Conference 20, Case 4
Signalment:
5-year-old, female-spayed , mixed-breed dog (Canis familiaris)
History:
The patient presented to a veterinary medical teaching hospital with left-sided hemiparesis, acute onset of bilateral blindness and circling to the right. Bloodwork revealed no significant abnormalities. Upon hospital admission, the patient's obtundation progressed to stupor so euthanasia was elected . The owners reported that the patient had no significant clinical history other than diagnosis of and recovery from canine distemper at 1-year-old.
Gross Pathology:
No significant gross abnormalities were appreciated.
Laboratory Results:
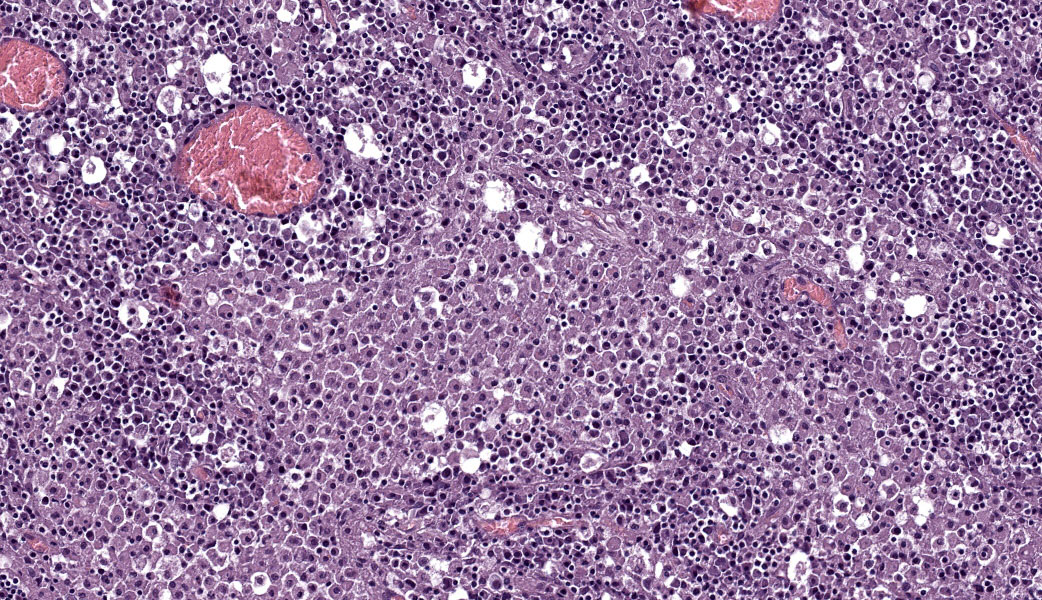
Canine distemper virus (Brain): Neurons and, less frequently, astrocytes exhibit strong cytoplasmic immunoreactivity.
Special stains: Luxol fast blue (Brain): Decreased positive staining supports demyelination of the cortical white matter.
Microscopic Description:
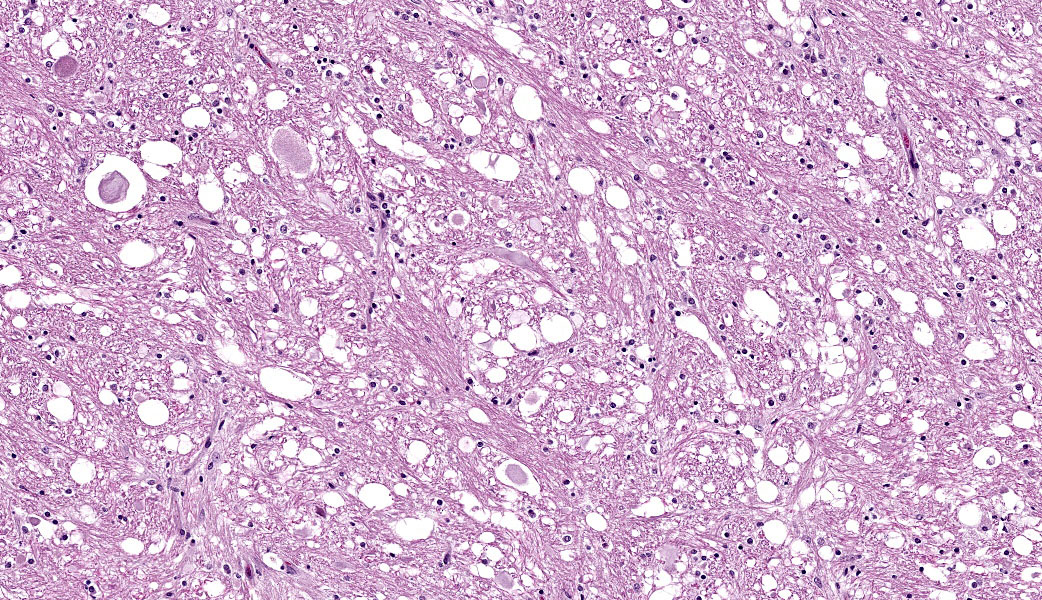
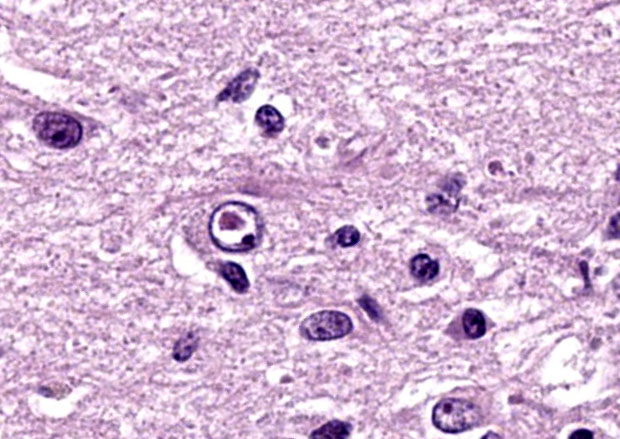
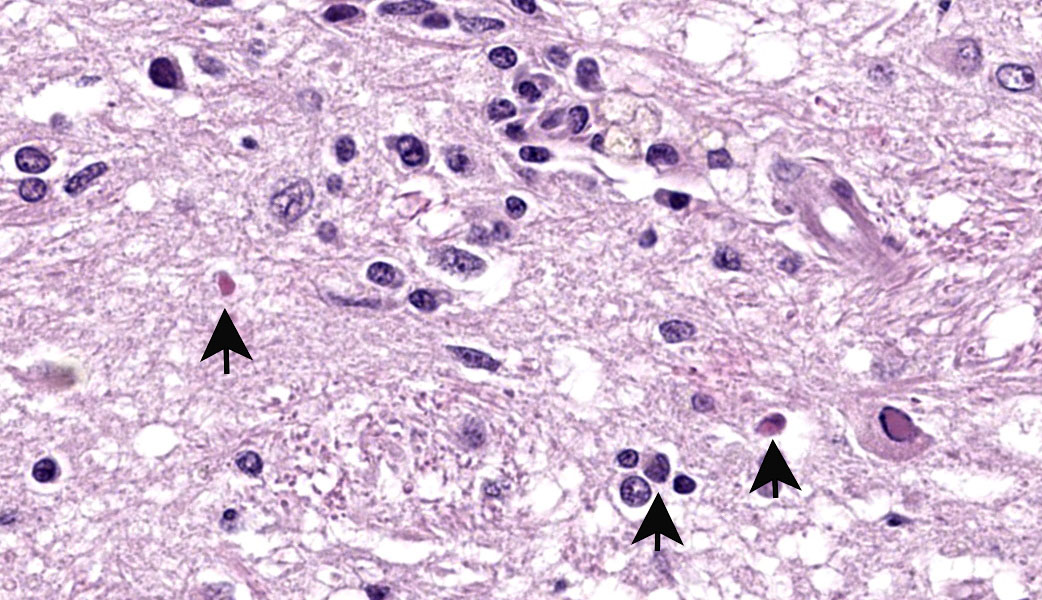
The slide contains one section of cerebrum at the level of the basal nuclei and corpus callosum. The white matter and, to a lesser extent, grey matter are moderately vacuolated and hypercellular. Vacuolation is defined by clear, well-demarcated vacuoles that reach up to 50 um diameter (spongiosis). The neuropil is additionally disrupted by increased numbers of proliferating glial cells (gliosis). Parenchymal vacuolation and gliosis of the grey matter is less severe than that described within the white matter. Neurons and astrocytes occasionally contain round intranuclear inclusion bodies that are approximately 5 um diameter and brightly eosinophilic. Morphologically-similar, intracytoplasmic inclusions are rare. Perivascular cuffing throughout the grey and white matter is characterized by mild to marked expansion of Virchow-Robbins space by small numbers of densely-packed lymphocytes and fewer plasma cells.
Contributor's Morphological Diagnosis:
Brain: Severe multifocal demyelination with lymphoplasmacytic perivascular encephalitis, gliosis and neuronal and astrocytic, intranuclear and intracytoplasmic inclusion bodies.
Contributor's Comment:
This case highlights an excellent example of canine distemper virus infection in the brain of a dog. Astrocytes and neurons contain both intracytoplasmic and intranuclear viral inclusion bodies, which are characteristic of Canine Distemper Virus (CDV) and other Paramyxoviruses including Newcastle Disease Virus, Measles Virus and Rinderpest Virus. The first supplementary image illustrates immunoreactivity for CDV within a neuron. The second supplementary image highlights demyelination and status spongiosis of the white matter, which has been proposed to be consequent to the direct effect of the virus on oligodendrocytes. Interestingly, there is no ultrastructural evidence of viral infection of oligodendrocytes.5 However, studies over the past decade have suggested that an axonopathy precedes demyelination, therefore promoting speculation that CDV encephalopathy is not a primary demyelinating disease.4
Four distinct presentations of CDV within the central nervous system have been characterized: classic canine distemper, multifocal distemper encephalomyelitis in mature dogs, old dog encephalitis and post-vaccinal canine distemper encephalitis.
Despite discrepancies in clinical presentation, initial disease pathogenesis remains similar across all four presentations. Upon inhalation, viral particles gain access to macrophages and epithelial cells through the signaling lymphocyte activation molecule (SLAM) and nectin-4 receptor, respectively. The virus travels within these infected leukocytes to regional lymph nodes and the tonsils, where viral replication occurs over the first twenty four hours of infection. Cell-associated viremia follows and the virus spreads to all lymphoid tissues and blood lymphocytes by five days post-infection. Viral infection of lymphoid tissues and lymphocytes results in lymphocytolysis and leukopenia, which ultimately leads to immunosuppression. Following initiation of immunosuppression, the clinical course depends largely on the host immune response, namely whether there is adequate, delayed/intermediate or a failure of humoral/cellular immunity.
Within the presented case, the patient's advanced age and reported history of canine distemper are most consistent with old dog encephalitis (ODE). Old dog encephalitis is rarely reported and therefore its pathogenesis remains poorly understood. Nonetheless, ODE is believed to develop within immunocompetent individuals with a subclinical, persistent infection. A replication-defective virus is suspected to persist within neurons of the central nervous system.2 Notably, a recent study provided evidence that non-cytolytic, cell-to-cell viral transmission occurs within astrocytes through a third receptor that is independent of SLAM and nectin-4.1
Although ODE appears to be a chronic disease, clinical presentation may be acute with sudden onset of neurologic abnormalities. This form of the canine distemper progresses over several months until the patient becomes comatose and eventually dies. At the time of this patient's workup, no effective treatment or preventative for ODE had been identified.
Contributing Institution:
University of California, Davis Veterinary Medical Teaching Hospital
https://www.vetmed.ucdavis.edu/hospital
JPC Morphologic Diagnosis:
Cerebrum: Demyelination, multifocal to coalescing, severe, with gliosis, perivascular cuffing, and neuronal and glial intranuclear and intracytoplasmic viral inclusions.
JPC Comment:
Wrapping up this conference was a stunning case of canine morbillivirus with readily identifiable viral inclusions. Canine distemper virus is a top infectious cause of demyelinating injury in dogs. This is due to direct infection and destruction of glial cells, such as oligodendrocytes (primary myelinating cell of the CNS), astrocytes, and microglia.3,5 CDV can also infect neurons, including Purkinje's cells and granular cells.3 The cerebellum is consistently affected in cases of CDV, with demyelinating lesions in the cerebellar white matter. Other common sites for similar lesions include the periventricular white matter (especially around the 4th ventricle) and the optic pathways.3 Occasionally, the white matter of the spinal cord may be involved. The Pesavento Lab also performed IHC on this case and was able to readily demonstrate the virus within glial cells and neurons in this case.
In acute and subacute cases of CDV, astrocytes and oligodendrocytes are infected in the early demyelinating phase. This manifests predominantly as vacuolation and/or dilation of myelin sheaths, demyelination, and axonal swelling (spheroids), +/- digestion chambers. The distribution is frequently multifocal but is usually concentrated in the caudal brainstem and cerebellum. The viral inclusion bodies of CDV (known as "Lentz bodies") can be seen either in the nucleus or cytoplasm of infected cells both within the brain and in other tissues.3,5
"Old dog encephalitis" (ODE) is a controversial condition. The paper that describes this condition had eight dogs total in the study, with one being experimentally infected.2 ODE is a disease of neurons and grey matter, especially in the cerebral cortex and thalamus, which contrasts starkly with the caudal brain predilection of acute/subacute CDV infection.2 Although viral antigen was densely expressed in neurons in the ODE paper, the demyelinating component of this disease was less pronounced.2 In cases of ODE, the cerebellum and caudal brainstem are comparatively spared.2 As such, there is still room for interpretation on the cause of ODE and the correlation, or lack thereof, with replication-deficient CDV infection.
CDV manifests in a variety of other ways outside of the CNS due to remarkable pleocellular tropism. These include pustular dermatitis and nasodigital hyperkeratosis/parakeratosis (also known as "hard pad disease"), enamel hypoplasia/dysplasia, ocular manifestations, myocardial necrosis, and metaphyseal osteosclerosis. Metaphyseal osteosclerosis occurs as CDV infects osteoclasts directly, causing a growth retardation lattice (also known as a "double line") within the bony trabeculae due to lack of trabecular resorption during bone growth/maturation. Currently, it is unknown which tissue sample is the most sensitive for testing due to CDV's incredibly broad cellular tropism. Another historical ruleout for spongiosis in the white matter of a dog that was mentioned by one participant (old enough to remember when it was banned by the FDA is hexachlorophene, which also results in a similar spongiotic and demyelinating injury).
References:
- Alves L, Khosravi M, Avila M, et al. SLAM- and nectin-4-independent noncytolytic spread of canine distemper virus in astrocytes. Journal of Virology. 2015;89:5724-5733.
- Axthelm M, Krakowka D. Experimental old dog encephalitis (ODE) in a gnotobiotic dog. Veterinary Pathology. 1998;35:527-534.
- Carvalho OV, Botelho CV, Ferreira CG, Scherer PO, Soares-Martins JA, Almeida MR, Silva Junior A. Immunopathogenic and neurological mechanisms of canine distemper virus. Adv Virol. 2012;2012:163860.
- Lempp C, Spitzbath I, Puff C, et al. New aspects of the pathogenesis of canine distemper leukoencephalitis. Viruses. 2014;6:571-601.
- Vandevelde M, Zurbriggen A. The neurobiology of canine distemper virus. Veterinary Microbiology. 1995;44:271-280.