Signalment:
Four 5-week-old, cross-bred pigs (Sus scrofa).The pigs were weaned two weeks previously, and treated with ivermectin at weaning. About 20 pigs died in a herd of 200 animals.
Gross Description:
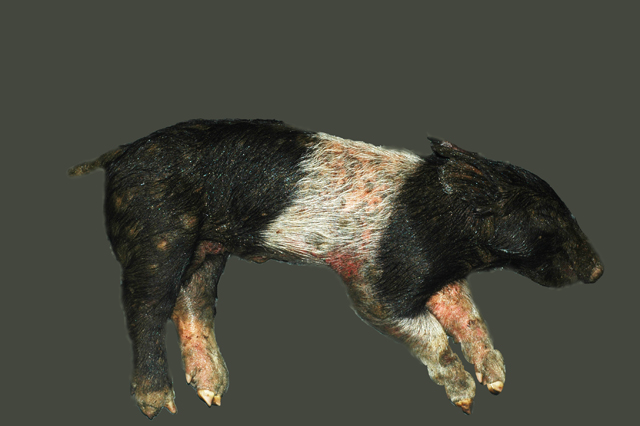
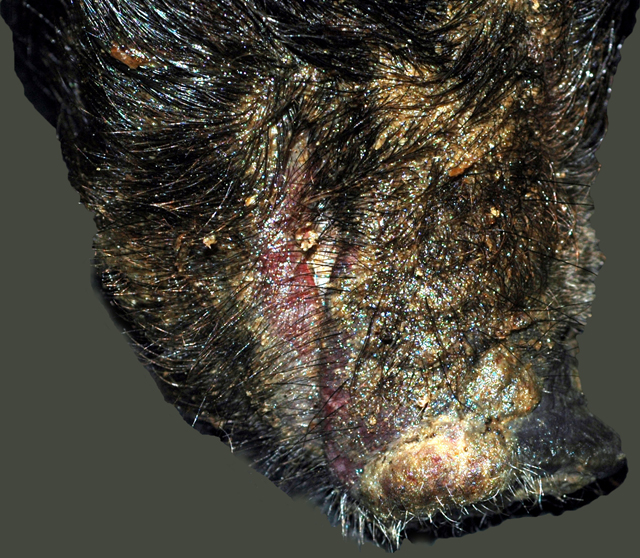
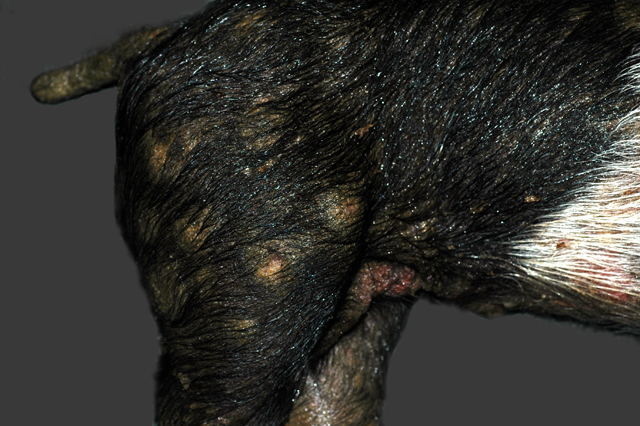
All four pigs had a generalized, greasy, moist appearance on the skin with multifocal crusting. Multifocally, there were multiple, raised, circular, crusty lesions (0.5-2.0 cm in diameter) around the eyes, and on the snout, chin, thorax, abdomen and legs. Exudate matted the eyes of one pig closed. There was mild separation of the hooves and erosions were present on the coronary bands.Â
Histopathologic Description:
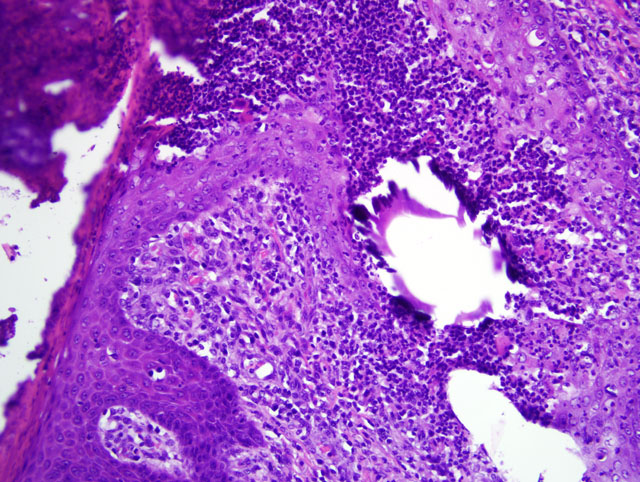
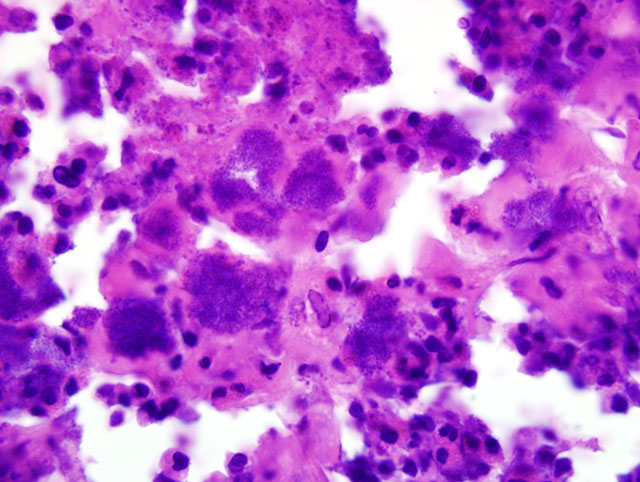
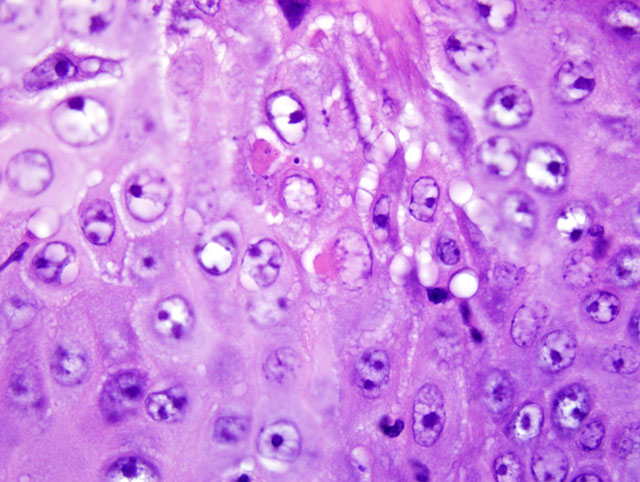
Multifocally, the epidermis is ulcerated with an overlying serocellular crust composed of neutrophils, fibrin, and serous proteinaceous material admixed with numerous colonies of bacterial cocci. Additionally, there are frequent intracorneal pustules composed of degenerate neutrophils that expand the stratum corneum. The epidermis in intact areas is multifocally, moderately hyperplastic with prominent rete pegs, and there are frequent neutrophils noted in exocytosis across the epithelial surface. Multiple follicular lumina are also filled with numerous degenerate neutrophils with occasional eosinophils. Multifocally, epithelial cells exhibit ballooning degeneration or hydropic change and contain occasional 5-7 μm diameter, eosinophilic, intracytoplasmic inclusion bodies (poxviral inclusions). There are occasional apoptotic cells scattered throughout the epidermis and follicular epithelium characterized by shrunken, hypereosinophilic cells with pyknotic or karyorrhectic nuclei. The dermis subjacent to the ulcerated areas is heavily infiltrated by lymphocytes, plasma cells, histiocytes, and scattered neutrophils with frequent fibroblasts and small capillaries (granulation tissue).
Morphologic Diagnosis:
1. Skin: dermatitis, proliferative and ulcerative, multifocal severe, with ballooning degeneration, granulation tissue, and intracytoplasmic eosinophilic inclusion bodies, porcine (
Sus scrofa).
2. Skin: epidermitis and folliculitis, suppurative (exudative), multifocal to coalescing, severe, with numerous bacterial cocci colonies, porcine (
Sus scrofa).
Lab Results:
Bacteriology:
Staphylococcus hyicus was cultured from multiple skin samples.Â
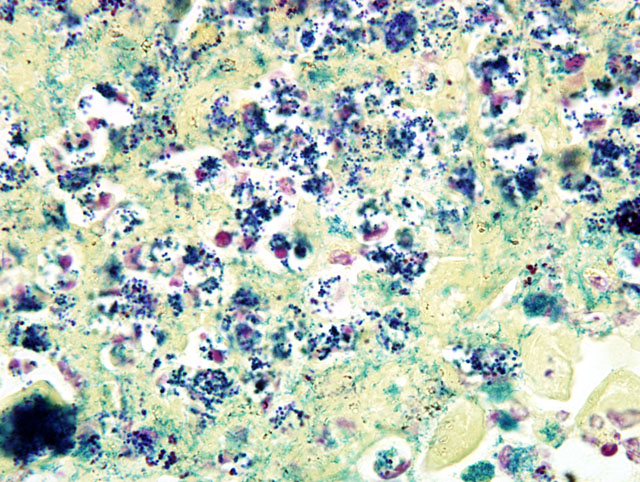
Virology: Swinepox virus was identified with electron microscopy from the circular, raised lesions.
Condition:
Primary swinepox with secondary Staphylococcus hyicus
Contributor Comment:
Greasy pig disease, or exudative epidermitis, is caused by
Staphylococcus hyicus, a gram positive bacterium. The disease occurs most commonly following introduction of carrier animals into a na+�-�ve herd.(12) The pathogenesis of exudative epidermitis is not completely known; however, trauma from fighting, unclipped teeth, rough bedding, or other factors leading to exposure of the dermis may allow the bacteria to establish the infection.(6,12) Initially, there is reddening of the skin with multiplication of bacteria on skin surface and growth between keratinocytes. In infected skin, there is generally marked inflammation with hyperplasia of the stratum corneum and neutrophilic infiltration followed by epidermal erosion.(12) The most important factor in pathogenesis of infection is the production of exfoliative toxins. The exfoliative toxins are known as: ExhA, ExhB, ExhC, ExhD, SHETA and SHETB.(3,10) Exudative epidermitis is regarded as a porcine homologue of Staphylococcal Scalded Skin Syndrome (SSSS) or bullous impetigo in humans. SSSS results in loss of keratinocyte cell-to-cell adhesion and leads to blister formation.(9) The virulent strains of
S. aureus produce exfoliative toxins, namely ETA, ETB and ETD.(1) It has been reported that these toxins are glutamate specific serine proteases that cleave a single peptide bond in the extracellular region of human and mouse desmoglein (Dsg) 1.(2) In human epidermis, Dsg1 and Dsg3 are present predominantly in stratified squamous epithelium.(11) Recent studies have suggested that pathophysiological mechanisms of intraepidermal splitting in exudative epidermitis are similar to human SSSS by cleavage of Dsg1.(5,8,9) The detailed differential list for exudative epidermitis has already been described in
WSC 2008-2009, Conference 9, case I.Â
Swine pox is a typical poxvirus primarily affecting young pigs and lesions are usually confined to the ventrolateral abdomen.(3) Transmission is typically through direct contact, although lice (Haematopinus suis) and other blood-sucking insects can be an important means of transmission in swine herds as well.(3) Lesions follow the typical pox progression: erythematous macules becoming papules, then vesicles progressing to pustules, leading to rupture and crust formation.(3) Differentials for swine pox must include other vesicular diseases, sarcoptic mange, and erysipelas.(3) Swinepox virus is the sole member of the suipoxvirus genus and is morphologically similar to vaccinia virus; it has a double-stranded DNA genome of 146 kilobase pairs and 150 predicted genes.(4) Following an abrasion to the skin, swinepox may enter the host and preferentially replicates in epidermal keratinocytes of the stratum spinosum; replication occurs exclusively in the cytoplasm, as indicated by the intracytoplasmic inclusion bodies.(4)
In this case, the swine pox infection was likely the predisposing factor leading to the staphylococcal infection. In addition to the skin lesions in these cases, there was evidence of embolic showering of bacteria within the lungs and lymph nodes with suppurative inflammation in all pigs examined (evidence for systemic infection). Therefore, the cause of death in these pigs appears to be sepsis secondary to epidermal infection with
Staphylococcus hyicus, made possible by the ulcerative lesions caused by the poxviral infection.
JPC Diagnosis:
Haired skin: Dermatitis, proliferative and necrosuppurative, chronic, diffuse, marked, with epidermitis, folliculitis, and many cocci.
Conference Comment:
During the conference, the moderator highlighted that the presence of many acantholytic keratinocytes in this case is reminiscent of the autoimmune condition of pemphigus foliaceus. This observation is meaningful in light of the contributors comments concerning the pathogenesis of staphylococcal cutaneous infections, and this case provides an exquisite example of two diseases sharing a common molecular link, i.e. desmogleins as the target of bacterial toxins and autoantibodies, resulting in histomorphological similarities. As noted by the contributor, Dsg1 and Dsg3 are both components of desmosomes, but differ with respect to their histologic distributions within the epidermis and anatomic locations. The Dsg1 protein, the target of autoantibodies in pemphigus foliaceus, is most abundant in the superficial layers of the epidermis and is scarce in oral mucosa, which accounts for the characteristic histologic pattern of acantholytic subcorneal and intragranular pustular dermatitis and an absence of oral lesions in the disease. The Dsg3 protein is more abundant in the basal layers of the epidermis and oral mucosa and, along with Dsg1, is targeted by autoantibodies in pemphigus vulgaris thus explaining the characteristic suprabasilar clefting in the epidermis and lesions found in the oral mucosa.(7)
There is substantial slide variation with respect to the ballooning degeneration and poxviral inclusions described by the contributor. Most conference participants slides lacked this feature or had only rare examples. This variation is not surprising given that the submission consisted of samples from multiple animals. The contributor provided a succinct review of swinepox, and readers are referred to
WSC 2009-2010, Conference 5, case II for additional discussion regarding poxviruses.
References:
1. Amagai M, Matsuyoshi N, Wang ZH, Andl C, Stanley JR: Toxin in bullous impetigo and staphylococcal scalded-skin syndrome targets desmoglein 1. Nat Med
6:1275-1277, 2000
2. Amagai M, Yamaguchi T, Hanakawa Y, Nishifuji K, Sugai M, Stanley JR: Staphylococcal exfoliative toxin B specifically cleaves desmoglein 1. J Invest Dermatol
118:845-850, 2002
3. Andresen L, Bille-Hansen V, Wegener H:
Staphylococcus hyicus exfoliative toxin: purification and demonstration of antigenic diversity among toxins from virulent strains. Microb Pathog
22:113-122, 1998
4. Cameron R: Diseases of the skin.Â
In: Diseases of Swine, eds. Straw BE, Zimmerman J, D'Allaire S, Taylor D, 9th ed., pp. 179-198. Blackwell Publishing, Ames, IA, 2006
5. Delhon G, Tulman E, Afonso C, Rook DL: Swine pox.Â
In: Diseases of Swine, eds. Straw BE, Zimmerman J, DAllaire S, Taylor D, 9th ed., pp. 483-487. Blackwell Publishing, Ames, IA, 2006
6. Fudaba Y, Nishifuji K, Andresen LO, Yamaguchi T, Komatsuzawa H, Amagai M, Sugai M:
Staphylococcus hyicus exfoliative toxins selectively digest porcine desmoglein 1. Microb Pathog
39:171-176, 2005
7. Ginn PE, Mansell JEKL, Rakich PM: Skin and appendages.Â
In: Jubb, Kennedy, and Palmers Pathology of Domestic Animals, ed. Maxie MG, 5th ed., vol. 1, pp. 647-650. Elsevier Saunders, Philadelphia, PA, 2007
8. Nishifuji K, Fudaba Y, Yamaguchi T, Iwasaki T, Sugai M, Amagai M: Cloning of swine desmoglein 1 and its direct proteolysis by
Staphylococcus hyicus exfoliative toxins isolated from pigs with exudative epidermitis. Vet Dermatol
16:315-323, 2005
9. Nishifuji K, Sugai M, Amagai M: Staphylococcal exfoliative toxins: "molecular scissors" of bacteria that attack the cutaneous defense barrier in mammals. J Dermatol Sci
49:21-31, 2008
10. Sato H, Watanabe T, Higuchi K, Teruya K, Ohtake A, Murata Y, Saito H, Aizawa C, Danbara H, Maehara N: Chromosomal and extrachromosomal synthesis of exfoliative toxin from
Staphylococcus hyicus. J Bacteriol 182:4096-4100, 2000
11. Shirakata Y, Amagai M, Hanakawa Y, Nishikawa T, Hashimoto K: Lack of mucosal involvement in pemphigus foliaceus may be due to low expression of desmoglein 1. J Invest Dermatol 110:76-78, 1998
12. Wegener H, Skov-Jensen E: Exudative epidermitis. In: Diseases of swine, eds. Straw B, Zimmerman J, D'Allaire S, Taylor D, 9th ed., pp. 675-679. Blackwell Publishing, Ames, IA, 2006