Signalment:
Gross Description:
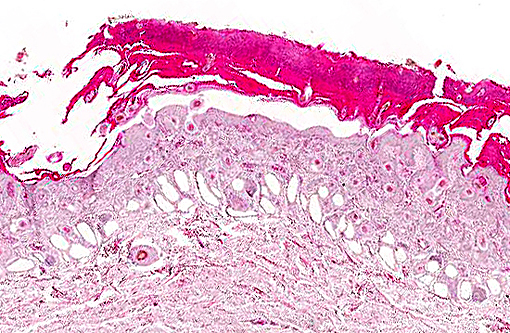
Histopathologic Description:
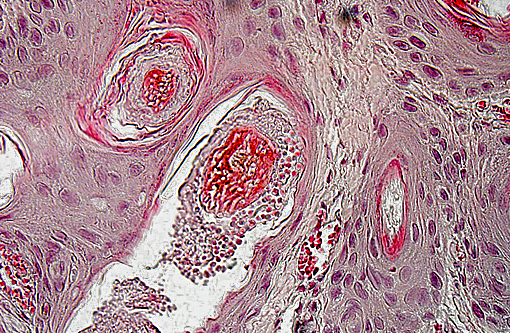
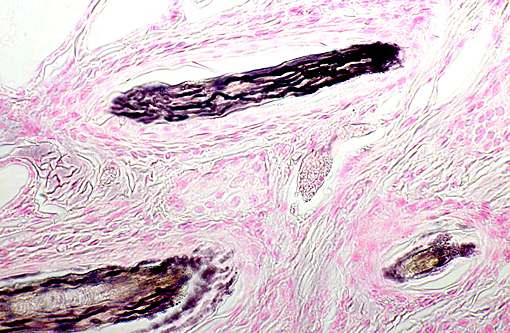
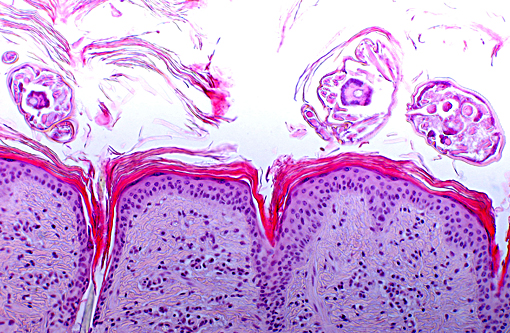
Within dilated hair follicles and around free hair shafts in epidermal crusts are numerous 2-3 μm sized basophilic arthrospores and hyphae with thin parallel walls and indistinct branching and septation (Trichophyton sp.). Often, hair follicles are additionally mildly expanded by clumped keratin (infundibular hyperkeratosis). Around blood vessels in the upper dermis is a mild inflammatory infiltrate composed of eosinophils, of lymphocytes and as well as of lesser neutrophils and macrophages. Sweat glands are mildly dilated.
Morphologic Diagnosis:
Lab Results:
Condition:
Contributor Comment:
Transmission occurs through contact while arthrospores may survive in the environment for more than a year. Dermatophytes colonize the superficial dermis and hair follicles and they are adapted to digesting keratin.(4,5) Histologically, dermatophytes are more easily detectable using PAS method or Grocott stain. Septated hyphae are detectable between the keratin of the stratum corneum and within the hair shafts. There is regularly a mild acanthosis and hyperkeratosis. Inflammatory changes are often mild.(1) Vaccination is effective for prophylaxis and treatment of bovine ringworm.(6)
Scab mites cause a highly pruritic dermatitis and psoroptic mange is like demodicosis of high economic importance in bovine species. In some countries it is a reportable disease.(1,3) Psoroptes ovis is the assumed species of mites responsible for psoroptic mange in cattle.(8) Mites can survive for about 7-12 weeks outside the host. Transmission results directly by contact or indirectly by vectors (e.g. bedding, contaminated objects like feed fences or cow brushes). Psoroptes mites complete their life cycle on the skin. In contrast to Sarcoptes sp., they do not invade the epidermis.(7)
Lesions in cattle can be detected at the skin of the head, neck, back, inguinal and sacral areas and may become generalized. They are characterized by alopecia, crusts, scales and exudation. Due to severe pruritus, excoriations and secondary bacterial infections may blur the picture. In more severe cases the skin shows increased thickness with diffuse alopecia and deep folds.(1,3,7)
In accordance with the clinical symptoms, histologically there is superficial perivascular dermatitis with eosinophils, lymphocytes, macrophages and mast cells. Due to an assumed hypersensitivity reaction there is often spongiosis and marked dermal edema. In chronic cases, hyperplasia of the epidermis can be observed.(1) Mites could be identified microscopically within skin scrapings or biopsies, but not with certainty.(7)
Sarcoptic mange is a differential diagnosis for the lesions in the present case. However, in the present case, the mites are detectable on the surface and between superficial keratin lamellae whereas Sarcoptes mites dig deeper. On the other hand there were no burrows filled with eggs and/or larvae within the layers of the epidermis, which are typical for Sarcoptes but not seen in Psoroptes infestation.(1)
In the present case, a combined infection with dermatophytes and mites occurred. The nutritional stage and concurrent debilitating diseases may have influenced the development of both infections. Both diseases, mange and dermatophytosis, are differentials for each other, and it has to be considered, that dermatophytosis is a zoonotic disease. In contrast to Sarcoptes mites Psoroptes mites are believed to be non-pathogenic for humans.(1)
JPC Diagnosis:
Conference Comment:
Dermatophytosis is more common in young animals and often associated with immunosuppression, which is supported in this case by the simultaneous symptoms of respiratory disease and oral ulcers. Notably, other than the neutrophils within the crusts, there is minimal inflammation present despite the fungal proliferation within nearly every hair shaft. Avoiding immune detection and subsequent provocation of an inflammatory response is instrumental to survival for these pathogens. The arthrospores are always along the periphery of the hair while hyphae are confined within the hair shaft.(3) The organisms never penetrate Adamsons fringe into the mitotic region of the follicle, and fungal growth is terminated along with growth of the hair. Dead telogen hairs, known as club hairs, are resistant to infection. Resolution of clinical signs typically occurs spontaneously within a few months.(3) This is in contrast to other fungal infections which invade the subcutaneous tissue, incite a tremendous inflammatory response, and often necessitate surgical excision such as pseudomycetomas, phaeohyphomycosis and sporotrichosis.(2)
References:
1. Ginn PE, Mansell JEKL, Rakich PM. Skin and appendages. In: Maxie MG, ed. Jubb, Kennedy, and Palmers Pathology of Domestic Animals. 5th ed. Philadelphia, PA: Elsevier Limited; 2007, vol 1:553-781.
2. Hargis AM, Ginn PE. The integument. In: Zachary JF, McGavin MD, eds. Pathologic Basis of Veterinary Disease. 5th ed. St. Louis, MO: Elsevier Mosby; 2012:1038-1039.
3. Scott DW. Large animal dermatology, Philadelphia, PA: WB Sauders Company; 1988, 174-182.
4. Chermette R, Ferreiro L, Guillot J. Dermatophytoses in animals. Mycopathologia. 2008;166(5-6):385-405.
5. Bond R. Superficial veterinary mycoses. Clin Dermatol. 2010;28(2):226-236.
6. Lund A, Deboer DJ. Immunoprophylaxis of dermatophytosis in animals. Mycopathologia. 2008 Nov-Dec;166(5-6):407-424.
7. Rommel M, Eckert J, Kutzer E, K+â-¦rting W, Schnieder T. Veterin+â-ñrmedizinische Parasitologie. 5th ed. Berlin: Parey; 2000.
8. Bates PG. Inter- and intra-specific variation within the genus Psoroptes (Acari: Psoroptidae). Vet Parasitol. 1999;83(3-4):201-217.