Conference 20, Case 2
Signalment:
4-year-old, male, Balinese cat, (Felis silvestris catus)
History:
Sudden death, after one month in which the subject manifested increased appetite.
Gross Pathology:
At necropsy, multiple organs were affected:
Abdominal cavity: in the cavity there was an effusion (approx. 30 ml) of pale, yellow, and transparent fluid which contained scattered strands of white, elastic, and insoluble material (fibrin).
On the serosal surfaces of the major abdominal and thoracic organs (spleen, kidneys, intestine, pancreas, lungs and heart) there were multifocal, well defined, irregularly round, raised, white, and compact plaques that varied in size from few millimeters to a centimeter in major diameter. Some of these lesions extended into the parenchyma of organs and presented poorly defined and irregular edges.
Thoracic cavity: the same fluid (approx. 10 ml) was found in the thoracic cavity.
Trachea: There was moderate intraluminal white-pink foam (edema). Lungs: multifocal, white, solid, slightly protruding lesions up to few millimeters in diameter were present under the pleura. At cut surface, they showed irregular well-defined margins and white coloration.
Liver: The serosal surface of the organ was covered by extensive and abundant yellow, firm material, with a granular superficial appearance. On cut surface, the material did not extend into the parenchyma. Many multifocal friable yellow adhesions were present between the liver and the wall of the abdominal cavity.
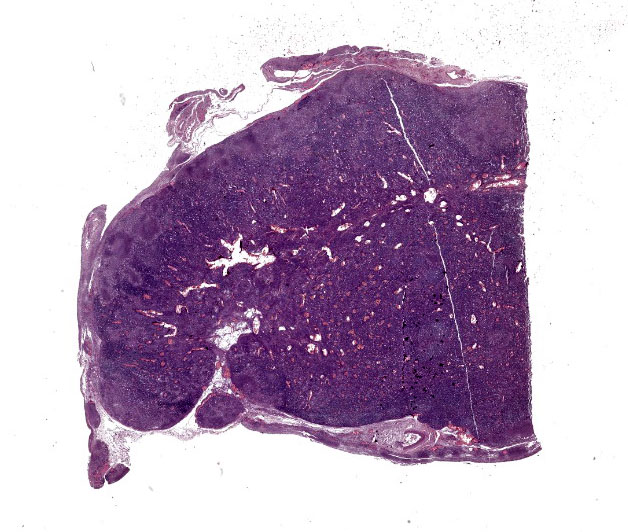
Lymph nodes: Thoracic and abdominal lymph nodes were enlarged, dark red, and firm. At cut surface, they presented a diffuse homogenous dark red coloration. The macroscopic morphological diagnosis was therefore: diffuse and severe fibrinous peritonitis and pleuritis with generalized severe hemorrhagic lymphadenitis.
Laboratory Results: N/A
Microscopic Description:
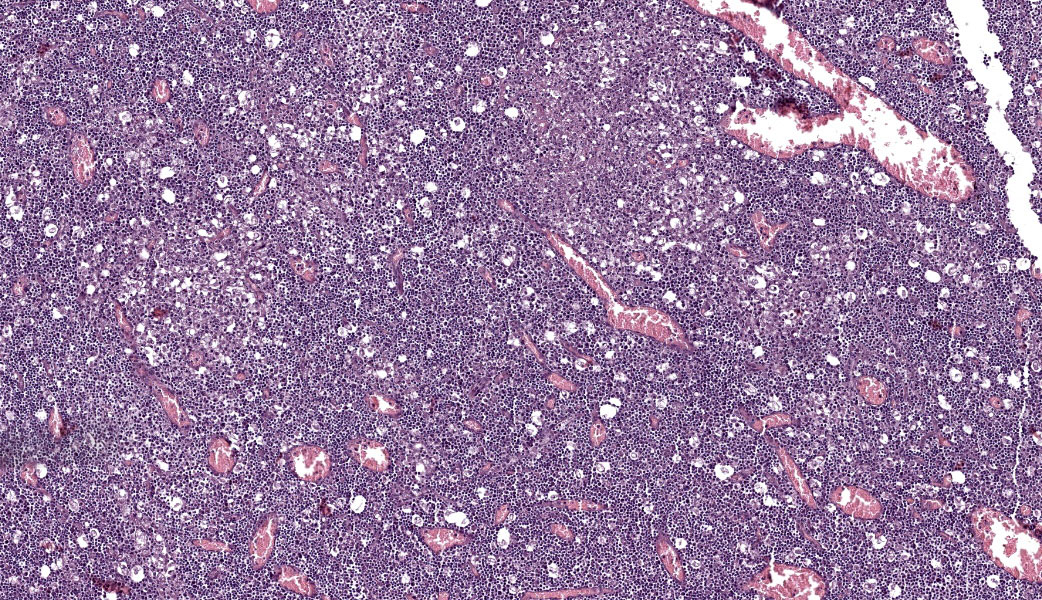
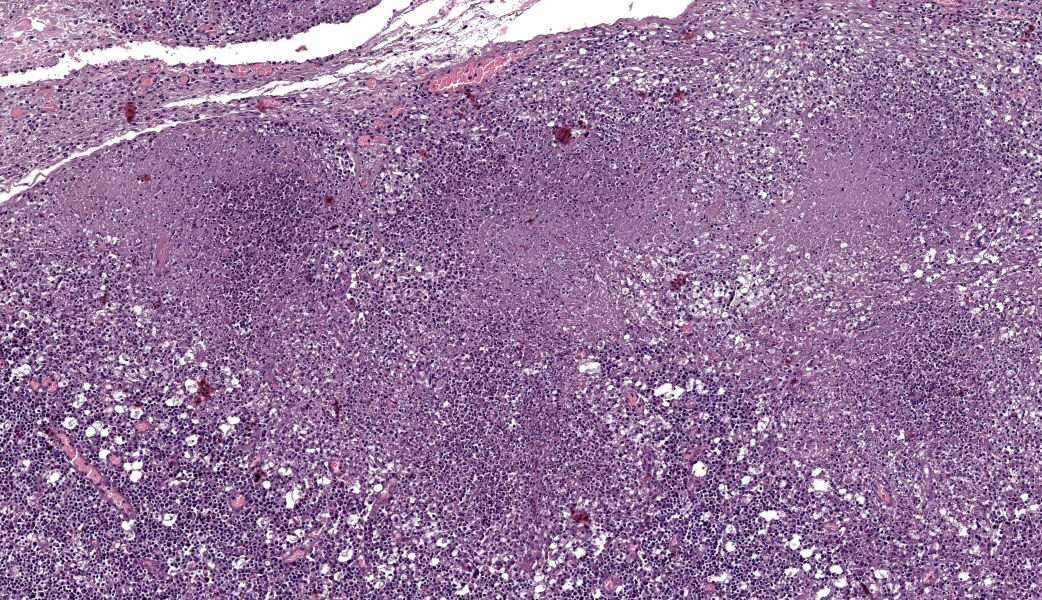
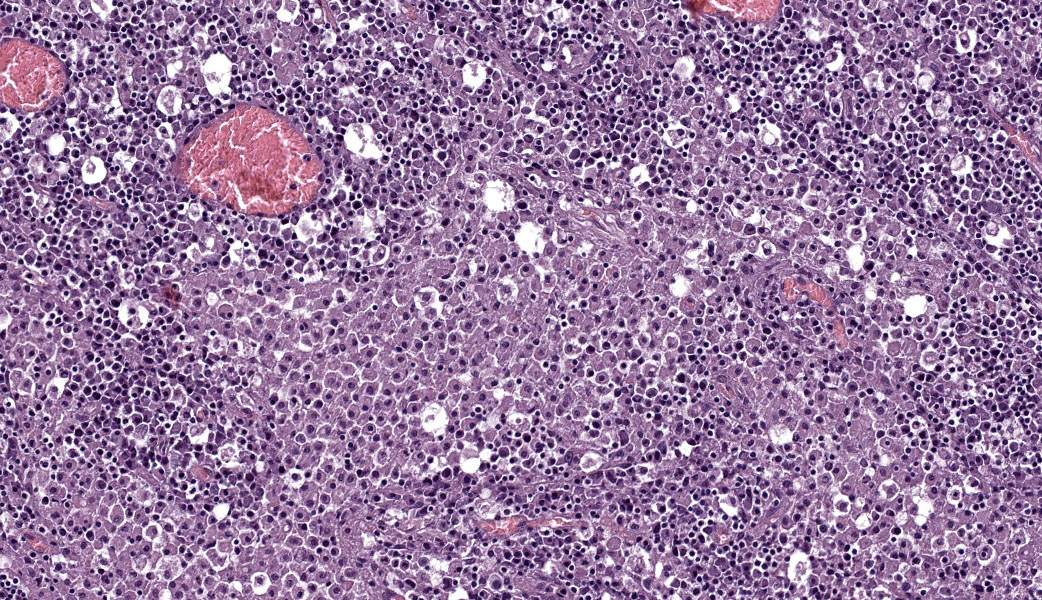
Abdominal lymph node: Multifocal, severe cellular infiltrates with undefined margins are randomly distributed within the lymph node parenchima especially in the subcapsular areas. They extend into the serosal expanding and effacing the mesothelial lining. The infiltrates are characterized by a severe mixed inflammatory population with largenumbers of both viable and lytic neutrophils surrounded by occasional large macrophages with abundant cytoplasm and rare lymphocytes and plasma cells. In the center of almost each infiltrate the neutrophils population shows karyolysis, pyknosis, karyorrhexis and the accumulation of extensive amount of amorphous eosinophilic, proteinaceous material admixed with cellular debris (lytic necrosis).
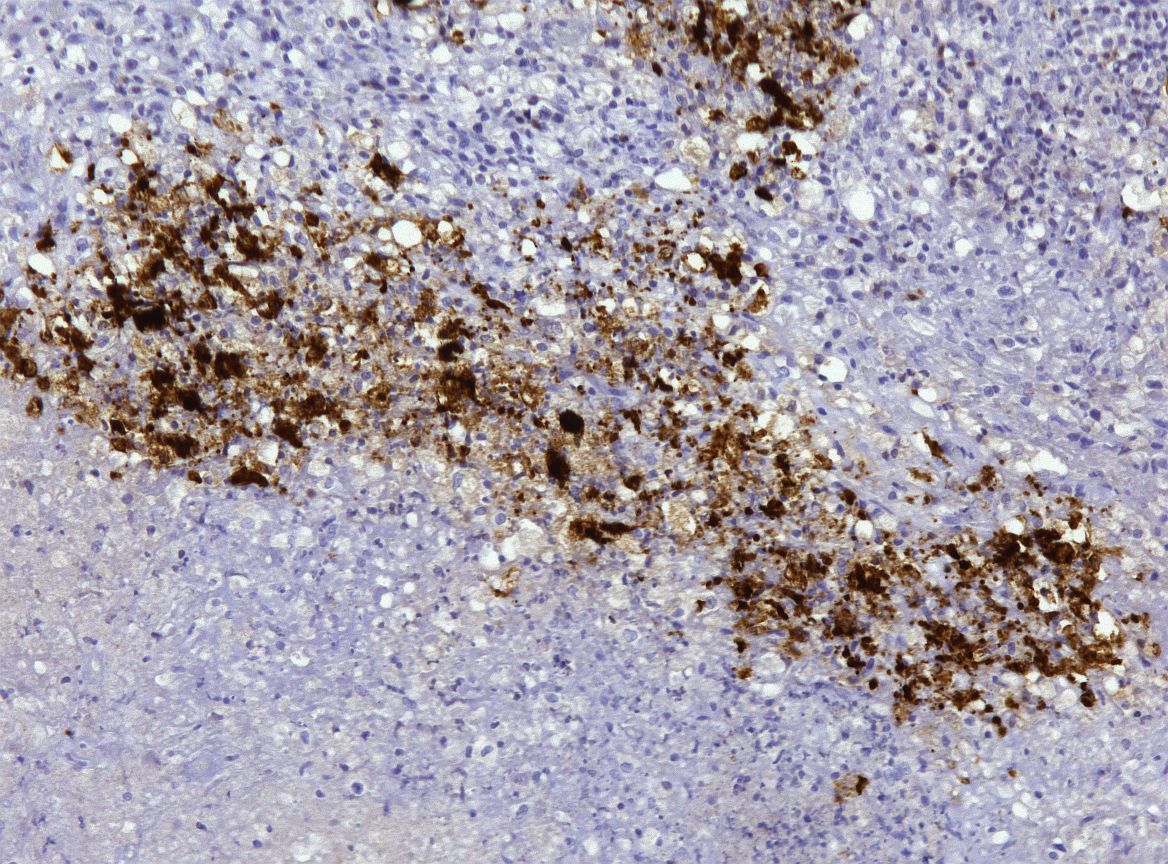
Immunohistochemical (IHC) staining of lymph node sections using a mouse anti-Feline Coronavirus antibody (clone FIPV3-70 Serotec, Oxford UK) demonstrates strong positivity for many elements within the inflammatory infiltrates, suggestive of presence of viral antigen.
Contributor's Morphologic Diagnosis:
Multifocal severe necrotizing and piogranulomatous lymphadenitis and peritonitis associated with Feline Coronavirus antigen. Feline infectious peritonitis.
Additional findings:
- Diffuse severe peritonitis
- Multifocal severe piogranulomatous hepatitis, pneumonia, pancreatitis, nephritis,myocarditis and enteritis.
- Lung: Mild diffuse edema and mild diffuse emphysema
Liver: Diffuse, severe hepatic hydropic degeneration.
Contributor's Comment:
Feline infectious peritonitis (FIP) is an uncommon, fatal, progressive, and immune-mediated disease of cats caused by feline coronavirus (FCoV) infection. At present, despite decades of research on its etiology, pathogenesis, transmission, and prevention, FIP is still one of the most frequent fatal and infectious feline disease for which there is, so far, no effective cure.1
Infection with FCoV is common in cats throughout the world, although, in most cats, the virus causes no clinical signs. However, in some cats, FCoV infection is associated with the development of the progressive and fatal disease manifestation of FIP.3
The current knowledge of genetic differences between feline enteric coronaviruses (FECV) and feline infectious peritonitis virus (FIPV) has been mainly based on comparative sequence analyses that revealed "discriminatory" mutations that are present in FIPVs but not in FECVs.6 Most of these mutations result in amino acid substitutions in the S protein and these may have a critical role in the switch from FECV to FIPV.1 In most cases, the precise roles of these mutations in the molecular pathogenesis of FIP have not been tested experimentally in the natural host, mainly due to the lack of suitable experimental tools including genetically engineered virus mutants.6
Cats that do not clear FIPV develop either dry or wet forms of the disease depending on whether ineffective cell-mediated or humoral immunity dominates the clinical disease. Although often described as distinct entities, the effusive and non-effusive forms of FIP are the farthest of a continuum of syndromes, characterized by vasculitis and pyogranulomatous inflammation. Effusive disease is more common than the non-effusive form, and mixed forms are common.6 In our case clear indication of vasculitis was not present and necrosis was diffusely associated with the pyogranulomatous infiltrate, therefore IHC was needed to confirm the etiology.
Contributing Institution:
Dept. Comparative Biomedicine and Food Science (BCA) -
Veterinary Medicine - University of Padua
AGRIPOLIS - Viale dell'Universita 16
35020 Legnaro (PD) - Italy
http://www.bca.unipd.it/en/
JPC Morphologic Diagnosis:
Lymph node: Lymphadenitis and capsulitis, necrotizing and granulomatous, chronic, multifocal to coalescing, marked, with paracortical hyperplasia.
JPC Comment:
This case was an excellent representation of a real-life manifestation of FIP, as classic vasculitis was not a significant part of this lesion (although areas of necrosis within the node suggested the presence of vascular damage. The top differentials for participants included FIP, Francisella tularensis, and Yersinia pestis (causative agent of bubonic plague) and other ?hot? gram-negatives, and less commonly, and rare cases of feline parvovirus infection. The Pesavento Laboratory confirmed the positive immunohistochemical identification of coronavirus in this case using a canine coronavirus antibody which cross reacts with many other related coronaviruses.
FIP is globally distributed and fatal if untreated. Today, however, antiviral treatment is over 80% effective.4 Certain breeds are more susceptible to infection, but the gene association in the host has not yet been identified.8 FIPV can cause disease in exotic big cat species as well, and has been reported in lions, servals, cheetahs, mountain lions, and others.2,5,7,9,10
Dr. Pesavento discussed how much we still have to learn about FIP, including the triggers of viral activation, how FIP persists in a host, the host range, susceptibility factors, and how variable manifestations of infection affect the outcome for the individual. While 98% of cats infected with FeCoV never develop FIP, what exactly causes or triggers the genetic mutations that result in FIP is still unknown. The general cause of FIP is enteric persistence of FeCoV within a host, specifically within the colon, coupled with systemic access and mutation of the virus into a pathogenic FIPV. There are multiple mutations and multiple genes involved in this transformation of FeCoV into FIPV, many of which are not fully understood.
Additionally, a phenomenon known as ?antibody-dependent enhancement? has been demonstrated in cats that are re-infected with an identical serotype of FIPV.11 Antibody-dependent enhancement (ADE) is a phenomenon in which non-neutralizing or weak antibodies bind to a virus but fail to neutralize it. Instead, the antibody actually facilitates the virus? entry into cells, resulting in increased viral replication and more severe disease.11 This makes this disease particularly challenging for vaccine development. Currently vaccine development targets the nucleocapsid (N) protein. This N protein is well-conserved across coronaviral strains, unlike the spike (S) protein that is prone to significant variation. The N protein can trigger a longer-lasting antibody response, but, perhaps more importantly, an N-specific cytotoxic T lymphocyte response. This elegant targeting of a cell-mediated, rather than a humoral, response may help avoid antibody-dependent enhancement in FIPV.
References:
- Brown MA, Troyer JL, Pecon-Slattery J, Roelke ME, O'Brien SJ. Genetics and Pathogenesis of Feline Infectious Peritonitis Virus. Emerg Infec Dis. 2009;15(9):1445-1452
- Evermann JF, Heeney JL, Roelke ME, McKeirnan AJ, O'Brien SJ. Biological and pathological consequences of feline infectious peritonitis virus infection in the cheetah. Arch Virol. 1988;102(3-4):155-171.
- Giordano A, Paltrinieri S, Bertazzolo W, Milesi E, Parodi M. Sensitivity of Tru-cut and fine-needle aspiration biopsies of liver and kidney for diagnosis of feline infectious peritonitis. Vet Clin Path. 2005;34(4):368-374.
- Kipar A, Meli ML. Feline Infectious Peritonitis: Still an Enigma? Vet Path. 2014;51 (2):505-526.
- Juan-Salles C, Domingo M, Herraez P, Fernandez A, Segales J, Fernandez J. Feline infectious peritonitis in servals (Felis serval). Vet Rec. 1998;143(19):535-536.
- Kipar A, Meli ML. Feline Infectious Peritonitis: Still an Enigma? Vet Path. 2014;51 (2):505-526.
- Mwase M, Shimada K, Mumba C, Yabe J, Squarre D, Madarame H. Positive immunolabelling for feline infectious peritonitis in an African lion (Panthera leo) with bilateral panuveitis. J Comp Pathol. 2015;152(2-3):265-268.
- Pesteanu-Somogyi LD, Radzai C, Pressler BM. Prevalence of feline infectious peritonitis in specific cat breeds. J Feline Med Surg. 2006;8(1):1-5.
- Stephenson N, Swift P, Moeller RB, Worth SJ, Foley J. Feline infectious peritonitis in a mountain lion (Puma concolor), California, USA. J Wildl Dis. 2013;49(2):408-412.
- Stout AE, Andr? NM, Whittaker GR. Feline coronavirus and feline infectious peritonitis in nondomestic felid species. J Zoo Wildl Med. 2021;52(1):14-27.
- Takano T, Kawakami C, Yamada S, Satoh R, Hohdatsu T. Antibody-dependent enhancement occurs upon re-infection with the identical serotype virus in feline infectious peritonitis virus infection. J Vet Med Sci. 2008;70(12):1315-1321.