Joint Pathology Center
Veterinary Pathology Services
Wednesday Slide Conference
2019-2020
Conference 25
6 May, 2020
CASE I: 15/692 (JPC 4137583).Tissue from a horse (Equus caballus).
Signalment: 11 years, mare, American Quarter Horse, Equus caballus, horse.
History: The horse had a firm, well defined tumor on the left side of the neck. The tumor was excised for pathological examination.
Gross Pathology: A skin biopsy measuring 4x2x2 cm was received. There was a tumor in the dermis, not adherent to the epidermis, that consisted of a brown, homogeneous but somewhat lobulated, medium firm tissue. The tumor was surrounded by a thin sheath of connective tissue in the deeper areas and on the sides.
Laboratory results: None.
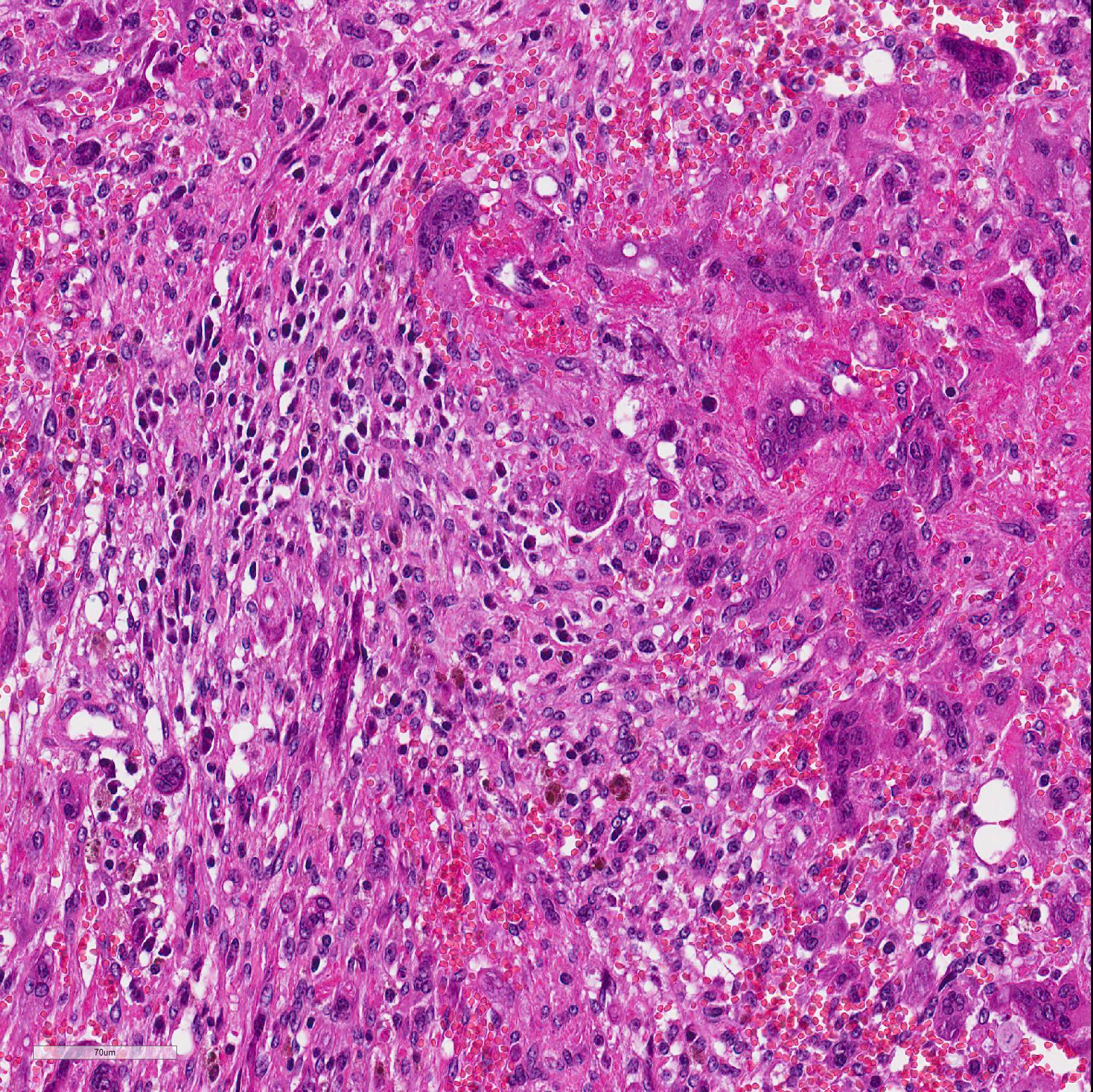
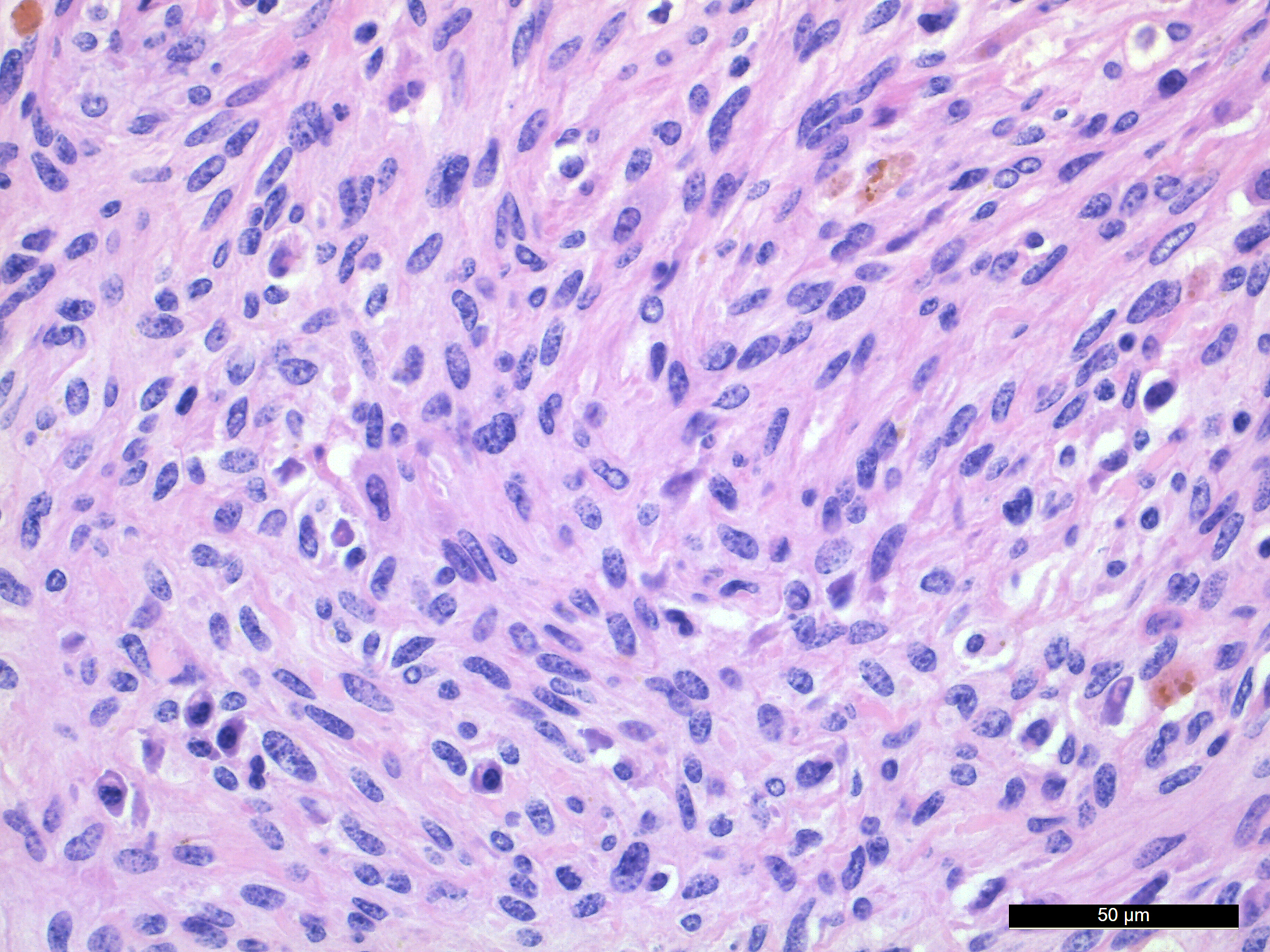
Microscopic Description: In the deep dermis of the skin there is a non-encapsulated, infiltrative, medium cell rich, moderately demarcated tumor that consists of spindle shaped cells and numerous multinucleated giant cells (MGCs), in sparse amounts of stroma. There are two growth patterns in the tumor, one with tightly packed spindle shaped cells growing in bundles with few MGCs and another with numerous MGCs and fewer loosely arranged spindle cells.
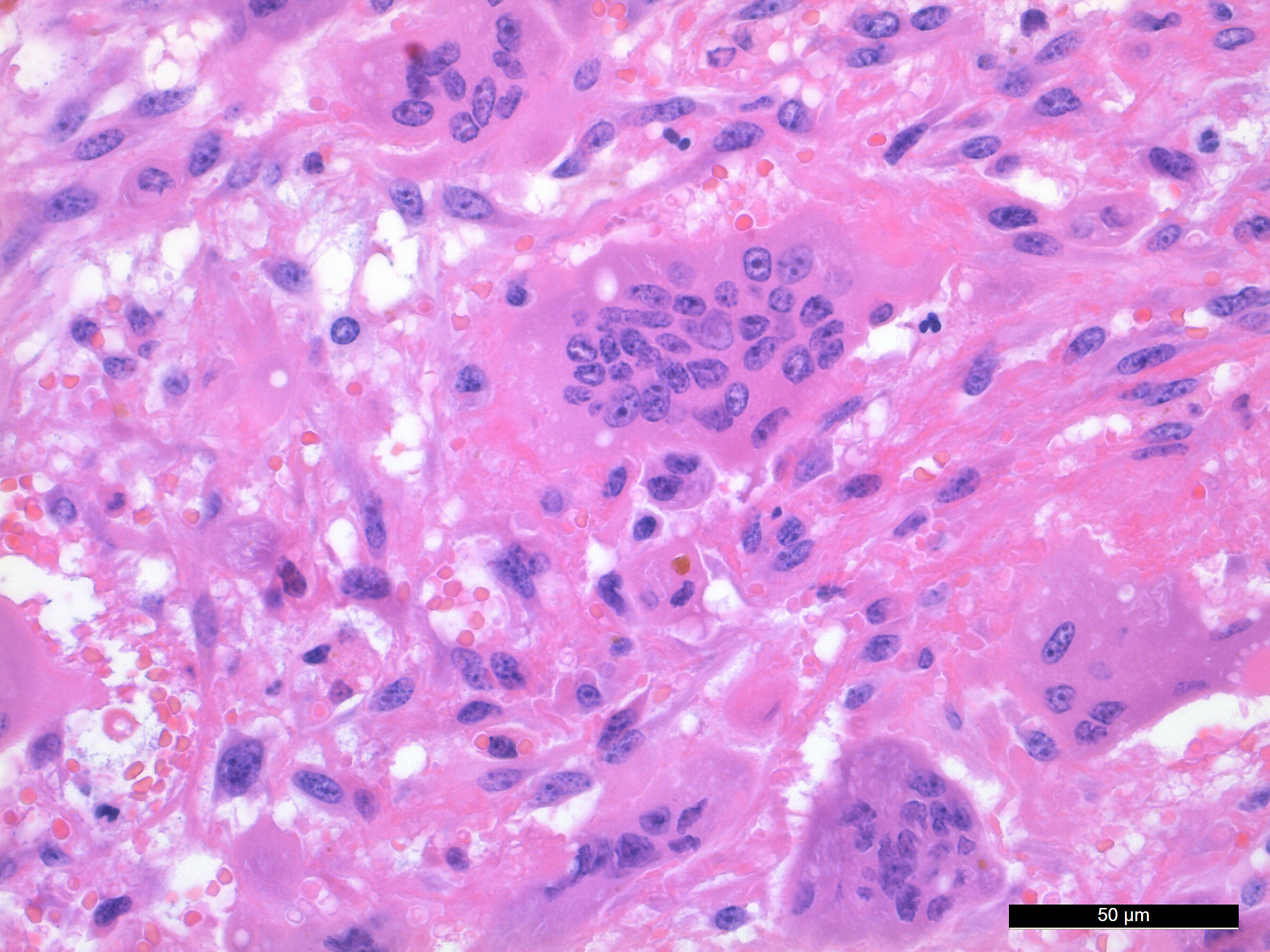
The spindle shaped tumor cells are medium sized, with small to moderate amounts of eosinophilic cytoplasm and an oval to cigar shaped nucleus with finely stippled chromatin and 1-3 small nucleoli. The spindle shaped cells have mild to moderate anisocytosis and anisokaryosis. The MGCs are elongated, oval or round, they have abundant eosinophilic cytoplasm and oval or round nuclei with finely stippled chromatin and 1-3 small nucleoli. The MGCs are highly variable in shape, size, and nucleus number (up to 37). Mitotic figures are 2 per 10 HPF.
There are some multifocal necrotic areas in the tumor tissue. The areas with numerous MGCs often have hemorrhage, and there are multifocal inflammatory cell infiltrates with lymphocytes, plasma cells and macrophages with yellow to brown pigment (hemosiderin). Small amounts of pigment is also present in the cytoplasm of some MGCs. No acid-fast mycobacteria was detected in a Ziehl-Neelsen stained section.
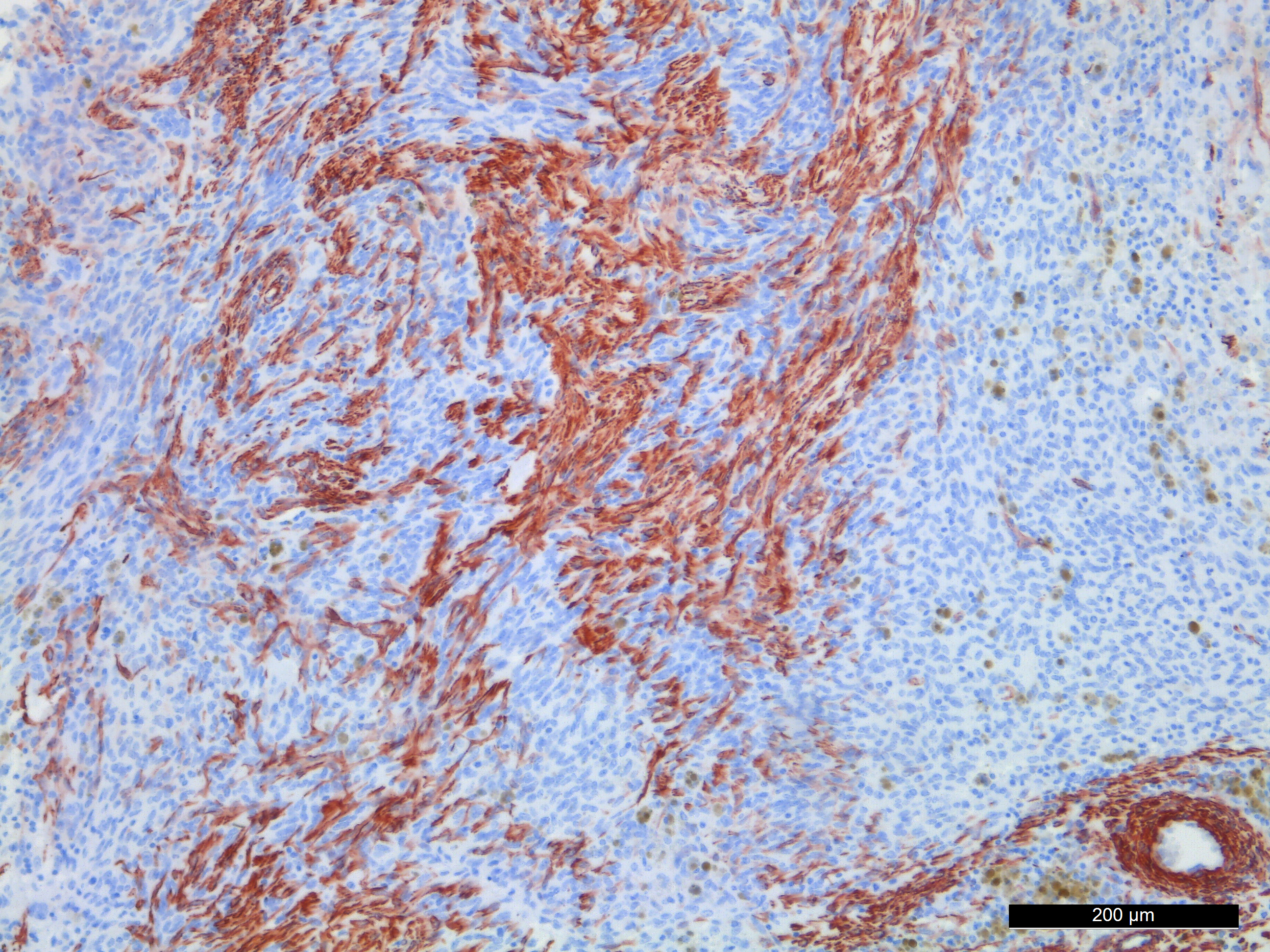
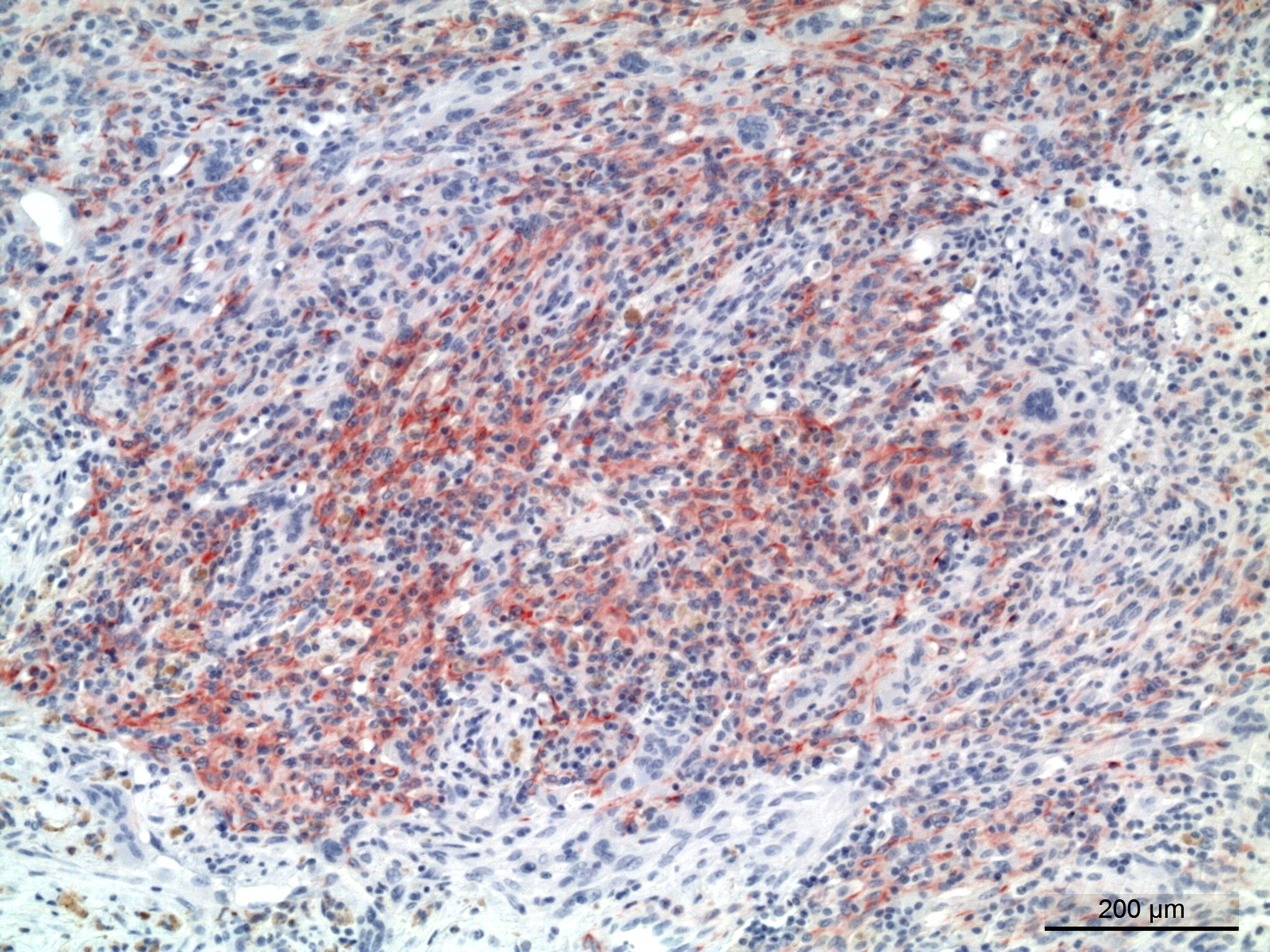
Immunohistochemistry staining with the muscle marker desmin showed positive staining in the spindle shaped cells. Immunohistochemistry staining with antibody for ? smooth muscle actin was variable. Areas with compact growth of spindle shaped cells had a combination of strongly positive cells and negative cells. In areas with numerous MGCs, the spindle cells were weakly positive. All MGCs were negative, in both desmin and ? smooth muscle actin stained slides.
Contributor Morphologic Diagnosis:
Skin: undifferentiated pleomorphic sarcoma with numerous MGCs
Contributor Comment: Undifferentiated pleomorphic sarcoma is a rare tumor that also has been termed malignant fibrous histiocytoma, giant cell tumor of soft parts, extraskeletal giant cell tumor and anaplastic sarcoma with giant cells. The cellular origin of the tumor cells are controversial. Sarcomas with combined spindle shaped cells, vacuolated histiocyte-like cells, and variable amounts of pleomorphic MGCs and a collagenous stroma have in the past often been diagnosed as malignant fibrous histiocytoma. But the term undifferentiated pleomorphic sarcoma is now preferred as these tumors most likely represent a diverse group neoplasms.5,6
A meta-analysis of 43 case reports and case series, totaling 82 human patients, showed that giant cell rich malignancies may be of highly variable origin, and may arise from both skeletal and many extraskeletal tissue types.4
The tumor in the present case is dominated by spindle shaped cells and numerous MGCs, with few mononuclear histiocyte-like cells. Also in areas with numerous MGCs, most other cells were spindle shaped. The spindle shaped cells were immunohistochemically positive for desmin and variably positive for ? smooth muscle actin.
In a study of 21 cases of equine giant cell tumor of soft parts,2 tumors with a morphology comparable to the present case were described. The tumor cells were immunohistochemically positive for vimentin and negative for cytokeratin, smooth-muscle actin, CD3, CD79a, CD31 and desmin. CD18 expression was detected only in the MGCs in the tumors, and it was suggested that the MGCs represent a secondary non-neoplastic cellular population.
It is suggested that within the undifferentiated pleomorphic sarcoma group there is a tumor of primitive myofibroblasts origin in dogs and cats that is analogous to the entity malignant fibrous histiocytoma (MFH) in humans.5 The fibroblastic/myofibroblastic cells in MFH stain positive for vimentin and variably for actin and desmin, and the MGCs should show the same positivity as the fibroblastic cells.5 This was however not the case in the present tumor, where desmin and actin negative MGCs were present between desmin and smooth muscle actin positive spindle shaped cells.
Specific antibodies for further differentiation between myofibroblasts and smooth muscle cell differentiation or origin were not available. Thus a distinction between a myofibroblastic fibrosarcoma with MGCs or a leiomyosarcoma with MGCs could not be made. In human leiomyosarcomas, a giant cell variant is recognized, and in these tumors the MGCs are proposed to be osteoclast-like cells.3
Contributing Institution:
Norwegian University of Life Sciences, Faculty of Veterinary Medicine www.nmbu.no
JPC Diagnosis: Subcutis (presumptive): Undifferentiated pleomorphic sarcoma with giant cells.
JPC Comment: The
contributor has provided a concise review of this tumor which has been shrouded
in controversy for many years. It has appeared twice in the WSC in the
subcutis of the horse (WSC 1994-1995, Conf 20 Case 3 and WSC 1991-1992 Conf 3,
Case 4). Two submissions of malignant fibrous histiocytoma in the skin of the
rat have also been submitted (WSC 1976-1977, Conf 29 Case 1 and WSC 1987-1988,
Conf 21, Case 4.)
Giant cell tumors of soft parts have been described in a number of species,
including baboons, Syrian hamsters, cats, and in a mule, as well as humans.2
A recent publication describes them in rabbits.1 In the rabbit,
they most often arise in the subcutaneous tissues as well. In humans, they may
be classified as superficial (affecting the skin and subcutis) or deep
(affecting tendons, fascia, and skeletal muscles of the thighs). Deep tumors
often recapitulate giant cell tumors of bone, and are positive for CD68 and
tartrate-resistant acid phosphatase (TRAP) and occasionally positive for smooth
muscle actin (as seen in this case.)1
One of the major differentials
for this neoplasm, both from a morphologic and a immunohistochemical
standpoint, is histiocytic sarcoma. In the study of 21 horses with this
neoplasm, 19/21 horses demonstrated CD18 positivity within the multinucleated
giant cell population, but not in the spindle cell population.2 For
this reason, several authors have posited that the multinucleated cell
population may be a reactive population of histiocytic cells in a spindle cell
neoplasm2,5, but exactly why they arise in a neoplasm with a fibroblastic/myofibroblastic
phenotype is yet unclear.
A recent report of a tumor of this type in a Warmblood horse discusses another
variant of this tumor, which has years ago been included under the umbrella
term "giant cell tumor of soft parts" but more recently termed "benign giant
cell tumor of tendon sheath".7 This particular variant has been
reported in dog, cats and horses. Oher authors believe this this neoplasm
actually represents a form of pigmented villonodular synovitis, as it often
arises in or near joints and shares a common feature of numerous
hemosiderin-laden macrophages.7 A case of pigmented villonodular
synovitis in a reticulated giraffe appeared in the WSC 2015-2016 as Conference
9, Case 1, and the case discussion reflects a similar controversy of nature and
origin.
The immunohistochemistry reported
by the contributor in this case is also somewhat problematic, as the 21 cases
in the retrospective by Bush et al. were negative for smooth muscle
actin and desmin, but this particular neoplasm demonstrated patchy
immunopositivity. This is in agreement that the current thinking that this
neoplasm, which has gone under many names over the years, is not a single
neoplasm with different variants, but a collection of several neoplasms which
are histologically and immunohistochemically diverse.2,5 Consultation
with a veterinary pathologist with extensive experience in similar tumors
suggested an alternative theory on the origin of these lesions, in that they
may be reparative in nature rather than neoplastic (personal communication, L. Craig,
University of Tenn.) A similar morphology is seen in peripheral giant cell
granulomas in the oral cavity of the dog and cat, that of a biphasic population of
spindle and multinucleate cells within a background of granulation tissue. (See
WSC 2017-2018 Conf 11, Case 4). While the origin of these tumors is not yet
elucidated in veterinary species, human pathologists have considered peripheral
giant cell granulomas to represent a reparative rather than neoplastic response.
References:
1. Bertram, CA, Garner MM, Reavill D, Klopfleisch R, Kiupel M. Giant cell sarcomas in domestic rabbits (Oryctolagus cuniculus). Vet Pathol 2020; DOI: 10.1177/0300985820921814
2. Bush JM, Powers BE. Equine giant cell tumor of soft parts: a series of 21 cases (2000-2007). J Vet Diagn Invest. 2008;20:513-516.
3. Gibbons CLMH, Sun SG, Vlychou M. et al. Osteoclast-like cells in soft tissue leiomyosardomas. Virchows Arch. 2010;456:317-323.
4. Gore MR. Giant cell rich malignancies: A metaanalysis. Ann Med Health Sci Res. 2018;8:225-232
5. Hendrick MJ. Mesenchymal tumors of the skin and soft tissues. In: Meuten DJ, ed. Tumors in domestic animals. 5th ed. Ames, Iowa, USA: Wiley Blackwell; 2017:142-175.
6. Mauldin E, Peters-Kennedy J. Integumentary system. In: Maxie MG, ed. Jubb, Kennedy, and Palmer?s pathology of domestic animals. Vol 1. 6th ed. St. Louis, Missouri, USA: Elsevier; 2016:509-736.
7. Zimmerman K, Almy F, Saunders G, Crisman M, Leonardi, L. An unusual case of giant cell tumor of soft parts in an American Wamrmblood horse. Open Vet J 2019, 9(1):44-48.