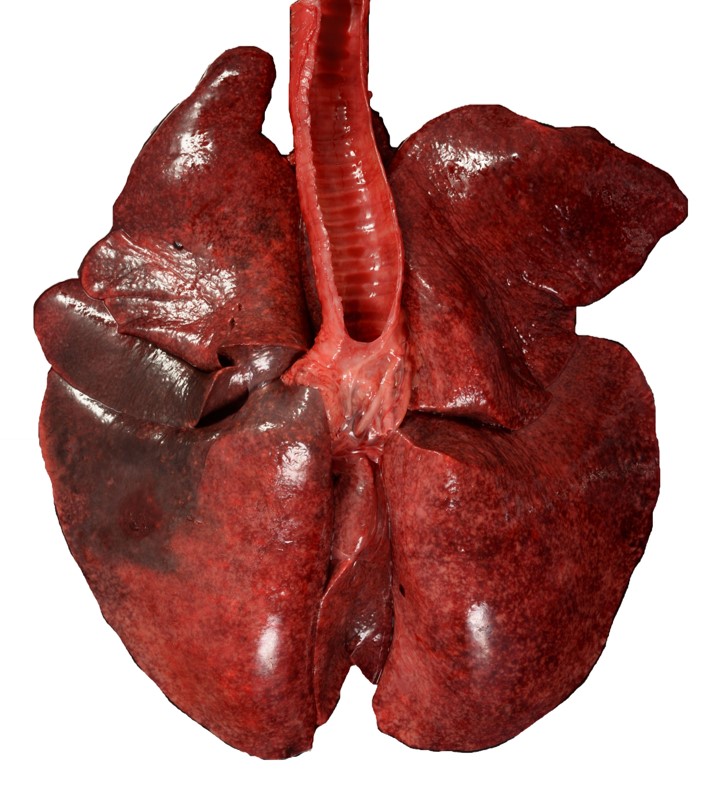
The lungs were diffusely mottled dark red and light pink, failed to collapse and mildly firm (interstitial pneumonia). Diffusely throughout the lungs were dozens of miliary, white-tan foci that extended into the parenchyma. The trachea contained approximately 2 mL of frothy, red fluid. On cut section, the large airways oozed a small amount of cloudy red to gray mucoid material (exudate).
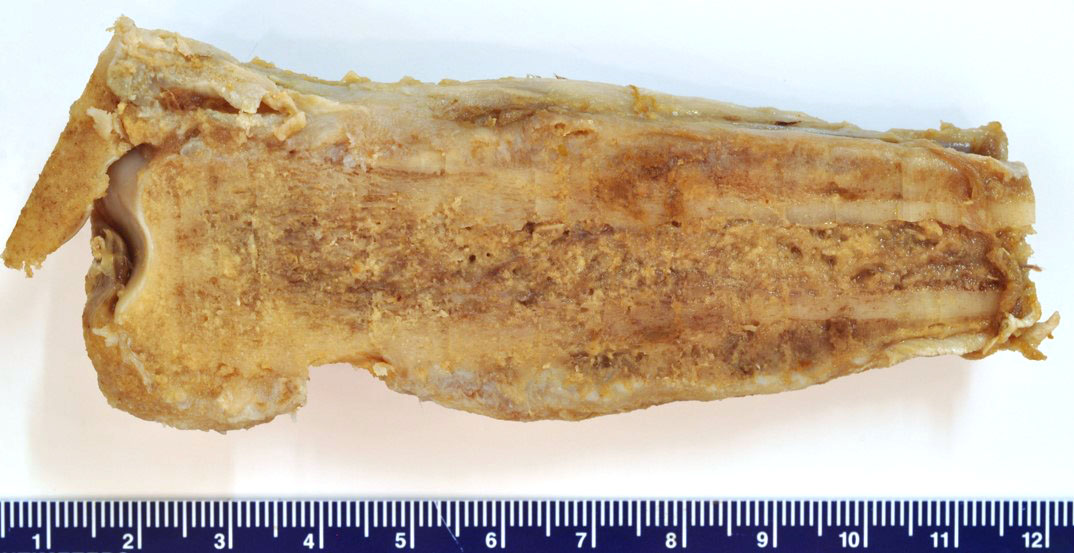
Bilaterally, the distal one-third of the radius and ulna were expanded approximately 7 cm in diameter on the left and 5 cm in diameter on the right. On cut section, the right and left carpal joints exuded approximately 1 mL of viscous, brown, cloudy fluid (suppurative synovitis and periarthritis). The periarticular fascia on the medial aspect of the right and left carpi had cystic spaces filled with similar brown, cloudy fluid. On sagittal section, the periosteum of the distal one-third of the radius, ulna and carpal bones was gradually expanded towards the carpal joint up to 1.7 cm thick. The outer cortical surface was rough and uneven.
Laboratory Results (clinical pathology, microbiology, PCR, ELISA, etc.):
Blastomyces dermatitidis was partially cultured from lung tissue (culture was not finalized due to human health risks). Blastomyces dermatitidis infection was confirmed by PCR of the lung tissue.
Microscopic Description:
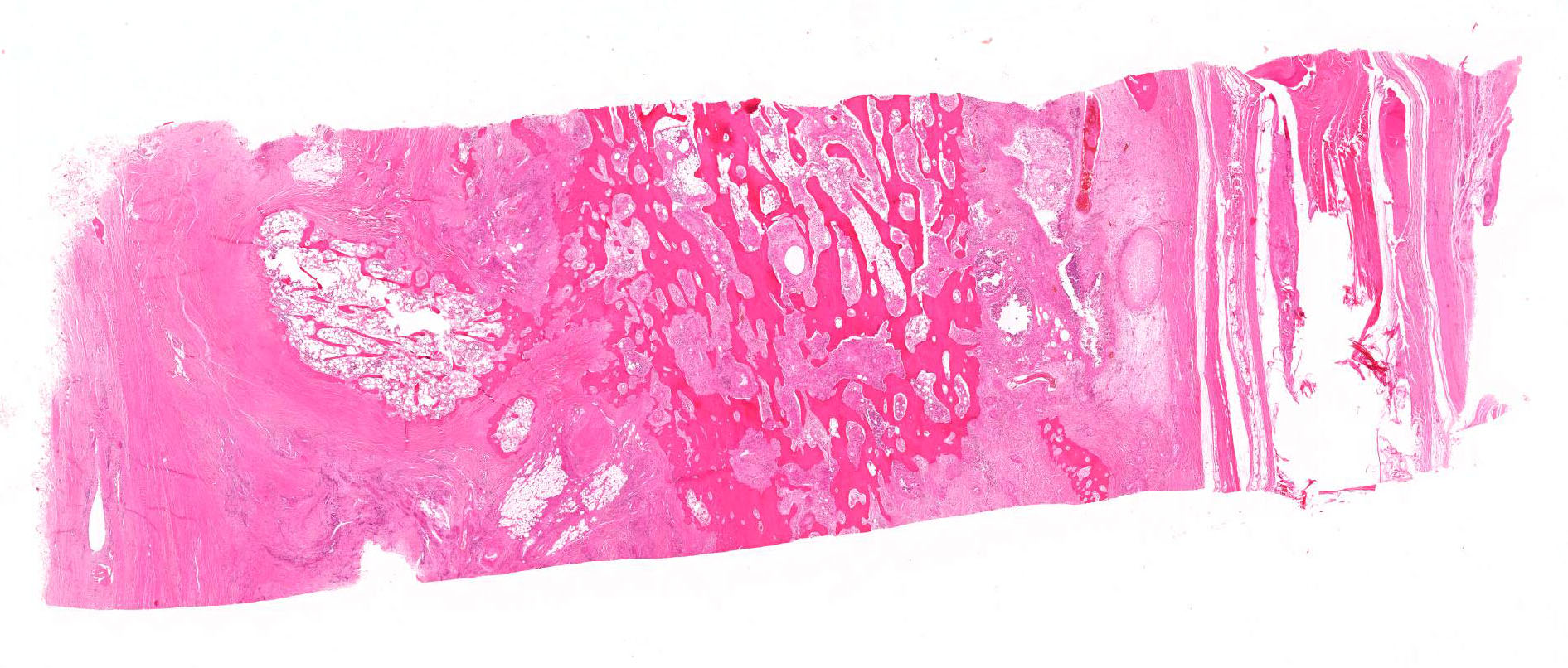
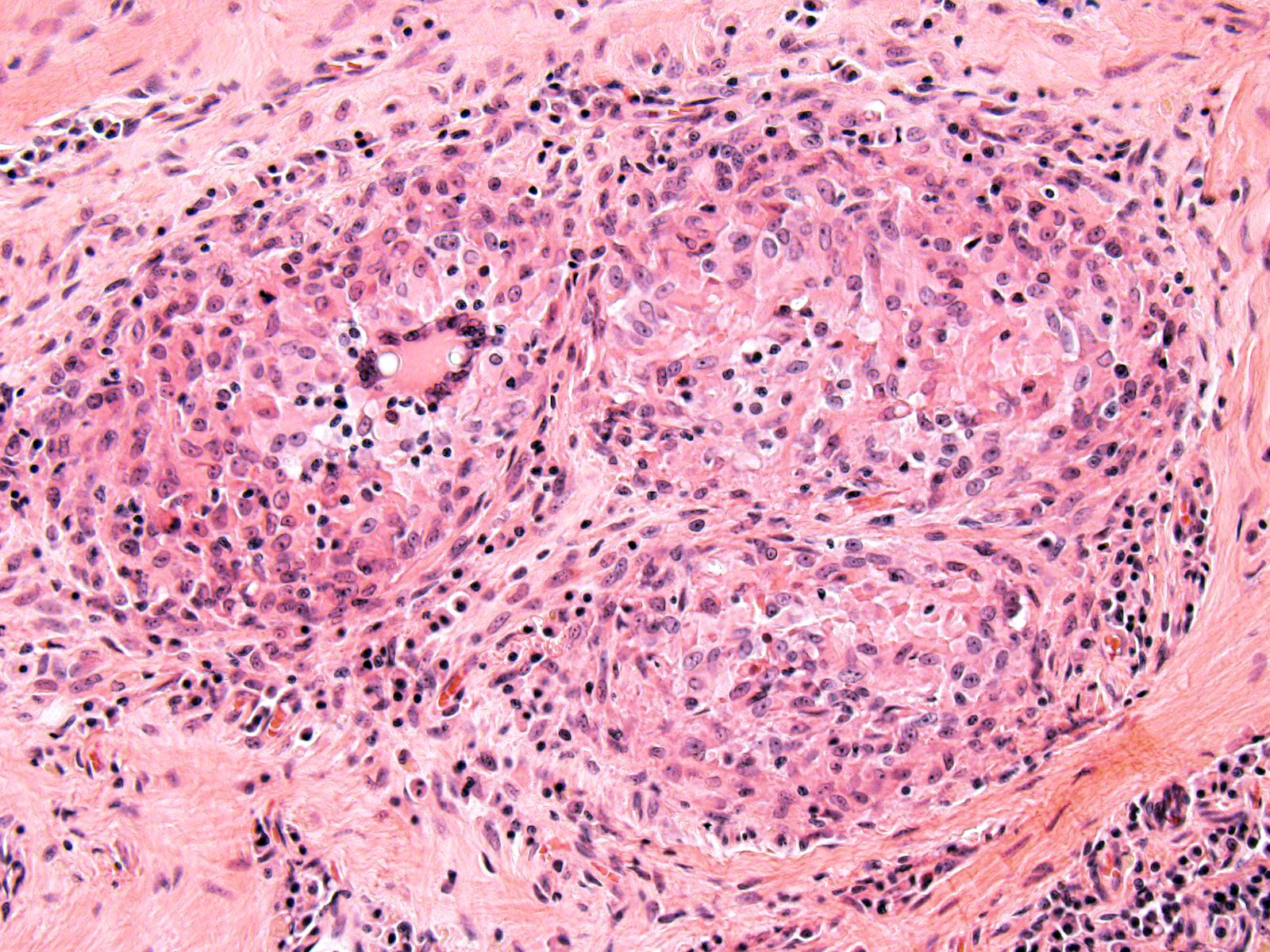
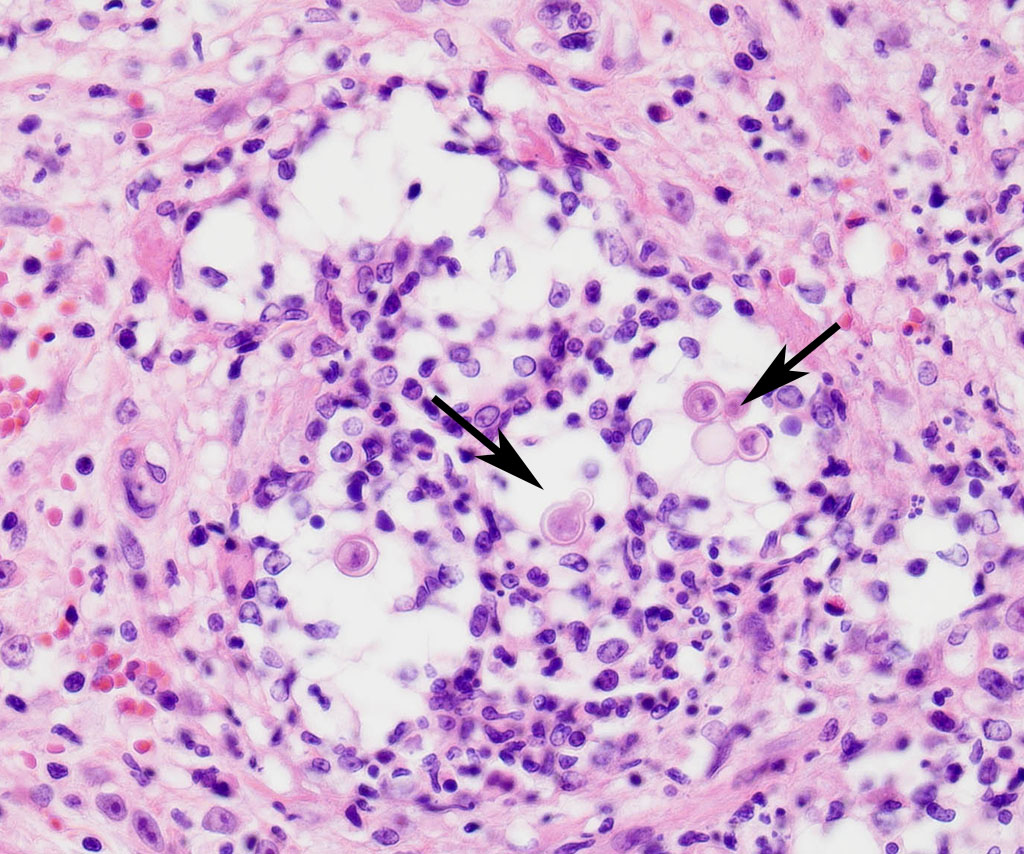
The cortical bone is expanded by numerous cavities filled with numerous macrophages together with fewer multinucleated giant cells, and rare lymphocytes (osteomyelitis). The inflammatory infiltrate varies, with multiple clusters of neutrophils and degenerated neutrophils in random areas. In multiple areas, the inflammatory cells are centered upon variable size islands of an intensely eosinophilic to magenta, acellular material, with indistinct margins (bone necrosis). The cortical bone is covered by an expanded layer of woven bone that is incompletely mineralized (periosteal reaction) and contains numerous osteocytes. The immature woven bone is lined by a single layer of plump osteoblasts (reactive). Frequently, the edges of the woven bone are scalloped, irregular with adjacent osteoclasts (Howship’s lacunae). Randomly scattered in the inflammatory infiltrate are numerous intrahistiocytic and extracellular, 8-20 μm in diameter, round yeast-like organisms with a 2-3 μm thick, clear to lightly basophilic, refractile capsule. The yeast-like organisms frequently exhibit broad-based budding consistent with Blastomyces spp. The inflammatory infiltrate extends to the bone marrow spaces and dissects through the periosteal reaction and adjacent skeletal muscles (myositis).
Contributor’s Morphologic Diagnosis:
Pyogranulomatous osteomyelitis and myositis, with bone necrosis, woven bone deposition and yeasts consistent with Blastomyces spp.
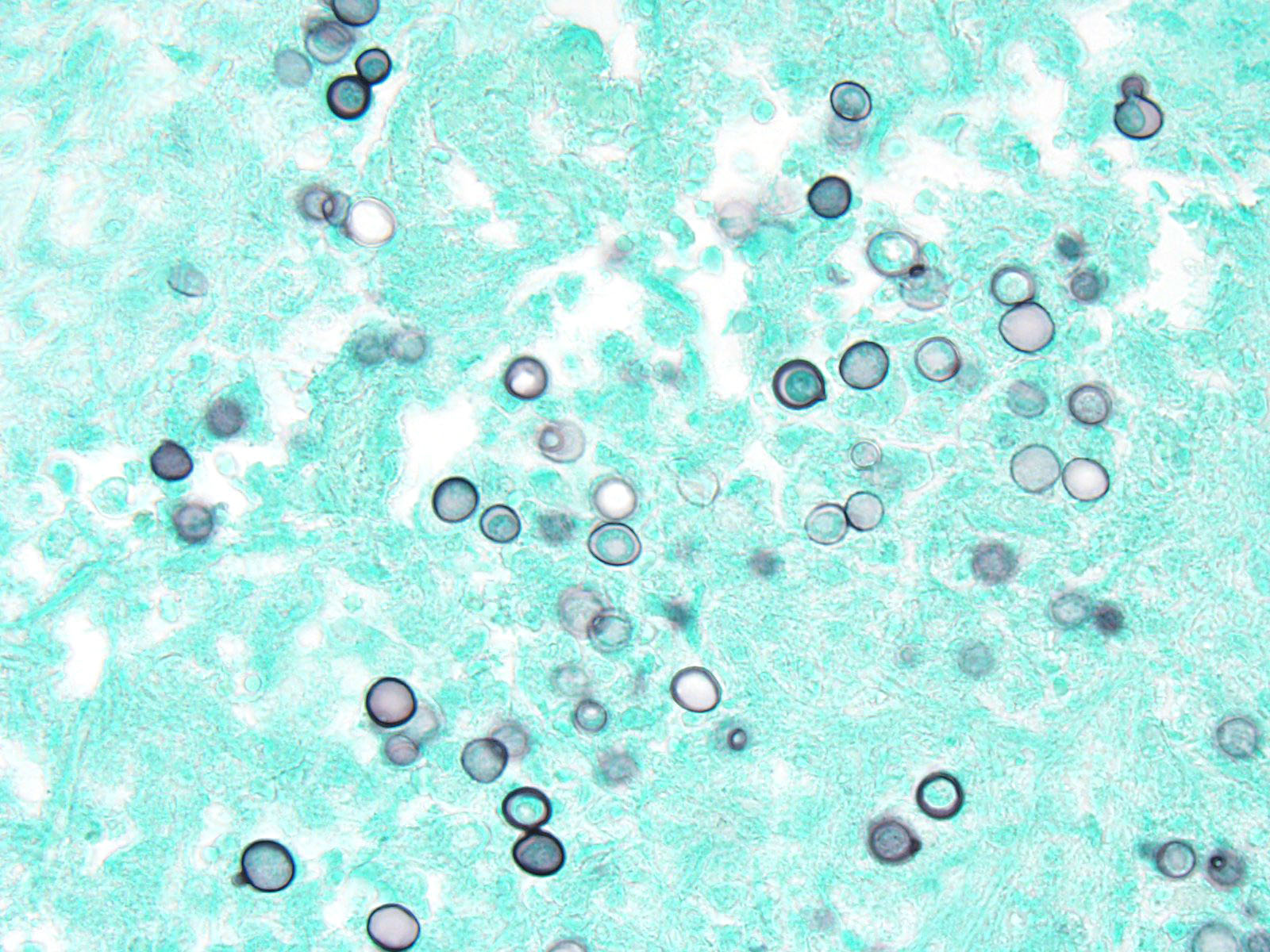
Contributor’s Comment: This is a classic case of disseminated blastomycosis caused by infection with Blastomyces dermatitidis with severe lung and skeletal involvement. Additionally, histopathology examination of the heart revealed severe, multifocal, chronic degeneration and necrosis of cardiomyocytes with dystrophic mineralization, suggestive of hypoxia attributable to the severe pneumonia. Histopathology examination of decalcified sections of distal radius/ulna and calvaria revealed a severe, chronic osteomyelitis with large numbers of yeast organisms, consistent with disseminated blastomycosis. Histochemical staining of lung sections using periodic acid-Schiff and Grocott-Gomori’s methenamine silver further highlighted the yeast organisms.
Although the classic presentation for fungal pneumonia on radiographs is a generalized, random, miliary nodular pattern, blastomycosis can have various presentations ranging from multiple pulmonary nodules, patchy or lobar lung consolidation (alveolar pattern), to a solitary pulmonary mass.3 Histopathology ruled out other potential causes of pulmonary nodules, such as neoplasia and infection with other mycoses including Coccidioides immitis, Cryptococcus neoformans, and Histoplasma capsulatum.
Blastomycosis is one of the most common systemic mycotic infection of dogs that live in endemic areas including the Ohio and Missouri river valleys, the southern Great Lakes, and southern mid-Atlantic states.4 In the state of New York, the incidence of blastomycosis in dogs has increased over the past 20 years and is endemic in the Adirondacks where this dog lived.5 Blastomycosis occurs in dogs and humans and is rare in other animals. Young, male, intact dogs living in endemic areas are at an increased risk of becoming infected.5 Sporting dogs such as Labrador and Golden retrievers and Doberman Pinschers are more frequently affected.6,7
Blastomyces dermatitidis is a saprophytic fungus found in moist acidic or sandy soil, with high organic content.6 It is a thermally dimorphic fungus, that forms septated mycelia in the environment, but becomes yeast in infected tissues. In its mycelial form, it produces conidia (spores) that are inhaled by the host and then phagocytized by alveolar macrophages.7 In the presence of higher body temperature, spores become yeast with thick double-contoured outer walls and characteristic broad-based budding.6,7 The yeast causes local suppurative to pyogranulomatous inflammatory response, which may be self-limiting.8 However, phagocytized yeasts can be transported into the pulmonary interstitium where they can disseminate hematogenously or through the lymphatic system to other organs.8 Inoculation directly through a skin wound is also possible, but rare.8 By contrast with other mycotic diseases such as Coccidioides immitis and Aspergillus sp. where infectious spores can be easily aerosolized from infected tissues and transmit the disease, the yeasts of B. dermatitidis are not infective, and thus, do not require special biosafety precautions during necropsy.
Clinical signs with blastomycosis depend on the organ system most severely affected; however, respiratory signs is the most common clinical presentation.7,8 In dogs, B. dermatitidis often disseminates to the lungs, lymph nodes, skin, eyes, bones, reproductive system, and nervous system.8 Lameness caused by osteomyelitis or paronychia is reported in 25% of dogs with blastomycosis, and fungal osteomyelitis in 10-15% of cases.
Itraconazole is the treatment of choice at a dosage of 5 mg/kg (2.27 mg/lb) every 24 hours for 60 days. Treatment should be continued for 30-60 days after resolution of clinical signs. Clinical signs often become more severe in the early phase of treatment, as the yeast organisms die off and elicit severe inflammation.8 Prognosis for blastomycosis is good if severe pulmonary or CNS involvement is not present. Relapse is possible in dogs with initially severe clinical disease or in dogs treated for an insufficient length of time. Prolonged administration of prednisone might have contributed to the severe disseminated disease seen in this case.
JPC Diagnosis: Bone: Osteomyelitis, pyogranulomatous, diffuse, severe with marked cortical and trabecular osteolysis and numerous yeasts, Doberman pinscher (Canis familiaris), canine.
Conference Comment: Generally, bacterial osteomyelitis is more common than fungal, but there are three dimorphic fungi that can affect bone: (1) Blastomyces dermatitidis, (2) Coccidioides immitis, and (3) Cryptococcus neoformans. These three are dimorphic fungi, meaning they are in the filamentous form in the environment and yeast form in tissues. Blastomyces dermatitidis is fully described by the contributor above.1,2
Coccidioides immitis, an endosporulator, is a common inhabitant of the desert climates in the southwestern United States, areas of Mexico, Central, and South America and is transmitted primarily by inhalation of arthroconidia (spores). Once at body temperature, the spores transition into spherules (yeast form) which can migrate along the pleura creating damage by inducing production of arginase I and coccidioidal urease by host tissues. Infection can be subclinical and manifestation in bone often takes years to develop, generally long after respiratory lesions have healed. Dissemination to bone is most common in dogs with the distal diaphysis of long bones affected by isolated nodules. Spherules have an outer wall glycoprotein which acts to modulate host immune responses by compromising cell-mediated immunity and preventing phagocytosis. Grossly, coccidiomycosis forms firm, fibrous nodules often with caseating centers which microscopically are composed of granulomatous to pyogranulomatous inflammation surrounded by a thick fibrous connective tissue capsule. The fungus is often present at various stages of maturation, from the immature spherule (10-20um in diameter) to the mature spherule (up to 200um in diameter) which typically has a double contoured wall and contains numerous endospores (2-5um in diameter). Other organisms that reproduce by endosporulation include: Rhinosporidium seeberi, Prototheca sp., Chlorella sp., and Batrachochytrium dendrobatidis.1,2
Cryptococcus neoformans is a common inhabitant of pigeon feces and soil which typically affects immunocompetent cats and dogs (cats more so) with transmission via inhalation and primary lesions in the lungs and nasal cavity. In cats, osteolytic lesions occur in the bones of the maxilla with soft tissue swellings over the bridge of the nose. Grossly, lesions appear gelatinous or cyst-like. Microscopically, there are numerous yeast which exhibit narrow based budding, with thick heteropolysaccharide capsules which form a clear halo and characteristic “soap bubble” appearance. Their mucopolysaccharide capsules stain positive with Mayer’s mucicarmine and Alcian blue. Inflammation is scarce in these lesions due to the action of multiple virulence factors. The polysaccharide (glucuronoxylomannin) capsule prevents phagocytosis as well as inhibiting migration and recruitment of inflammatory cells. Additionally, an enzyme called laccase is produced which forms a melanin-like pigment that acts as an antioxidant. Two other enzymes, serine protease and urease aid in tissue invasion and promote sequestration in microcapillaries, respectively.1,2
During the conference, the moderator pointed out the marked loss of cortical bone which had been remodeled by osteoclasts, resulting in a trabecular appearance. Osteoclasts are of monocyte-macrophage lineage and many granulomatous reactions affecting bone result in marked bone loss and remodeling through osteoclastic activation.
Rdiographically, osteosarcoma and granulomatous inflammation may appear very similar. A key difference identified by the moderator is that infectious organisms usually cross the joint with abrupt transition from lesion to normal bone, whereas most neoplastic processes generally do not cross joints and have a longer zone of transition from lesion to normal. Additionally, clinically neoplasia is generally one large mass, whereas, with infectious organisms several joints may be infected. Radiographic interpretation and clinical history is essential for accurate histologic diagnosis.
Contributing Institution:
http://vet.cornell.edu/biosci/pathology/
References:
- Caswell JL, Williams KJ. The respiratory system. In: Maxie MG, ed. Jubb, Kennedy, and Palmer’s Pathology of Domestic Animals, Vol 2, 6th ed. St. Louis, MO: Elsevier; 2016:582-584.
- Craig LE, Dittmer KE, Thompson KG. Bones and joints. In: Maxie MG, ed. Jubb, Kennedy, and Palmer’s Pathology of Domestic Animals, Vol 2, 6th ed. St. Louis, MO: Elsevier; 2016:103-104.
- Crews JL, Feeney DA, Jessen CR, et al. Radiographic findings in dogs with pulmonary blastomycosis: 125 cases (1989–2006). J Am Vet Med Assoc. 2008; 232(2):215-221.
- Bromel C, Sykes JE. Epidemiology, diagnosis, and treatment of Blastomycosis in dogs and cats. Clin Tech Small Anim Pract. 2005; 20(4):233-239.
- Cote E, Barr SC, Allen C, et al. Blastomycosis in six dogs in New York State. J Am Vet Med Assoc. 1997; 210(4):502-504.
- Woods KS., Barry M, Richardson D. Carpal intra-articular blastomycosis in a Labrador retriever. Can Vet J. 2013; 54:167–170.
- Taboada J, Grooters AM. Systemic Mycoses. In: Ettinger SJ, Feldman EC, eds. Textbook of Veterinary Internal Medicine. 6th ed. St. Louis, MO: Saunders Elsevier; 2005:671-690.
- Legendre AM. Blastomycosis. In: Green CE, ed. Infectious Diseases of the Dog and Cat. 3rd ed. St Louis, MO: Elsevier Inc.; 2006:569-576.