Signalment: A 3.5 year old, male New Zealand White rabbit (Oryctolagus cuniculus)
History: A 3.5 year old, intact male, NZW, apo-AI and LCAT (apolipoprotein and lecithin:cholesterol acyltransferase) double transgenic rabbit initially presented with a discrete raised 0.5 x 1.0 x 1.0 cm mass on the face approximately one cm rostral to the medial canthus of the left eye. The facial mass was surgically removed as an excisonal biopsy. Within three months the mass had recurred and was raised and ulcerated, measuring 4.0 x 2.0 x 1.5 cm. A second surgery was performed to resect the tumor with wider margins. Over the next three weeks the rabbit?s condition declined and the rabbit developed respiratory distress and died acutely.
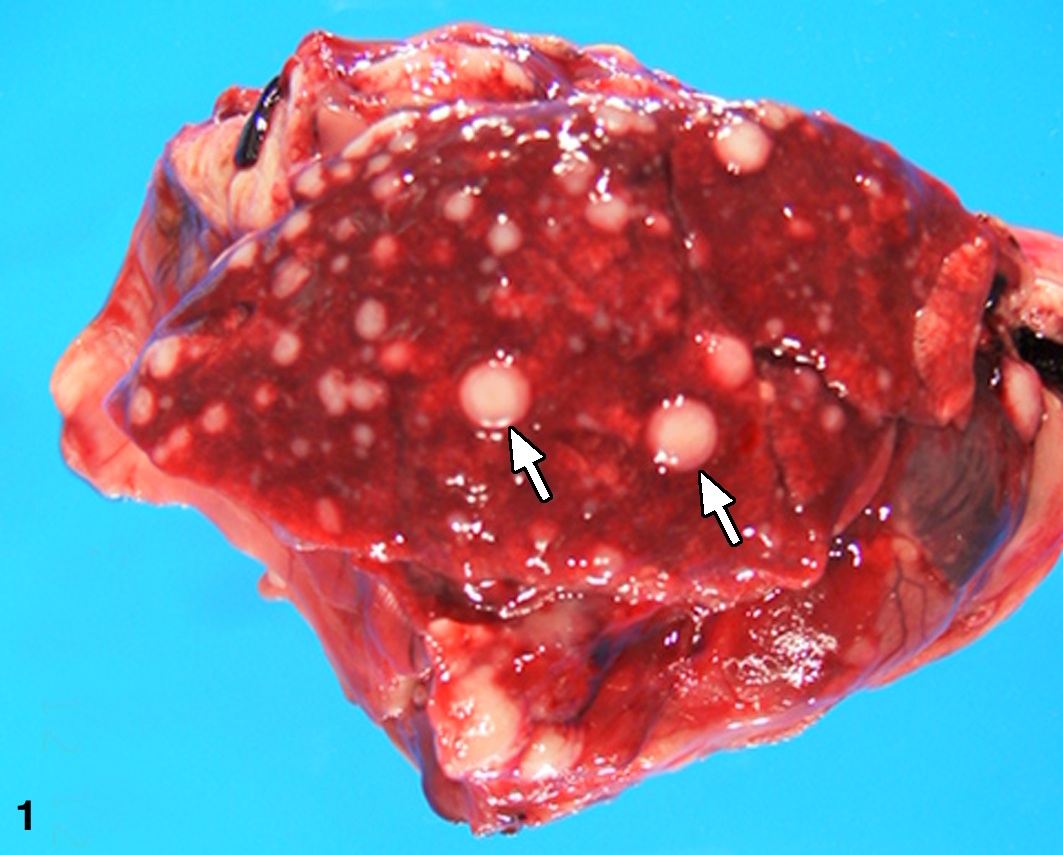
Gross pathologic findings: At necropsy, there was a healed incision at the site of the excisional biopsy. One facial mass was 1.5 X 2.5 X 3.5 cm white, resilient, somewhat lobulated and a similar, 1.0 X 1.0 X 1.5 cm mass present caudal to the first mass. Two subcutaneous masses were present between the rami of the mandible. Within all lung lobes there were multifocal to coalescing firm grayish-white variably sized masses which measured up to 7 mm in diameter (Figure 1). The masses extended into the parietal pleura and multifocally within the diaphragm. Lesions were not observed in any other major organs.
Histopathological description: Histological examination of the original excised skin biopsy revealed a raised mass that invaded into the dermis which was partially ulcerated, non-encapsulated and well-delineated composed of tightly packed polygonal to spindloid cells with a fine fibrovascular stroma. The cells were moderately anisocytotic and anisokaryotic with moderate lightly eosinophilic finely granular cytoplasm with indistinct cell borders. Nuclei were round to oval with finely stippled chromatin, 1-4 nucleoli and mitoses were frequent, 5-7/hpf. Vascular invasion was evident with tumor cells present within multiple venules and lymphatic vessels in multiple tissues. Within the skin, neoplastic cells were seen fingering into the adjacent dermis and extended to the cut border of the facial mass. Similar neoplastic foci with a packeting of cells were present in the submandibular lymph nodes, lungs, parietal pleura, and liver. Tumor cell morphology in these metastatic sites resembled the morphology of the tumor from the excisional biopsies.
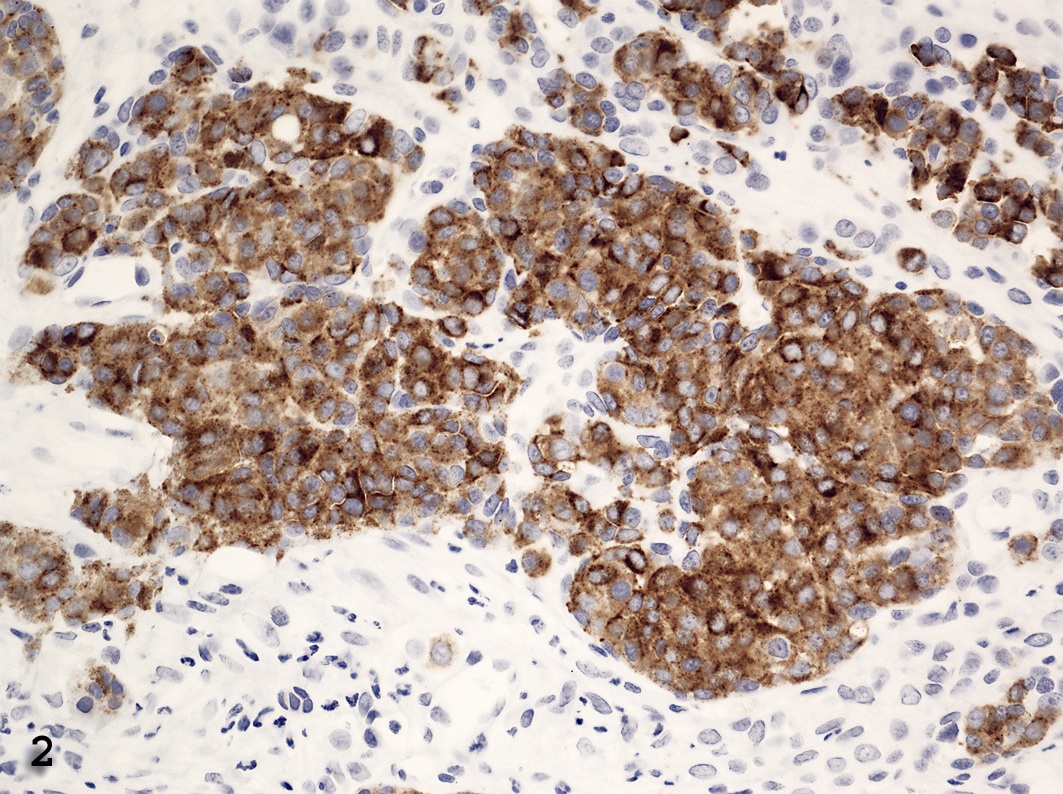
The masses from the biopsies and necropsy were stained for Fontana-Masson method (melanin stain). Results were negative. Immunohistochemistry demonstrated positive cytoplasmic staining for vimentin in both polygonal and spindloid cells. There was positive cytoplasmic staining for Mart-1 and S-100 in the polygonal cells, but negative staining in the spindloid cells (Figure 2). Staining with actin, HHF35, AE1/AE3, HMB-45, SMA and tyrosinase were negative.
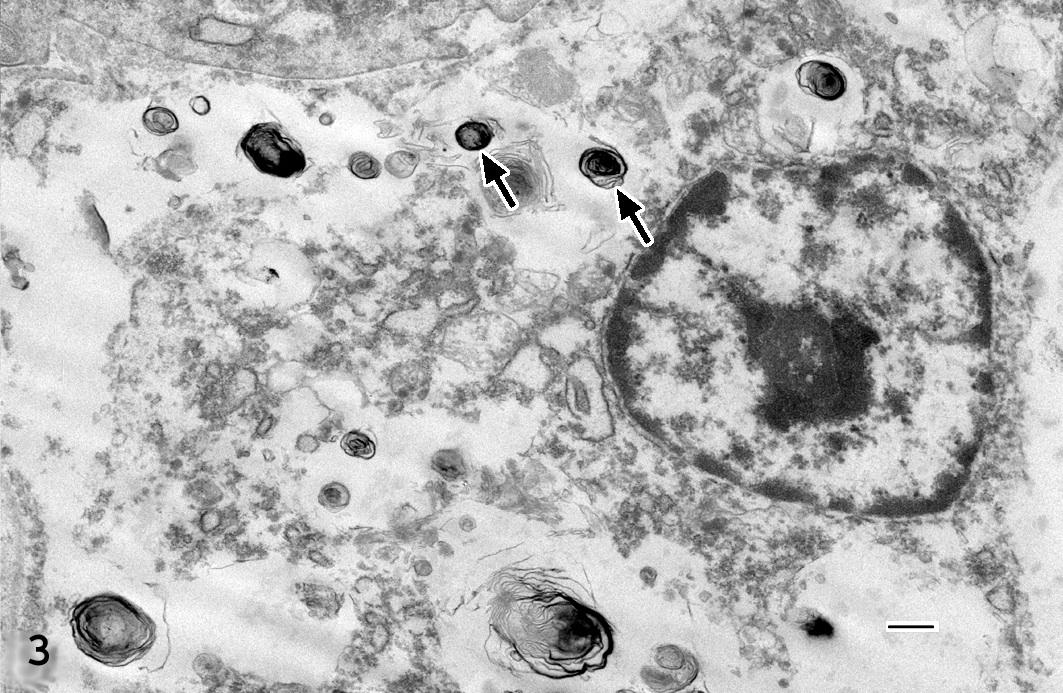
Transmission electron microscopy of the neoplastic cells revealed small numbers of clustered stage II melanosomes with a size range of 100-900 nm (Figure 3) with myelin-like membranes in sheets.
Contributor?s morphological diagnosis:
Skin, Melanoma, Amelanotic
Contributor?s Comment: This case of an amelanotic melanoma in an albino rabbit is rare. Albinism is an autosomal recessive disorder in which there are an adequate number of normally distributed melanocytes, but the melanin is not synthesized in a great enough quantity due to a point mutation in the gene for tyrosinase. Amelanotic melanomas have been reported in human albinos and experimentally induced in albino guinea pigs. 13, 16 Human amelanotic melanomas are rare only accounting for 5% of all melanoma cases.16 While melanomas are most commonly found in skin, any tissue that has melanin can be a site of tumor development.7
Melanomas are uncommon in rabbits. 1, 2, 3, 5, 6, 15 Amelanotic melanomas being extremely rare with only one report in the literature.6 The first case of melanoma in a rabbit was reported over 80 years ago.15 Melanomas have been reported in pet and laboratory rabbits; with NZW strain being over-represented.6 A retrospective study at the School of Veterinary Medicine at the University of Pennsylvania of 179 pet rabbits with a total of 190 tumors, eight were diagnosed as malignant melanoma by H&E and IHC.2 These melanomas were located on the skin of the pinna, eyelid, head, limb and scrotum. These melanomas had the typical appearance of aggressively growing tumors with high cellular pleomorphism and large numbers of mitotic figures. All tumors contained abundant melanin.2
Melanomas have been reported in other species including cat, 12 opossum, 10 monkey, 14 rat,9 dog,8 ferret18 and horse.11 Melanomas account for 7% of all malignant tumors in dogs and are most often found in the oral cavity, with metastases commonly occurring in the lung and lymph nodes. Metastases can be found in the brain, heart and spleen and in rare cases in the bone marrow.8 Melanomas are very rare in cats with only 4 diagnosed out of 3145 cats or 0.1% necropsied at the Animal Medical Center in New York. The most common site was intraocular with metastasis in 63% of the cases. Tumors thicker than two mm in human cases often metastasize to lymph nodes, skin, subcutaneous tissue, lung, liver, small intestine, pancreas, heart, brain, and spleen.7
The ability for melanomas to express a wide range of antigens complicates diagnoses. Antibodies key to differentiating melanomas include S-100, HMB-45, Mart-1 and MiTF.17 Many times IHC is necessary for a definitive diagnosis of melanomas due to their ability to be the ?great pretender?. In the present case, the neoplasm was undifferentiated and contained both spindloid and polygonal cells. The initial biopsy closely mimicked a basal cell carcinoma; however pan-cytokeratin stain was negative. To rule out melanoma, a Fontana-Masson stain was performed and was negative for melanin. Next, a panel of antibodies was used revealing the mass to be diffusely vimentin positive, yet only Mart-1 and S-100 positive for polygonal cells. The inconclusiveness of negative spindloid staining warranted TEM.
Transmission electron microscopy can be utilized to show melanin granules that may be few in number and small in size.4 The melanosomes generally are granular, myelin-like with shapes including oval, round, spindle-shaped, rod-like or irregular and are found singular not in groups. The present case demonstrated features of type II melanosomes (Figure 3).
Contributing Institution: National Institutes of Health, Division of Veterinary Resources, Office of Research Services, 9000 Rockville Pike, Building 28A, Room 106, Bethesda, Maryland, 20892
References
1. Beniashvili DS: Spontaneous rabbit melanoblastoma. Voprosy Onkologii 18:84-85, 1972
2. Bomhard W, Goldschmidt MH, Shofer FS, Perl L, Rosenthal KL, Mauldin EA: Cutaneous Neoplasms in Pet Rabbits: A Retrospective Study. Vet Pathol 44:579-588, 2007
3. Brown WH, Pearce L: Melanoma (sarcoma) of the eye in a syphilitic rabbit. J. Exp Med 43:807-813, 1926
4. Eyden B, Moss J, Shore I, Banerjee SS: Metastic small cell malignant melanoma: a case requiring immunoelectronmicroscopy for the demonstration of lattice-deficient melanosomes. Ultrastruct Pathol 29:71-78, 2005
5. Holz K, Heutgens W: Multiple Melanombildungen bein einem Kaninchen. Stsch Tierarztl Wochenschr 62:146-148, 1955
6. Hotchkiss CE, Norden H, Collins BR, Ginn PE: Malignant Melanoma in Two Rabbits. Lab Anim Sci 44:377-79, 1994
7. Hussein MR: Extracutaneous Malignant Melanomas. Cancer Investigation 26:516-534, 2008
8. Kim DY, Royal AB, Villamil JA: Disseminated Melanoma in a dog with involvement of Leptomeninges and Bone Marrow. Vet Pathol 46:80-83, 2009
9. Kurotaki T, Tomonari Y, Kanno T, Wako Y, Tsuchitani M: Malignant amelanotic melanoma behind the left eye in a female Crj:CD(SD)IGS Rat: A case report. Vet Pathol 45:681-84, 2008
10. Kusewitt DF, Applegate LA, Bucana CD Ley RD: Naturally occurring malignant melanoma in the South American opossum (Monodelphis domestican). Vet Pathol 27:66-68, 1990
11. Murphy J, Young S: Intraocular melanoma in a horse. Vet Pathol 16:539-42, 1979
12. Patnaik AK, Mooney S: Feline Melanoma: A comparative study of Ocular, Oral, and Dermal Neoplasms, Vet Pathol 25:105-112, 1988
13. Pawlowski A, Heaberman HF, Menon A: Skin melanoma induced by 7, 12-dimethylbenzanthracene in albino guinea pigs and it similarities to skin melanoma of Humans. Cancer Res 40:3652-3660, 1980
14. Pellegrini, G, Bienvenu JG, Meehan JT, Mischler SA, Perry RW, Scott DW, Anderson WI: Cutaneous melanoma with metastasis in a cynomolgus monkey (Macaca fascicularis). J Med Primatol 38:444-47, 2009
15. Sustmann: Multiple Melanombildungen bein Kaminchen. Dtsch Tierarztl Wochenschr 30:402, 1922
16. Terenziani M, Spreafico F, Serra A, Podda M, Cereda S: Amelanotic melanoma in a child with oculocutaneous albinism. Med Petiatr Oncol 41:179-80, 2003
17. Tong LCB, Kamil ZS, Habeeb A Al, Ghazarian D: Non-melanocytic mimics of melanoma, Part II: intradermal and intraepidermal mimics. J Clin Pathol 62:290-307, 2008
18. Tunev SS, Wells MG: Cutaneous melanoma in a Ferret (Mustela putorius furo). Vet Pathol 39:141-43, 2002