Signalment:
Gross Description:
Histopathologic Description:
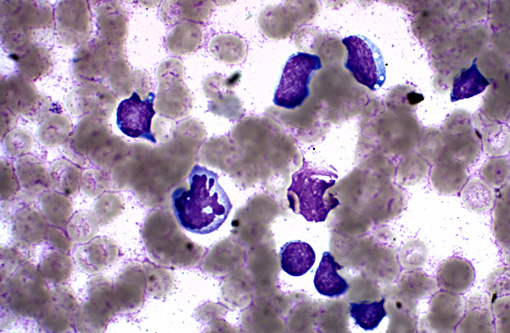
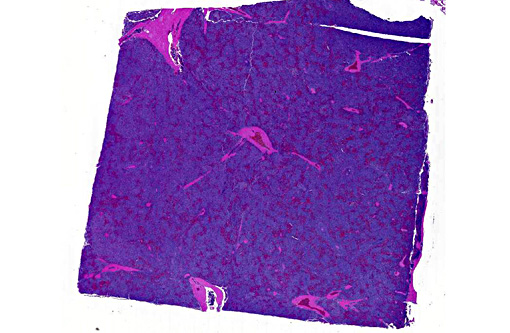
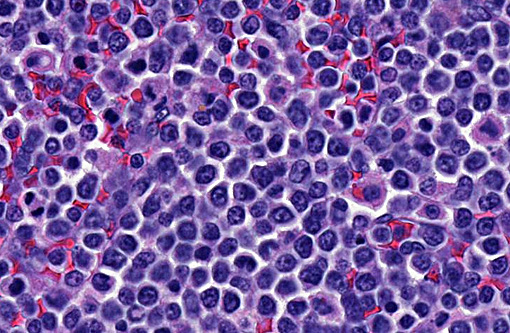
Spleen: In several sections of the spleen, the white and red pulps were completely replaced by a highly cellular mass. The mass was composed of round lymphoid cells arranged in sheets and supported by a delicate fibrovascular stroma. The neoplastic cells had distinct margins with scant to moderate amounts of eosinophilic cytoplasm. Nuclei were round to pleomorphic, had a finely clumped chromatin pattern, and 1 to 2 nucleoli. The nuclear diameter of the neoplastic cells was equal to the diameter of two regional erythrocytes. Mitotic figures ranged from 5 to 12 in ten 400x random fields. The tumor was not immunophenotyped.
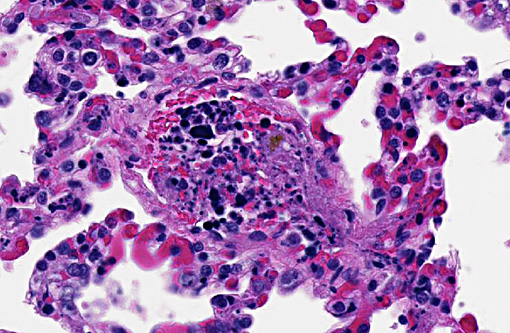
Lungs: Within the lumen of some of the small-size pulmonary arteries, there were clusters of neoplastic lymphoid cells and karyorrhectic debris. Fibrin thrombi were present in many pulmonary vessels.
Neoplastic infiltrates were also present in the bone marrow, liver, heart and multiple visceral lymph nodes (not shown).
Morphologic Diagnosis:
1) Multicentric lymphoblastic lymphosarcoma.
2) Intravascular chromatin emboli compatible with tumor lysis syndrome.
Lab Results:
The total WBC on this slide was 85, 600/microliter with lymphocytes composing 83,032/microliter of the total, neutrophils were at 2568/microliter. The HCT was decreased at 32% (38-56) and the platelets were decreased, 58,000/microliter (150,000-500,000).
K+ 6.1 (4-5.4)
Phosph 12.4 (2.2-6.4)
Creatinine 2.1 (0.7-1.6)
BUN 109 (9-26)
Condition:
Contributor Comment:
Clinical signs, which can include vomiting, lethargy, respiratory distress and cardiac arrest, are related to the massive release of intracellular purines, phosphorus, uric acid, potassium and lactate.(1) Common clinicopathologic abnormalities include hyperphosphatemia, hyperkalemia and acidosis. Individuals with renal insufficiency may be at greater risk for ATLS (due to reduced ability to clear metabolites from the blood) and ATLS patients are also at risk for developing renal failure if realkalinization of blood leads to calcium and phosphorus precipitation in the kidney.(2)
ATLS has been reported in dogs(5), a cat(1) and in mice with experimentally induced lymphoid malignancies.(6) The histologic lesion of ATLS, large chromatin emboli in blood vessels, especially in the lung, was recently described in a 129/SvEv mouse with experimentally induced acute myeloid leukemia that had been treated with valproic acid, a histone deacetylase inhibitor known to produce tumor lysis.(6)
In dogs, ATLS has been associated with dramatic reduction in tumor mass.(5) In this case, the dog had severe clinical signs and may have been manifesting ATLS, DIC or both at the time of presentation. However, both chemotherapeutic agents the dog received have been associated ATLS. Although no dramatic reduction in tumor size was noted clinically or pathologically, there was necrosis of tumor cells in the spleen, and the characteristic histologic lesions in the lung indicate that ATLS was most likely at least partly responsible for the dog's demise.
JPC Diagnosis:
1. Lung, Spleen: Lymphoma, intermediate to large cell, high grade.
2. Lung, septal capillaries: Neoplastic thrombi with necrosis.
Conference Comment:
Lymphoblastic lymphoma (LBL) is a diffuse lymphoma characterized by a dispersed chromatin pattern which obscures nuclear detail, and thus has indistinct nucleoli. Of note, the term lymphoblast has been commonly misused in veterinary medicine. By definition, the lymphoblasts of LBL are intermediate-sized cells and not the large lymphocytes seen in cases of diffuse large B cell lymphoma or peripheral T cell lymphoma. LBL can look very similar to hepatosplenic lymphoma (HS-TCL) and both are comprised of T-cells. HS-TCL is centered on the liver and spleen; however, it occurs without significant lymph node involvement which was identified in this case.(4) Additionally, another distinct entity in the liver has been proposed called hepatocytotropic lymphoma (HC-TCL) for those subtypes that are not confined to the sinusoids but rather invade hepatic cords.(4) All three discussed subtypes, (LBL, HS-TCL, HC-TCL) are associated with a poor prognosis in dogs.(4,7)
References:
1. Calia CM, Hohenhaus AE, et al. Acute tumor lysis syndrome in a cat with lymphoma. J Vet Intern Med. 1996;10:409-411.
2. Fleming DR, Doukas MA. Acute tumor lysis syndrome in hematologic malignancies. Leuk and Lymphoma. 1992;8:315-318.
3. Howard SC, Jones DP, Pui CH. The tumor lysis syndrome. N Engl J Med. 2011;364(19):1844-1854.
4. Keller SM, Vernau W, Hodges J, et al. Hepatosplenic and hepatocytotropic T-cell lymphoma: two distinct types of T-cell lymphoma in dogs. Vet Pathol. 2013;50(2):281-290.
5. Myonakis ME, Koutinas AF, et al. Acute tumor lysis syndrome in a dog with B cell lymphoma. Austr Vet J. 2007;85:206-208.
6. Radaelli E, Marchesi F, et al. Diagnostic excercise: Sudden death in a mouse with experimentally induced acute myeloid leukemia. Vet Pathol. 2009;46:1301-1305.
7. Valli VE, Kass PH, Myint MS, Scott F. Canine lymphomas: Association of classification type, disease stage, tumor subtype, mitotic rate, and treatment with survival. Vet Pathol. 2013;50(5):738-748.