Signalment:
Gross Description:
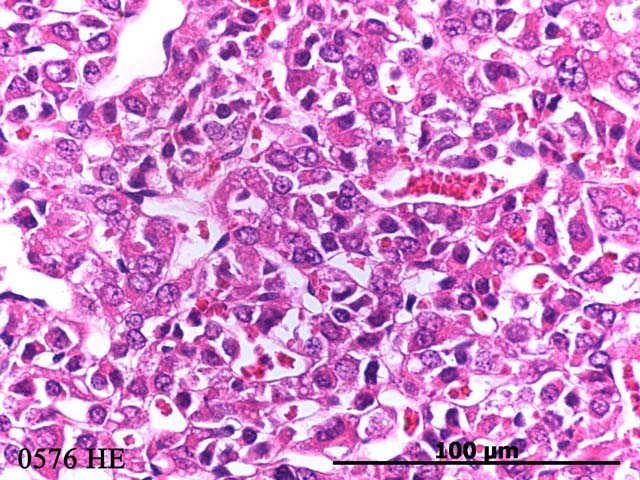
Histopathologic Description:
The nasal pharynx and nasal cavity are full of blood admixed with fibrin. At the level of the eyes, nasal cavity and upper molars, there is a unilateral focal area where one molar and the adjacent tissue are altered. There is a single large hair (whisker) and other cross sections of hair. The large hair extends from within the oral cavity along the medial aspect of one molar sulcus and is embedded into the overlying soft tissues of the palate. Surrounding the larger piece of hair there is a thin layer of stratified squamous epithelium and beyond this there is abundant fibrous connective tissue admixed with neutrophils, macrophages, and scattered lymphocytes and plasma cells. At the margin of the oral cavity there are greater numbers of neutrophils. There are similar inflammatory cells surrounding the smaller sections of hair. There is marked loss of bone in the subjacent maxilla. The adjacent molar occlusal surface shape is altered with the lingual aspect longer and curved toward the buccal side. There is loss of the roots and intervening bone with no visible pulp cavity. There is an inflammatory infiltrate comprised predominantly of neutrophils at the base of the root. In the ipsilateral buccal mucous tunic to the affected molar, the oral mucosa is absent and the underlying lamina propria is acutely necrotic with a large amount of fibrin and hemorrhage admixed with blood.
While the pituitary mass was considerable in size, it appears to have been an incidental finding in this animal, as the cause of impending death was respiratory failure resulting from the large blood clot within the nasal cavity. This can occur in rats because the anatomy of the oronasal cavity of rats is unique rendering them obligate nose-breathers. In rats, the soft palate is long, and the anterior opening of the esophagus, the epiglottis, and the larynx lies anterior to the nasopharyngeal opening. Therefore, an obstruction in this area would prevent a rat from being able to breathe.
Morphologic Diagnosis:
Lab Results:
Table 4-1. Complete Blood Count and Serum Biochemistry Panel Results
| Complete Blood Count | Results | Reference Range (Units) | Serum Biochemistry Panel | Results | Reference Range (Units) |
| Hemoglobin | 10.4 L | 11.4-19.2 g/dL | Glucose | 465 H | 60-125 mg/dL |
| Hematocrit | 28.0 L | 33-50 % | Urea Nitrogen | 22 | 9-30 mg/dL |
| WBC | 6.8 | 5.5-11 x103/uL | Creatinine | 0.5 | 0.4-1.0 mg/dL |
| RBC | 5.53 | 5.5-10.5 x106/uL | Total protein | 6.4 | 4.5-6.5 g/dL |
| MCV | 51 | Fl | Albumin | 3.2 | 2.0-6.2 g/dL |
| MCH | 18.8 | Pg | Total bilirubin | 0.1 | 0-1 mg/dL |
| MCHC | 37.1 | g/dL | Alkaline Phosphatase | 91 H | 15-45 U/L |
| Platelet Count | 634 | x103/uL | ALT | 69 H | 10-35 U/L |
| Platelet Estimate | Increased | AST | 177 H | 10-45 U/L | |
| Differential | Units | Cholesterol | 122 | 50-250 mg/dL | |
| Neutrophils | 4148 | 61% | Calcium | 12.0 | 8-12 mg/dL |
| Bands | 0 | Phosphorous | 10.8 H | 4.2-8.5 mg/dL | |
| Lymphocytes | 2448 | 36% | Sodium | 142 | 140-160 mEq/L |
| Monocytes | 136 | 2% | Potassium | 7.1 H | 4.3-5.8 mEq/L |
| Eosinophils | 68 | 1% | Chloride | 93 | 90-110 mEq/L |
| Basophils | 68 | 1% | Albumin/ Globulin Ratio | 1.0 | 0.4-1.1 |
| Polychromasia | Slight | BUN/Creat Ratio | 44 | ||
| Prothrombin | Time | 13.1 Secs | Globulin | 3.2 | 2.5-4.8 |
| PTT | 14.3 | Secs | CPK | 241 | U/L |
| Fibrinogen | 262 | Mg/dL | |||
| D-Dimer | < 250 | Ng/mL |
Condition:
Contributor Comment:
Table 2. Endocrine cells and hormones of the anterior pituitary (3)
| Cell type | Hormone | Cell characteristics |
| Somatotroph (type 2 acidophil) | Growth Hormone | H&E: acidophilic granules PAS: negative EM: abundant, dense granules 350nm |
| Lactotroph (type 1 acidophil) | Prolactin | H&E: acidophilic or chromophobic granules PAS: negative EM: sparse, dense granules 600-900nm |
| Gonadotroph (type 2 basophil) | Follicle-stimulating Hormone Leutinizing Hormone | H&E: basophilic PAS: positive EM: dense granules 200-250nm |
| Thyrotroph (type 1 basophil) | Thyroid-stimulating Hormone | H&E: basophilic PAS: positive EM: dense granules 150nm |
| Corticotroph (type 3 basophil) | Adrenocorticotropic Hormone | H&E: basophilic PAS: weakly positive EM: variably dense granules 200-400nm Cytoplasmic filaments |
| Melanotroph | Melanocyte-stimulating Hormone | H&E: basophilic PAS: positive |
Most pituitary tumors are adenomas and grow by expansion thereby creating a space-occupying lesion potentially interfering with the normal function of the cells within the pituitary, hypothalamus, thalamus and other surrounding structures. The clinical signs associated with the lesion are often linked to which types of hormones are secreted. Although they occur infrequently, metastatic pituitary neoplasms have been reported in a variety of species. These lesions can produce destructive effects on the pituitary, hypothalamus and thalamus leading to a multitude of clinical signs.(3)
Even though pituitary carcinomas occur with much less frequency than pituitary adenomas in most rats, in this case the tumor invasion into the overlying brain suggests that this tumor is a carcinoma. In addition, the fact that the tumor cells had high pleomorphism with bizarre forms suggests carcinoma.Â
In a study by McComb, et al (1984), it was found that in rats over 24 months of age, pituitary adenomas were found in 85% of male and 79% of female SD rats. Of these tumors, 47% were prolactin (PRL)-containing and 16% were leutinizing hormone (LH)-containing adenomas. The remaining 37% were made up of tumors containing thyroid-stimulating hormone (TSH), growth hormone (GH), adrenocorticotropic hormone (ACTH) or some combination thereof, as well as immunonegative adenomas.(5) In another study done by Nagatani, et al (1987), 736 rats of various inbred strains ranging from 13 to 24 months of age were screened for pituitary tumors. Pituitary tumors were found in 284 of the 736 rats, with some rats having more than one lesion.(3) In addition to spontaneously occurring pituitary tumors, chronic estrogen treatment can induce prolactin-secreting tumor growth in the anterior pituitary of Fischer 344 rats.(8)
In dogs, ACTH-secreting tumors are the most common of the functional pituitary tumors. While these tumors can arise from the pars intermedia or the pars distalis, most commonly they are chromophobic adenomas composed of either large or small cells arising from the pars distalis. ACTH-secreting tumors frequently result in the development of adrenocortical hyperplasia and hyperfunction and cause pituitary-dependent Cushing's disease.(3) While cats are not considered a species that commonly develops pituitary tumors, one study found that 16 out of 16 diabetic cats with insulin resistance also had pituitary adenomas manifesting as acromegaly or hyperadrenocorticism.(2)
The most frequently occurring pituitary neoplasm in horses is the adenoma of the pars intermedia leading to a variety of clinical signs including hirsutism, polyphagia, muscle wasting, hyperglycemia, and diabetes insipidus, among others.(1)
In humans, pituitary neoplasms represent approximately 10% of the intracranial tumors, the most common of which are prolactin secreting pituitary adenomas.(3,5,4) Spontaneous pituitary adenomas have also been described in parakeets and mice.(5)
JPC Diagnosis:
Conference Comment:
References:
2. Elliot DA, Feldman EC, Koblik PD, Samii VF, Nelson RW: Prevalence of pituitary tumors among diabetic cats with insulin resistance. JAVMA, 216 (11): p. 1765-1768, 2000
3. Jones TC, Hunt RD and King NW: Veterinary Pathology, 6th ed., Williams & Wilkins, Baltimore, MD, pp. 1224-1232, 1997
4. McComb DJ, Hellmann P, Kovacs K, Scott D, Evans WS, Burdman JA, and Thorner MO: Spontaneous sparsely-granulated prolactin-producing pituitary adenomas in aging rats: Neuroendocrinology, 41: 201-211, 1985
5. McComb DJ, Kovacs K, Beri J, and Zak F: Pituitary adenomas in old sprague-dawley rats: a histologic, ultrastructural, and immunocytochemical study. JNCI, 19(5): p 1143-1157, 1973
6. Nagatani M, Miura K, Tsuchitani M, Narama I: Relationship Between Cellular Morphology and Immunocytological Findings of Spontaneous Pituitary Tumours in the Aged Rat. J Comp Path, 97: 11-20, 1987
7. Percy DH, Barthold SW: Pathology of Laboratory Rodents and Rabbits. 3rd ed., pp.108, 122, 173-174, 306. Blackwell Publishing, Ames, IA, 2007
8. Wendell DL, Platts A, Land S: Global Analysis of Gene Expression in the Estrogen Induced Pituitary Tumor of the F344 Rat. J Steroid Biochem & Mol Biology 101: 188-196, 2006