Signalment:
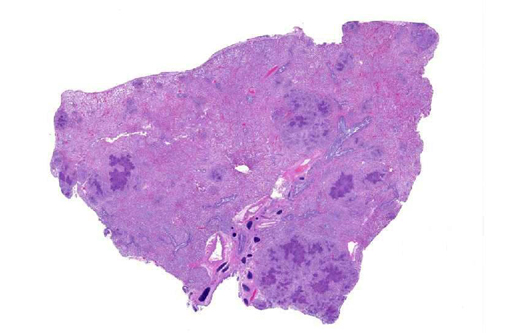
Gross Description:
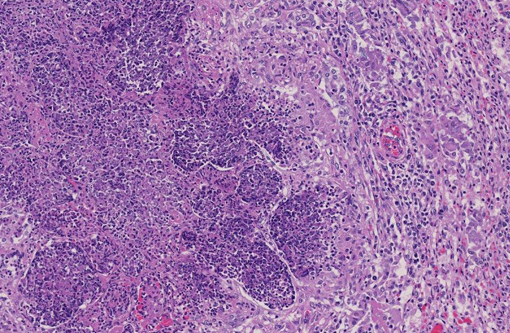
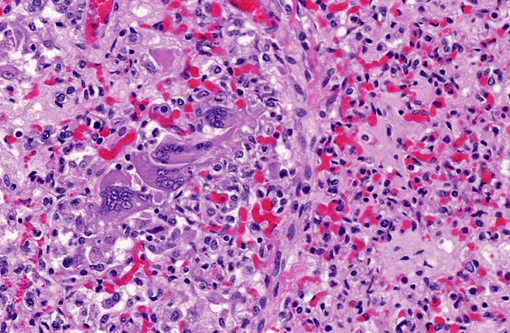
Histopathologic Description:
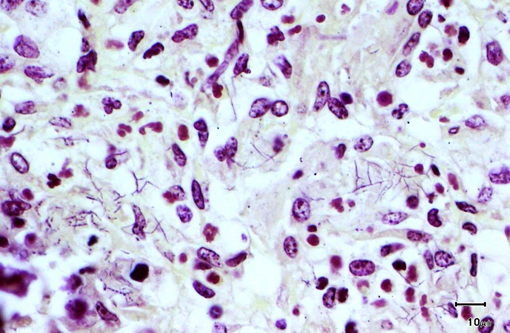
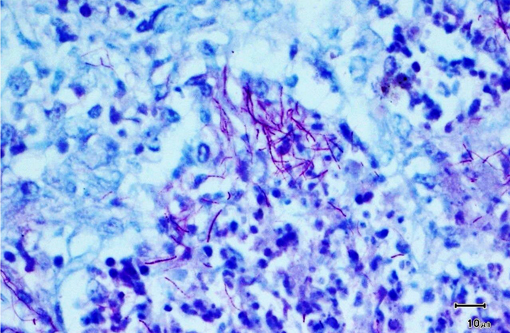
Special stains revealed numerous, approximately 1 micron thick, beaded to filamentous and occasionally branching, gram-positive (Brown & Brenn) and variably acid-fast (Fite-Faraco) bacteria within areas of inflammation.
Morphologic Diagnosis:
Lab Results:
Condition:
Contributor Comment:
The primary differential diagnosis for pulmonary nocardiosis includes infections with Actinomyces and some atypical mycobacteria.(9) Actinomycosis is more commonly associated with sulfur granules and Splendore-Hoeppli material, and the organisms are not acid fast.(10) Atypical mycobacteria are much shorter beaded bacilli and do not exhibit true branching.Â
Nocardia are commonly found in the environment, particularly soil enriched with decayed organic matter. Routes of infection include inhalation, inoculation, and ingestion. In people, Nocardia are generally considered pathogenic bacteria with relatively low virulence. With the exception of cutaneous disease, human infection is typically opportunistic, occurring more commonly in the elderly and people with compromised T cell-mediated immunity, as with prolonged steroid treatment or HIV infection.(6) In the present case, it is not clear if the absence of a normal appearing thymus at necropsy was due to a primary condition predisposing the animal to infection (thymic hypoplasia) or a secondary response to the severe infection (thymic atrophy).Â
Nocardiosis is an important cause of pneumonia, pleuritis and empyema in dogs, particularly hunting/sporting dogs in which, following inhalation or ingestion of plant material, migrating grass awns can contaminate the pleural spaces with bacteria resulting in a putrid tomato soup-like hemorrhagic pyothorax. In most other species, pulmonary nocardiosis more closely resembles the nodular pyogranulomatous and necrotizing inflammation observed in this lamb. Nocardia infections have been infrequently reported in primates, cats, horses, cattle and marine mammals.(1,2,5,8) In cattle, infections have been associated with mastitis, pneumonia, dermatitis, placentitis with abortion, and disseminated disease.(1) Nocardia infections in other ruminants are apparently very uncommon, but have been reported in sheep, goats, llama, bison and reindeer.(3,7,10) Nocardiosis is reportedly a significant and often fatal disease in marine mammals.(8) Pinnipeds, particularly hooded seals, and cetaceans appear to be prone to a systemic form of infection which typically involves the lung and thoracic lymph nodes and, to lesser extent, brain and skin.Â
JPC Diagnosis:
Conference Comment:
References:
2. Bolon B, Buergelt CD, Cooley AJ. Abortion in two foals associated with Nocardia infection. Vet Pathol.1989;26(3):277278.
3. Chang CD, Boosinger TR, Dowling PM, et al. Nocardiosis in a llama. J Vet Diagn Invest. 1993;5(4):631-4.
4. Erol E, Sells SF, Williams NM, Kennedy L, Locke SJ, Labeda DP, et al. An investigation of a recent outbreak of nocardioform placentitis caused abortions in horses. Vet Microbiol. 2012:17;158(3-4):425-30.
5. Klumpp SA, McClure HM. Nocardiosis, lung. In: Jones TC, Mohr U, Hunt RD, eds. Monographs on Pathology of Laboratory Animals: Nonhuman Primates II. Berlin and New York: Springer-Verlag; 1993:99103.Â
6. McAdam AJ, Sharpe AH. Infectious diseases. In: Kumar V. Abbas A. Fausto N, eds. Robbins and Cotran: Pathologic Basis of Disease. 8th edition. Philadelphia, PA: Elsevier Saunders; 2010:362-363.Â
7. Pal M. Nocardia asteroides as a cause of pneumonia in a buffalo calf. Review of Scientific Technical Office International Des Epizootics. 1997;16:881-884.Â
8. St. Leger JA, Begeman L, Fleetwood M, et al. Comparative pathology of nocardiosis in marine mammals. Vet. Pathol. 2009;46:299308.
9. Travis WD, Colby TV, Koss MN, et al. Lung Infections. Atlas of Nontumor Pathology. Non-neoplastic Disorders of the Lower Respiratory Tract. Washington, DC: American Registry of Pathology; 2002:557-563.
10. Vemireddi V, Sharma A, Wu CC, et al. Systemic nocardiosis in a reindeer (Rangifer tarandus tarandus), J. Vet. Diagn. Invest. 2007;19(3):326-329.