Joint Pathology Center
Veterinary Pathology Services
Wednesday Slide Conference
2017-2018
Conference 18
February 7th, 2018
CASE I: 1704415 (JPC 4101081).
Signalment: 21-year-old, female, American Quarter Horse (Equus caballus), equine.
History: The mare had a well-circumscribed hairless mass in the right nuchal region. The mass was reported to be pruritic and was excised surgically and submitted whole for histopathologic evaluation.
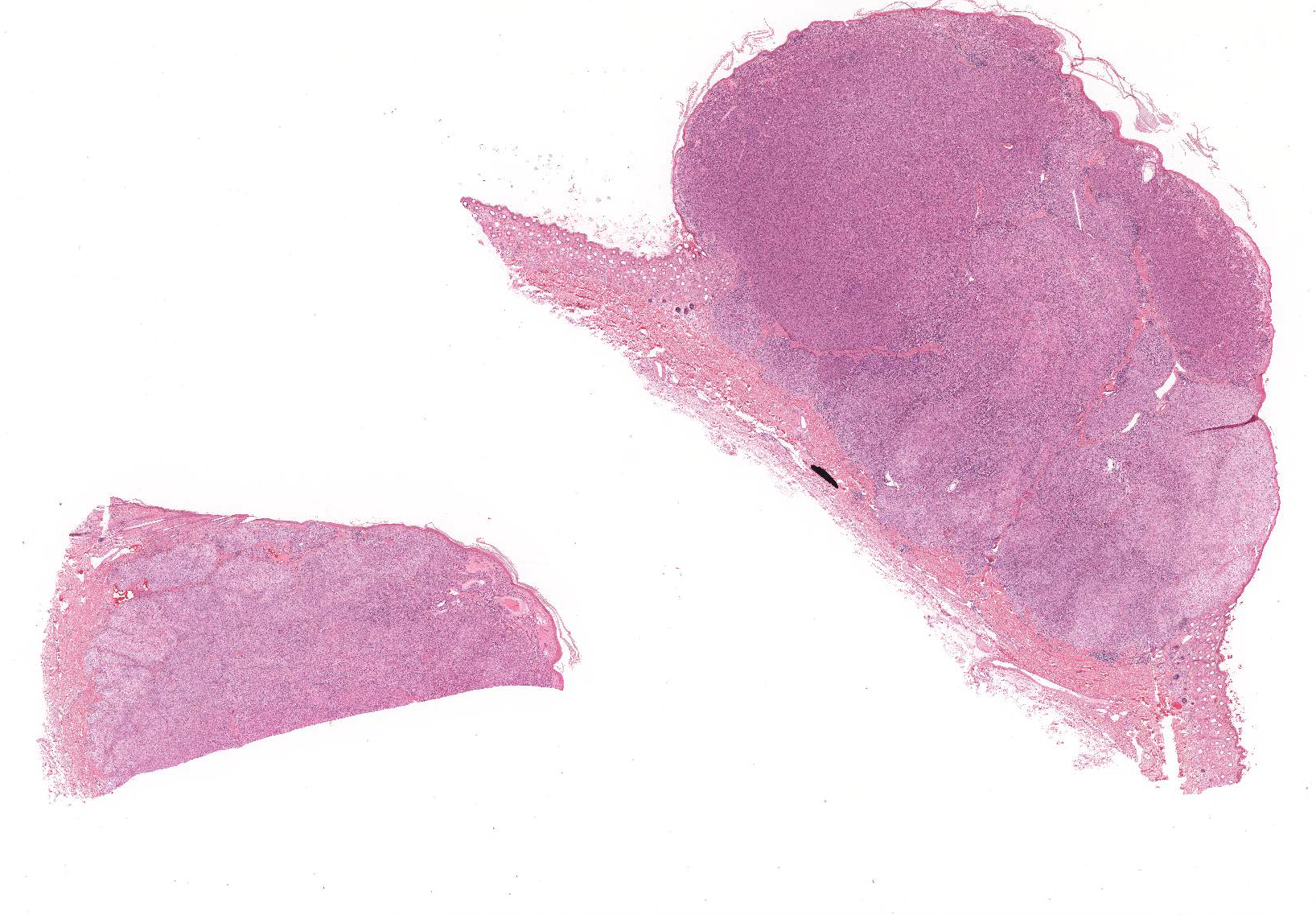
Gross Pathology: The submitted formalin-fixed sample consisted of haired skin with an approximately 14 mm diameter, slightly firm, tan, raised and hairless mass that markedly expanded the superficial dermis and elevated the epidermis.
Laboratory Results (clinical pathology, microbiology, PCR, ELISA, etc.):
None provided.
Microscopic Description:
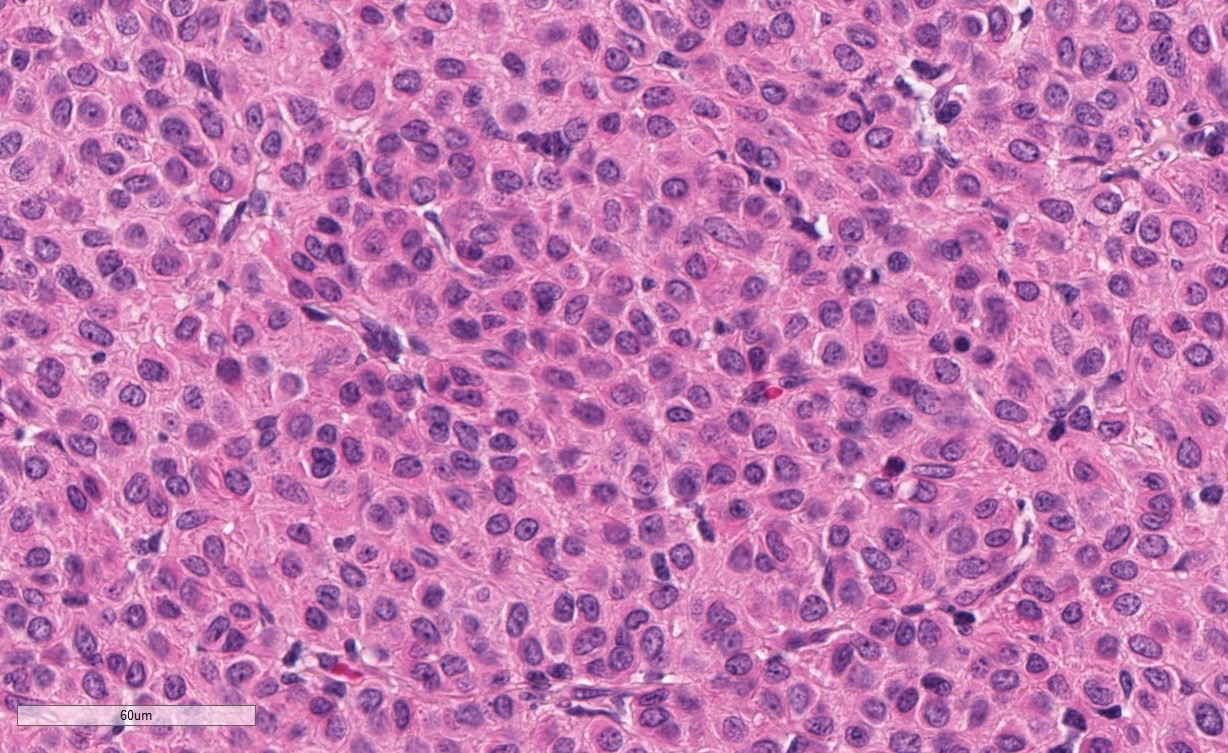
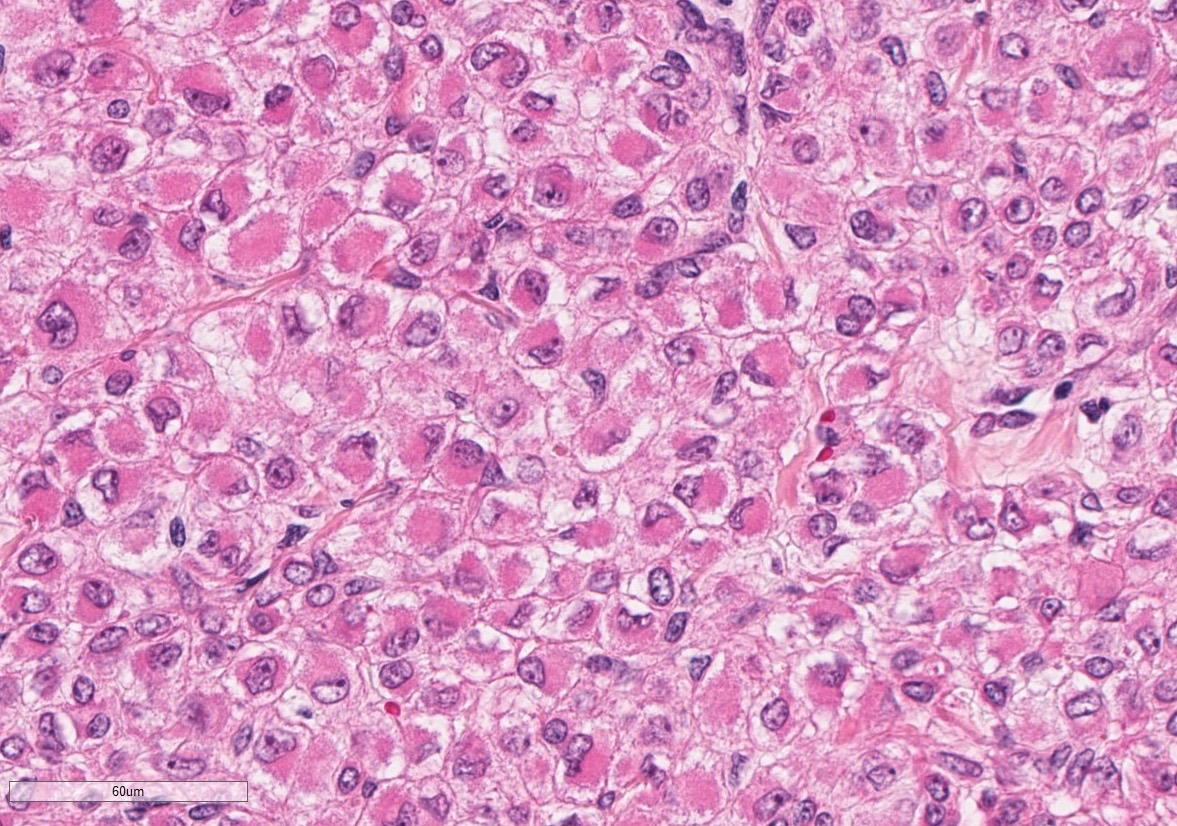
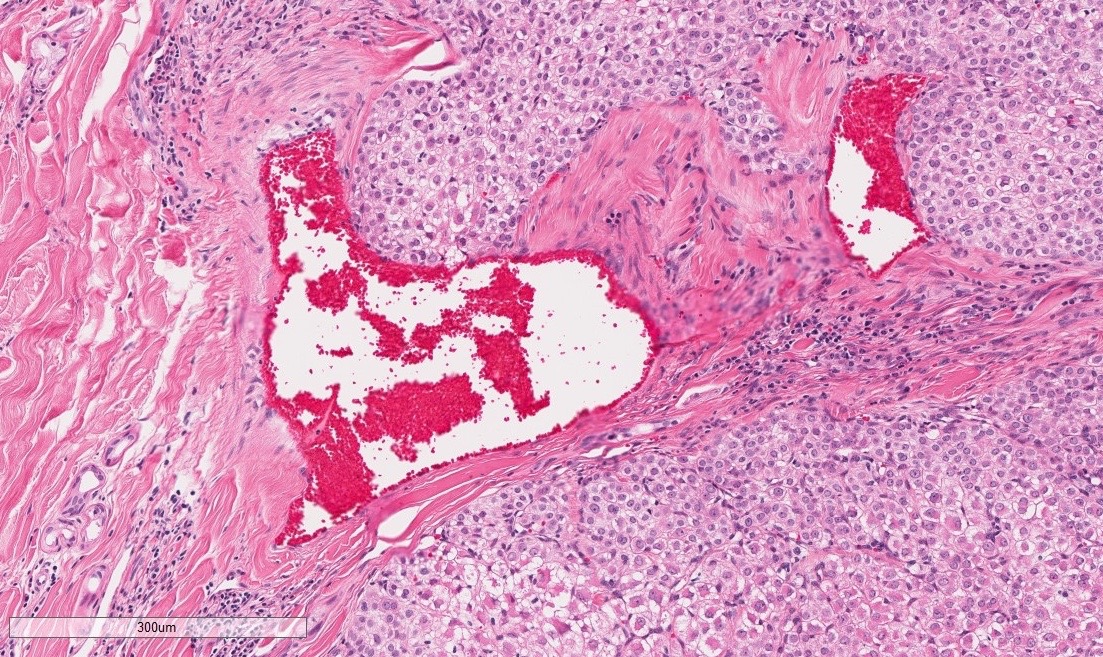
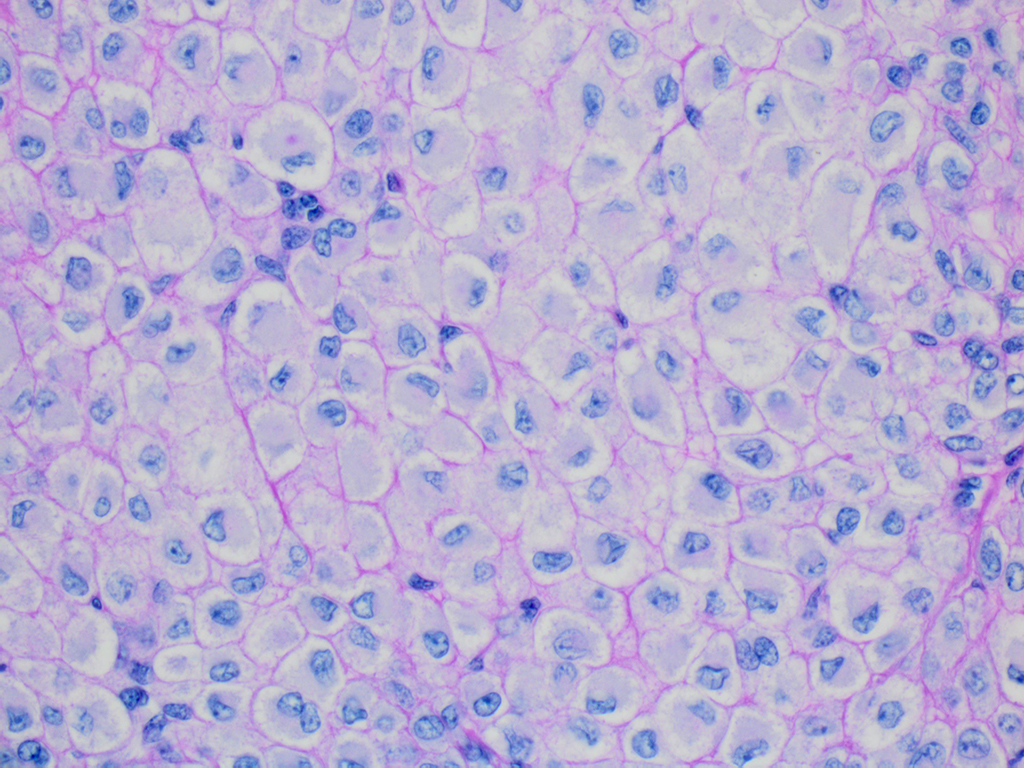
The superficial dermis is focally expanded by an unencapsulated, multilobulated and densely cellular neoplasm that replaces adnexal units and elevates the epidermis. The neoplastic cells have variable arrangements, ranging from dense sheets and nests to trabeculae or thin ribbons separated by a delicate fibrovascular stroma. Multifocally throughout the neoplasm, cells closely abut or palisade along vascular channels and impinge on the lumina without disrupting the endothelium. The cells are cuboidal to polygonal with variably distinct cell borders, a moderate amount of pale eosinophilic cytoplasm, and a round to ovoid or irregular nucleus with finely stippled chromatin and a small inconspicuous nucleolus. Occasionally, the cells have abundant hypereosinophilic cytoplasm that peripheralizes the nucleus (epithelioid-type cells). Anisocytosis and anisokaryosis are mild to moderate and mitotic figures are infrequent, with an average of 0 to 1 per single high power field (12 per 50 consecutive 40X high power fields). Binucleation and individual cell necrosis are occasionally observed. Low numbers of lymphocytes and plasma cells multifocally infiltrate the intervening stroma. The overlying epithelium is mildly hyperplastic with orthokeratotic hyperkeratosis and is focally ulcerated.
A periodic acid-Schiff (PAS) stain highlights a thin PAS-positive basement membrane surrounding individual or small nests of cells.
Immunohistochemistry:
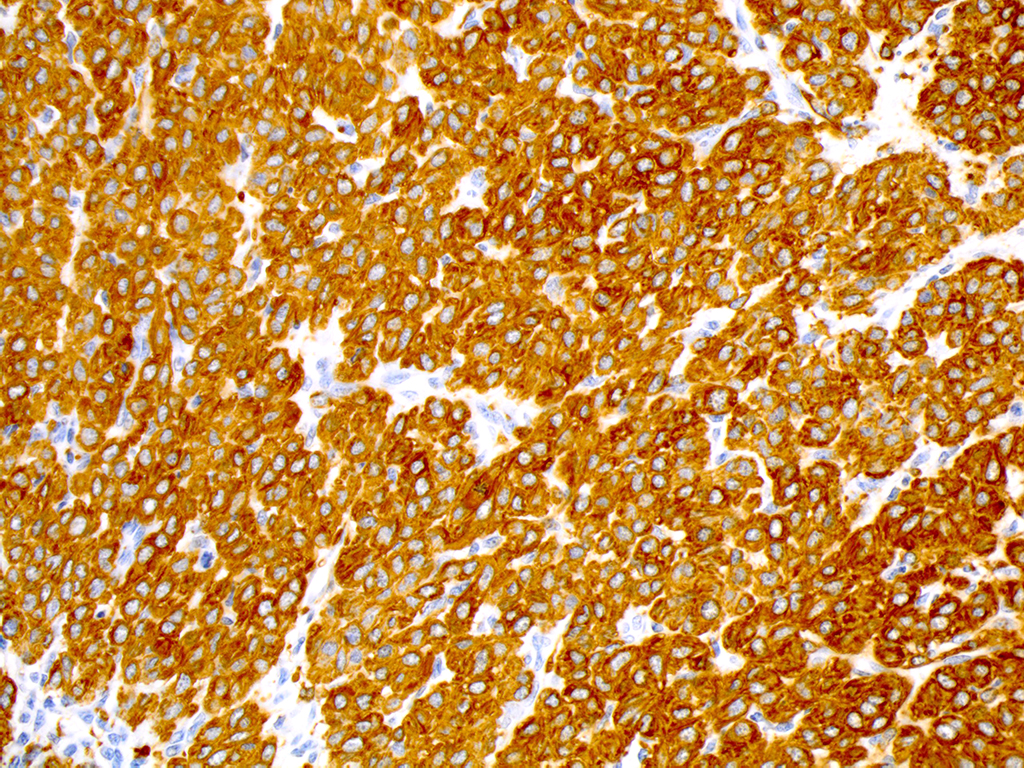
The neoplastic cells exhibit diffuse and strong positive cytoplasmic immunoreactivity for alpha-smooth muscle actin (α-SMA), vimentin, and desmin, and are negative for pancytokeratin (AE1/AE3) and CAM 5.2.
Contributor’s Morphologic Diagnosis:
Horse, cutaneous mass: glomus tumor
Contributor’s Comment: Glomus tumors (GT) are rare, typically benign neoplasms that are thought to arise from the modified perivascular smooth muscle cells of the glomus body, a structure that plays a role in body temperature regulation by allowing arterio-venous shunting of blood and is most commonly found in the subungual regions, subcutis of the extremities, or other specific locations such as the precoccygeal region.5,9
Glomus tumors in humans are usually solitary tumors that appear as small circumscribed nodules in the deep dermis or subcutis of the upper and lower extremities, with the subungual region of the finger being the most common location. Many other, less common locations have been described, such as the gastrointestinal tract, penis, urinary bladder, lungs etc.5
The clinical diagnosis in humans is based on the typical red-blue appearance, combined with a history of paroxysmal pain from cold exposure and light touch, regardless of the size of the tumor.4,5 Depending on their histologic appearance, GT are classified as classic (or sporadic/solid), glomangiomas, glomangiomyomas, glomangiomatosis and symplastic GT, which have a higher grade of nuclear atypia.4 The classic GT are by far the most common type and account for approximately 75% of tumors.5 Although predominantly benign, a small subset of tumors displays clinical or histological features of malignancy.3 Folpe et al., in 2001, proposed the following classification system for this subset of tumors. Malignant GT, or glomangiosarcoma, are tumors larger than 2 cm in diameter and located deep in the subcutis and underlying tissues, or that have atypical mitotic figures, or a moderate to high nuclear grade and ≥ 5 mitotic figures/50 consecutive 40x high power fields. Symplastic GT have a high nuclear grade in the absence of other malignancy features. Tumors that lack the criteria for malignant GT or symplastic GT, but exhibit one of the following characteristics, high mitotic activity and superficial location, or large size, or deep location only, are classified as GT of uncertain malignant potential. Finally, glomangiomatosis are tumors resembling diffuse angiomatosis with increased numbers of glomus cells. Only malignant GT were associated with metastasis, which included the brain, bone, lung, liver, small intestine, mediastinal lymph nodes, and bowel mesentery.3
Histologically, classic glomus tumors are well-defined, multilobulated, often partially encapsulated neoplasms composed of nests of small rounded cells often closely associated to capillaries and supported by a hyalinized or myxoid stroma. Cells have eosinophilic cytoplasm and a round, centrally located nucleus.4 A more oncocytic or epithelioid appearance of cells is occasionally reported.4 Cellular and nuclear pleomorphism and mitoses are not prominent features.4 Immunohistochemical staining is consistently positive for α-smooth muscle actin and vimentin.8
Similarly to humans, glomus tumors in domestic animals are rare and typically benign, and have been described in non-human primates, cats, dogs, cows, and horses.7,10,11 In horses, these tumors have been reported to occur in the subcutis of the head and neck and in the foot.1,2 A single case of glomus tumor with neuroendocrine differentiation was reported in the maxilla of a 13-year-old Icelandic crossbred mare.10 Most cases reported in horses displayed features of malignancy, either by local invasion, or by cytological criteria, according to the human classification of malignant glomus tumors.1-3,10 However, no metastasis to the local lymph nodes or distant sites were reported.
In the case presented herein, the preliminary diagnosis of a trichoblastoma was ruled out based on negative immunoreactivity for pancytokeratin (AE1/AE3) and CAM 5.2, while the positive immunoreactivity for alpha-smooth muscle actin (α-SMA), vimentin, and desmin instead supported a diagnosis of glomus tumor. Given the mitotic activity and nuclear pleomorphism, the tumor in this horse would be classified as a malignant glomus tumor (glomangiosarcoma) based on the human classification criteria. The malignant behavior of glomus tumors reported in horses was typically associated with increased cellular pleomorphism and the difficulty to achieve a complete surgical excision.1,2,10 In the present case, the small circumscribed nature of the neoplasm and complete surgical excision suggest a favorable prognosis.
JPC Diagnosis: Haired skin: Glomus tumor, American Quarter Horse (Equus caballus), equine.
Conference Comment: Glomus tumors arise (not surprisingly) from glomus cells which are specialized modified smooth muscle cells controlled by the sympathetic nervous system. They are typically associated with arteriovenous shunts or anastomoses that act to regulate flow within the shunt vessels and influence temperature regulation. In humans, these are frequently found in extremities.6 In animals, glomus tumors are most frequently reported in dogs where they also tend to occur along extremities. Additionally, there have been isolated reports in cats, horses, non-human primates, and several cows.2,7,9,11 In horses, there appears to be a predisposition for the head or neck (as seen in this case); however, examination of additional cases is necessary to determine whether there is indeed a true site predilection.2 A recent case described in a Holstein-Friesian cow identified a large primary glomus tumor within the liver.7
Microscopically, glomus tumors are well-demarcated, dermal or subcutaneous masses with a fibrous capsule that often contains subcapsular vessels and nerve fibers. In domestic animals and humans, most are considered round cell type, or glomus tumor proper, in which round cells are densely packed with centrally located, prominent nuclei and moderate amounts of pale eosinophilic cytoplasm. Rarer are the spindle cell type in which cells are more elongated with oval nuclei. In both types, mitotic figures and multinucleated cells are few, and each cell or groups of cells is surrounded by a PAS-positive basement membrane. Ultrastructurally, the basement membrane can be seen as well as actin-like filaments, cytoplasmic dense bodies, pinocytotic vesicles, and glycogen granules.6
Differentials for the round cell type of glomus tumor include: Merkel cell tumors, plasma cell tumors, nonepitheliotropic lymphomas, histiocytomas, mast cell tumors, and transmissible venereal tumors (TVTs). In general, all of the above differentials lack the association with peripherally located blood vessels and nerve fibers that are prominent in glomus tumors. Additionally, Merkel and plasma cell tumors have prominent “packeting” of cells which are separated by a fine stroma and not surrounded by basement membrane. The location of the nuclei can separate glomus tumors from plasma cell tumors and histiocytomas. Also, histiocytomas have a characteristic “top heavy” appearance and often have intraepidermal neoplastic cells. Mast cells can be clearly identified using toluidine blue or Giemsa stains to highlight metachromatic cytoplasmic granules. Finally, lymphomas and TVTs are most difficult to distinguish based on H&E cytologic characteristics alone. Due to its smooth muscle origin, glomus cell tumors retain positivity for vimentin, α-smooth-muscle actin, and pan-muscle-specific actin which will be negative in lymphomas and TVTs.6
Contributing Institution:
University of Pennsylvania
School of Veterinary Medicine
Department of Pathobiology
http://www.vet.upenn.edu/research/academic-departments/
References:
- Brounts SH, Adams SB, Vemireddi V, Holland CH. A malignant glomus tumour in the foot of a horse. Equine Vet Educ. 2008;20:24-27.
- Burns RE, Pesavento PA, McElliott VR, Ortega J, Affolter VK. Glomus tumours in the skin and subcutis of three horses. Vet Dermatol. 2011;22:225–231.
- Folpe AL, Fanburg-Smith JC, Miettinen M, Weiss SW. Atypical and malignant glomus tumors: analysis of 52 cases, with a proposal for the reclassification of glomus tumors. Am J Surg Pathol. 2001;25:1-12.
- Folpe AL, Brems H, Legius E. Glomus tumours. In: Fletcher CDM, Bridge JA, Hogendoorn PCW, Mertens F. World Health Organization Classification of Tumours: WHO Classification of Tumours of Soft Tissue and Bone, 4th ed., Lyon, France: International Agency for Research on Cancer; 2013:116-117.
- Goldblum JR, Folpe AL, Weiss SW. Perivascular Tumors In: Enziger and Weiss’s Soft Tissue Tumors, 6th ed., Philadelphia, PA: Saunders Elsevier; 2014:749-765.
- Gross TL, Ihrke PJ, Walder EJ, Affolter VK. Perivascular tumors. In: Skin Diseases of the Dog and Cat Clinical and Histopathological Diagnosis. 2nd Ames, IA: Blackwell Science Ltd.; 2005:759-762.
- Horiuchi N, Komagata M, Shitamura K et al. Glomus tumor of the liver in a cow. J Vet Med Sci. 2015;77:729–732.
- Mravic M, LaChaud G, Nguyen A, Scott MA, Dry SM, James AW. Clinical and histopathological diagnosis of glomus tumor: an institutional experience of 138 cases. Int J Surg Pathol. 2015;23:181–188.
- Park CH, Kozima D, Tsuzuki N, Ishi Y, Oyamada T. Malignant glomus tumour in a German shepherd dog. Vet Dermatol. 2009;20:127-130.
- Peters M, Grafen J, Kuhnen C, Wohlsein P. Malignant glomus tumour (glomangiosarcoma) with additional neuroendocrine differentiation in a horse. J Comp Pathol. 2016;154:309-313.
- Roperto S, Borzacchiello G, Brun R et al. Multiple glomus tumors of the urinary bladder in a cow associated with bovine papillomavirus type 2 (bpv-2) infection. Vet Pathol. 2008;45:39–42.