CASE I:
Signalment:
Canine fetus and placenta. 40 days gestation, Dog, Welsh Corgi Pembroke (Canis lupus familiaris)
History:
Two fetuses and placenta. Previous litter same stud - 4 pups - normal gestation. No new dogs brought into kennel in past year. Another dam bred to same stud had a litter of 12 with 4 mummified fetuses, a 3rd dam aborted 4 fetuses around day 40 gestation - bred to same stud as the other two females. No other previous history of abortion in kennel. Dam had temperature of 103 °F day after abortion. Physical examination of dam unremarkable. No vaccine history at clinic.
Dogs will go next door and scavenge chicken carcasses from time to time.
Gross Pathology:
No significant external lesions were present on the puppies. Puppy A was enclosed in an intact placenta, which had no visible lesions.
Laboratory Results:
Microbiology: Campylobacter jejuni 3+ cultured from lung and placenta.
Microscopic Description:
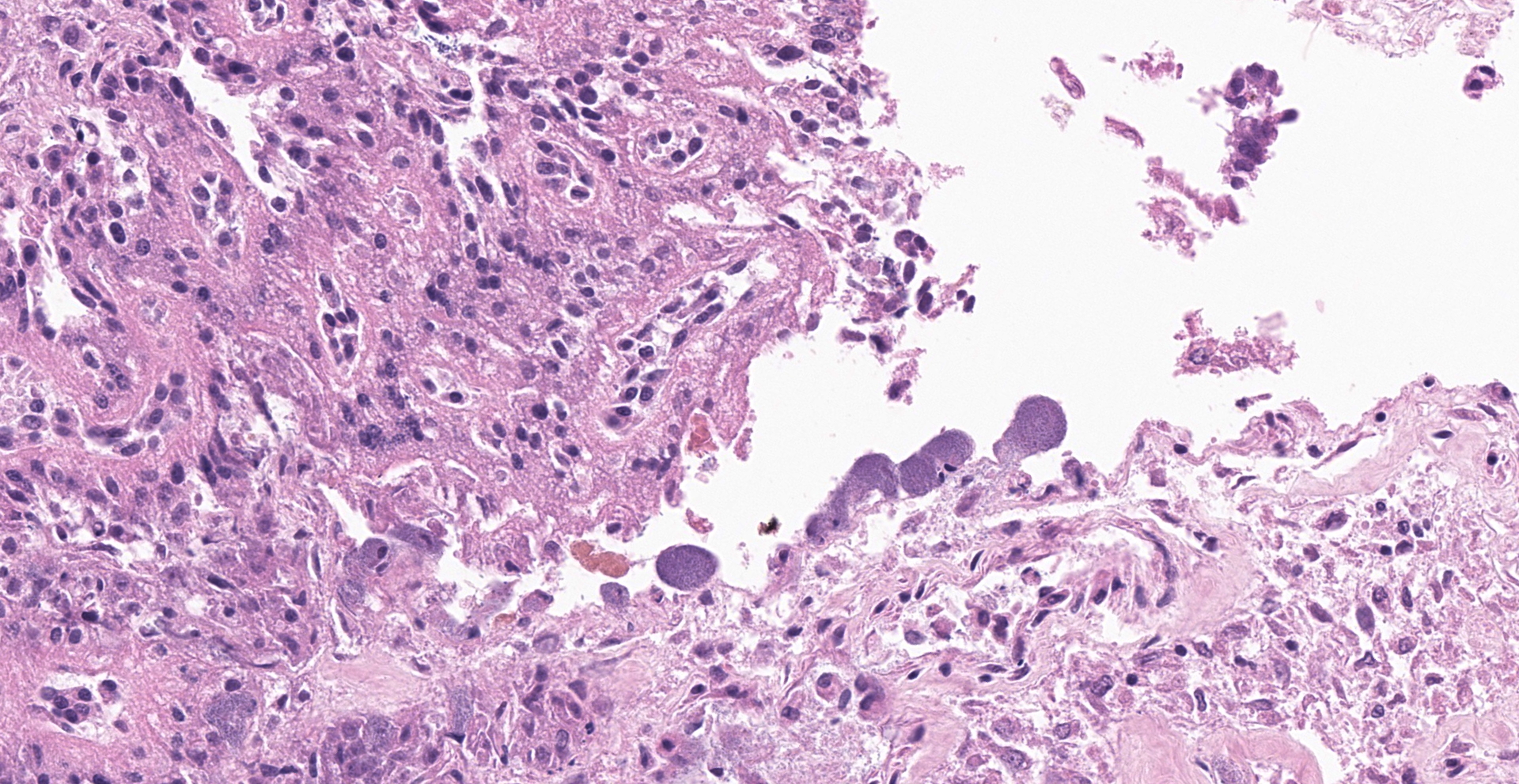
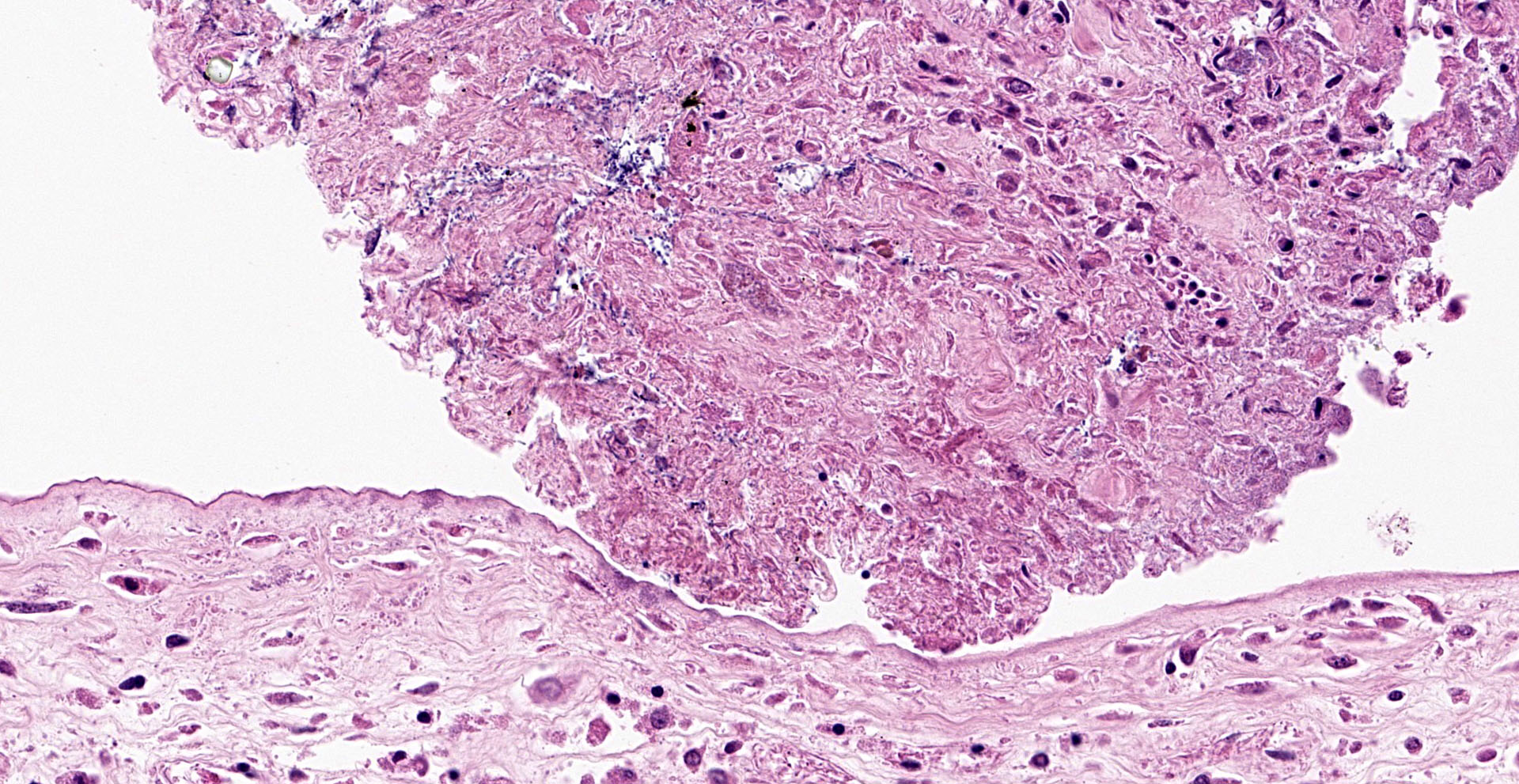
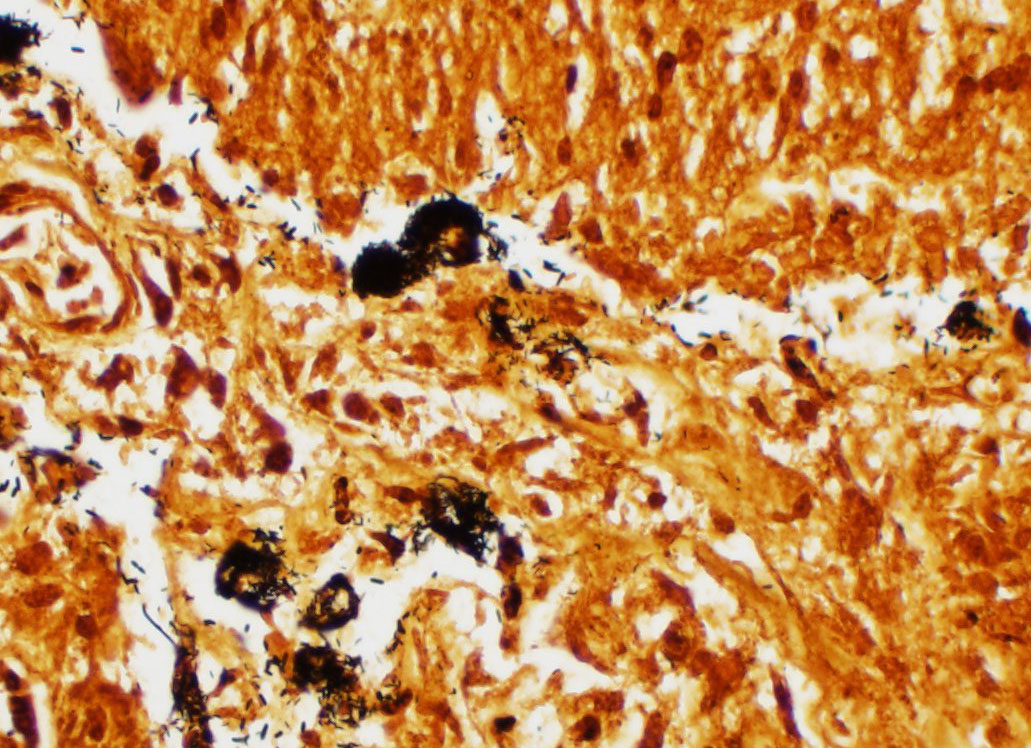
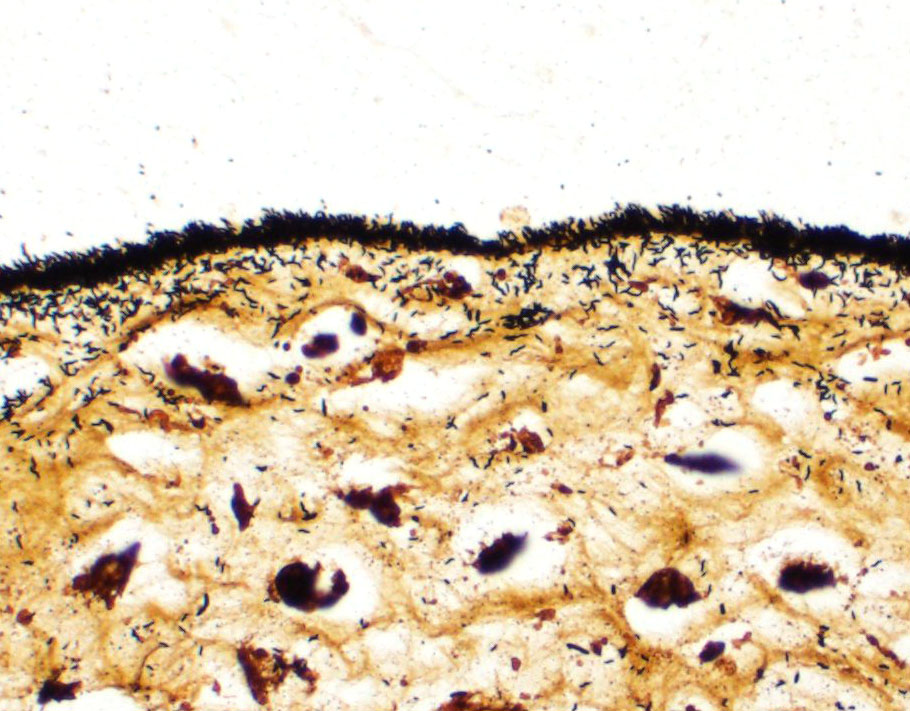
Lesions present in the placenta are very subtle and involve the region of the marginal hematoma and chorioallantois. At the edge of the placental labyrinth at the region of the marginal hematoma there is proteinaceous material that is eosinophilic or hypereosino-philic and clumped. Around or within this are 20 µm diameter circular to oval structures with a defined outer boundary and containing multiple basophilic stippled material (trophoblasts filled with bacteria). Attached to and part of the non-labyrinthine chorion are trophoblasts with a distended cytoplasm and fine basophilic stippling (intratropho-blastic bacteria). In places there are no trophoblasts and there is basophilic stippling of the eroded surface. Neutrophils and or macrophages in the adjacent membrane are generally minimal. Throughout the chorioallantois, in particular, and between amnion and allantois in the amniotic membrane are histiocytic cells that sometimes form clusters. There are extra cells within the walls of blood vessels or adjacent tissue. Some clumps of bacteria are within small blood vessels.
Contributor's Morphologic Diagnoses:
Intratrophoblastic bacteria of non-labryinthine placenta and marginal hematoma of placenta, and mild fibrinous, necrotic and histiocytic placentitis.
Contributor's Comment:
The lesions of bacterial failure of pregnancy, including abortion, in dogs are generally very subtle. The most important change is the presence of trophoblasts distended by bacteria. These are typically found at the tips of the labyrinthine villi and particularly in the region of the marginal hematoma. Inflammatory changes are usually mild and involve low numbers of neutrophils in the chorion near the distended trophoblasts, and mononuclear cells, particularly histiocytic cells, in other parts of the placenta particularly the allantois/amnion. The lesions in puppies can be variable, with multifocal neutrophilic pneumonia and hepatitis being reported.1,3,4
The lesions of Brucella canis are well reported in naturally acquired and experimental cases.2 Even then, intratrophoblastic bacteria are the predominant change with minimal inflammation being present. There are a couple of individual case reports of Campylobacter jejuni as a cause of reproductive failure with abortion and perinatal mortality.1,3,4 These cases may be accompanied by maternal illness including diarrhea, and diarrhea in human contacts. Acquisition of Campylobacter jejuni is reported from chicken carcasses or raw food diet, however subclinical persistent carriers do occur. The incubation period is typically short and of the order of several days. Bacteremia and localization in the placenta, phagocytosis by trophoblasts, fibrinous inflammation and then infection results in abortion, stillbirth or perinatal mortality.
Contributing Institution:
Department of Pathobiology
Ontario Veterinary College
University of Guelph
JPC Diagnosis:
Placenta, edge of marginal hematoma: Placentitis, necrotizing, multifocal to coalescing, mild, with intratrophoblastic, intravascular, and extracellular bacilli.
JPC Comment:
The contributor provides a concise overview of the histologic features and pathogenesis of canine abortion due to Campylobacter jejuni, an entity more commonly associated with reproductive failure in ruminants. C. jejuni is also the leading bacterial etiology associated with human foodborne gastroenteritis in both North America and Europe.5
Additional Campylobacter sp. associated with ruminant reproductive failure include C. fetus subsp. fetus and C. fetus subsp. veneralis. Although closely genetically related, there is significant variability between the two C. fetus subspecies in regard to their respective host range and epidemiologic features. C. fetus subsp. veneralis' distribution is essentially limited to the bovine reproductive tract and is the leading cause of venerally transmitted infectious infertility and embryotic death in this species. Conversely, both C. fetus subsp. fetus and C. jejuni have broad host ranges, often colonize the gastrointestinal tract of cattle, sheep, and goats as commensals, and are associated with abortion in sheep and cattle.5
McFadyean and Stockman first reported Campylobacter's association with ovine abortion following its isolation from an aborted sheep fetus in 1906. The genus has since been identified as a worldwide cause of infertility and abortion in ruminants. C. jejuni and C. fetus subsp. fetus are associated with 10.3-25.2% of abortions in sheep, making Campylobacter one of the one of the leading causes of ovine abortion. Abortions typically occur during the last trimester and may affect up to 50% of ewes, with the disease colloquially known as epizootic abortion. Goats and cattle are more sporadically affected by C. jejuni and C. fetus subsp. fetus, with these entities being detected in 0.9-1.8% of aborted caprine fetuses and only 0.19% cattle fetuses were positive for C. jejuni during a study of approximately 9,000 aborted fetuses. However, abortion storms have also been reported in both goats and cattle.5
Historically, C. fetus subsp. fetus was the leading species of Campylobacter associated with ovine abortion worldwide. However, C. jejuni became the leading cause of Campylobacter abortion in ewes in the United States during the 2000's and is becoming increasingly common around the world. C. fetus subsp. fetus continues to be the primary species causing ovine abortions in the United Kingdom and New Zealand, although C. jejuni is also a major cause of ovine abortion in these countries as well.5
Campylobacter isolates associated with ovine abortion have historically demonstrated a high level of genetic diversity. However, recent studies have revealed the vast majority of ovine abortions in the United States are attributed to a single C. jejuni clone since identified as SA for "sheep abortion". A unique feature of this clone is its capability to cause both systemic infections and abortions, features that have been replicated in a well-established laboratory model for Campylobacter-induced ovine abortions, the pregnant guinea pig.5
Ewes are commonly infected by C. fetus subsp. fetus and C. jejuni via the fecal-oral route as well as from aborted fetuses and fetal membranes. Most flocks are not affected by abortion storms despite Campylobacter's presence on many farms. However, abortion storms are often associated with the introduction of animals from flocks with previous history of abortion into a naïve flock, which may affect up to 70% of ewes. Large numbers of Campylobacter are associated with the products of abortion, particularly the placenta and uterine discharge, and the bacteria may be continually shed in vaginal secretions for weeks afterward. Additional ewes often become infected after licking aborted fetuses. Affected flocks often have a history of one or two abortions followed by an abortion storm.5
Bulls asymptomatically harbor C. fetus subsp. veneralis in the epithelial crypts of the penis and prepuce, which is subsequently venerally transmitted cows during breeding. C. fetus subsp. veneralis infection does not prevent fertilization in infected cows, instead resulting in endometriosis, salpingitis, and embryonic death most often between 15-80 days gestation, with a delayed return to estrus in up to 50% of cows in a herd. C. fetus subsp. veneralis may also result in abortions between 4-7 months of gestation in less than 10% of infected cows, likely due to smaller doses of inoculation or slower than typical bacterial replication. In contrast, neither C. fetus subsp. fetus nor C. jejuni are transmitted venerally by rams nor do they result in an ascending infection of the reproductive tract.
Many animals carry C. fetus subsp. fetus and C. jejuni in the intestinal tracts and gall bladder but do not demonstrate clinical disease. However, when animals carrying these Campylobacter sp. become immunocompromised (such as pregnant ewes), the bacteria undergo translocation with systemic distribution and subsequent colonization of the uterus, resulting in placentitis, fetal infection, and abortion, most often during the final trimester.5
Macroscopic lesions of the placenta are often apparent but subtle, with intercotyledonary thickening, edema, congestion, and opacity while the cotyledons often have a superficial brown-red exudate. Gross findings in the fetus may include serosanguinous fluid within the body cavities with strands or mats of fibrin and there may be hepatic rupture with hemoabdomen. In addition, a very characteristic (although not pathognomonic) gross lesion associated with Campylobacter in sheep is a liver with multifocal, pale, circular to targetoid foci measuring up to 4cm in diameter. However, this lesion is only present in approximately 25% of cases and is also associated with Flexispira rappini, another commensal of the ovine gastrointestinal tract associated with sporadic ovine abortion.5
The most common histologic lesions associated with Campylobacter abortion in sheep is placentitis (100%) which is often necrosuppurative and associated with placental vasculitis. As in this case, the most characteristic features are the presence of bacteria within trophoblasts, adjacent stroma, and within vascular channel remnants. Additional common findings include fetal pneumonia (92%), gastroenteritis (54%), serositis (41%), hepatitis (35%), and encephalitis (8%).5
The gold standard for diagnosis of Campylobacter abortion in ruminants is culture in conjunction with the previously described gross and histologic lesions. Optimal locations of culture collection include placenta and fetal stomach contents, followed by fetal lung, and fetal liver.5
In regard to canines, the first case report of Campylobacter-associated abortion originated from the northwestern United States in 1984, with the second case report emerging a decade later in South Africa, both of which described late term abortions. Although the pathogenesis C. jejuni-induced abortion in canines is unclear, it is suspected to closely mirror the process previously described in sheep since both species are asymptomatic carriers of the pathogen and are affected by late term abortion.4
Based on the moderator's experience, Campylobacter sp. infection often presents with two unique histologic features in both canine and ruminant placenta; 1) the superficial aspect of the chorioallantois (most commonly the allantois in ruminants) has a "fuzzy" appearance due to myriad bacteria adhered to the superficial aspect; and 2) bacterial emboli within blood vessels. These findings are in addition to the presence of intratrophoblastic bacteria; however, this feature is also associated with other bacterial etiologies known to cause canine abortion, including (but not limited to) Brucella canis, Listeria monocytogenes, and Salmonella spp. Feline abortion may also occur as the result of infection from the same etiologies, with the exception of Brucella canis. In addition, the moderator discussed felines being implicated as a source of zoonotic Coxiella burnetii infections, with the organism being isolated from the placenta from feline abortions.
The moderator discussed common features of bacterial canine placentitis. Inflammation is most often present in the non-zonary chorioallantois but is almost always mild and subtle, even in the face of dramatic infection. Key regions to evaluate for infected trophoblasts include those adjacent to the marginal hematoma and the surface of the zonary placenta.
Finally, participants noted the presence of haired skin within the submitted section of placenta. The moderator noted that since conjunctival lesions can be an indication of amnionitis some institutions include fetal eyelids during regular tissue collection.
References:
1. Bulgin MS, Ward AC, Sriranganathan N, Saras P. Abortion in the dog due to Campylobacter species. Am J Vet Res. 1984;45(3):555-556.
2. Carmichael LE, Kenney RM. Canine abortion caused by Brucella canis. J Am Vet Med Assoc. 1968;152(6):605-616.
3. Odendaal MW, de Cramer KG, van der Walt ML, Botha AD, Pieterson PM. First isolation of Campylobacter jejuni from the vaginal discharge of three bitches after abortion in South Africa. Onderstepoort J Vet Res. 1994;61(2):193-195.
4. Sahin O, Burrough ER, Pavlovic N, Frana TS, Madson DM, Zhang Q. Campylobacter jejuni as a cause of canine abortions in the United States. J Vet Diagn Invest. 2014;26(5):699-704.
5. Sahin O, Yaeger M, Wu Z, Zhang Q. Campylobacter-Associated Diseases in Animals. Annu Rev Anim Biosci. 2017;5:21-42.